Introduction
What is Stevens-Johnson syndrome (SJS) and toxic epidermic necrolysis (TEN)?
It is an adverse cutaneous drug reaction and a complex immunological complex also, involving at least 2 mucous membranes.1, 2 It is called erythema multiforme major and causes acute blistering. The skin detachment is <30% in SJS and more extensive in TEN.2
What are the causes of Stevens-Johnson syndrome (SJS) and toxic epidermic necrolysis (TEN)?
The commonest causes are listed below:
- Infections: Herpes simplex virus (HSV), Mumps
- Drugs: large list including NSAIDs, Antibiotics, anticonvulsants4
- Genetic: seems to be more common in certain populations like East Asian
What is the pathogenesis of Stevens-Johnson syndrome (SJS) and toxic epidermic necrolysis (TEN)?
It is not clearly understood. However, recently there is increased understanding in the apoptotic pathway of the keratinocytes and the immunological changes and the processes that lead to this adverse drug reaction.4
Symptoms of Stevens-Johnson Syndrome (SJS) and Toxic Epidermic Necrolysis (TEN)
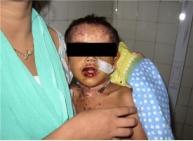
It is often misdiagnosed and begins with fever, sore throat like a prodrome. It is characterized by flat, atypical, target lesions and epidermal detachment is <10% of the total body surface area.1 Corneal and conjunctival sequelae are common.2 (figure 1 and 2)
Figure 1: Resolving Steven Johnson syndrome
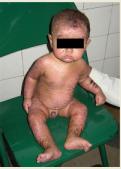
Figure 2: Steven Johnson syndrome involving the mucus membranes and skin
Treatment
It is considered a dermatological emergency and is treated aggressively. It had high mortality and morbidity.2, 4 Supportive care in the form of IV fluids and other management similar to that of a burn's patient is given. No certain line of therapy exists and various treatment options are available. The specific infections need specific antibiotics (like diagnosed mycoplasma needs doxycycline). Various drugs like prednisolone and cyclosporine have been used to halt disease progression.5 Anti TNF-alpha drugs like infliximab have shown positive results.5 Off label use of high dose, IVIG has been reported with certain benefits.5However, its use as an accepted line of therapy is still uncertain.3 Plasmapheresis seems to be a good alternative treatment option.5
1. Sharma VK, Sethuraman G, Minz A. Stevens Johnson syndrome, toxic epidermal necrolysis and SJS-TEN overlap: a retrospective study of causative drugs and clinical outcome. Indian J Dermatol Venereol Leprol. 2008; 74: 238-240.
2. De Rojas MV, Dart JK, Saw VP. The natural history of Stevens Johnson syndrome: patterns of chronic ocular disease and the role of systemic immunosuppressive therapy. Br J Ophthalmol 2007; 91: 1048?1053.
3. Metry DW, Jung P, Levy ML. Use of Intravenous Immunoglobulin in Children with Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis: Seven Cases and Review of the Literature. Pediatrics. 2003; 112: 1430-1436.
4. Yamane Y, Aihara M, Ikezawa Z. Analysis of Stevens-Johnson syndrome and toxic epidermal necrolysis in Japan from 2000 to 2006. Allergol Int. 2007; 56: 419-425.
5. Teo L, Tay Y K, Liu T T, Kwok C. Stevens-Johnson syndrome and toxic epidermal necrolysis: efficacy of intravenous immunoglobulin and a review of treatment options. Singapore Med J 2009; 50: 33.