Kevin Immanuel1, Gartika Sapartini2, Reni Ghrahani2, Budi Setiabudiawan2.
1Faculty of Medicine, Universitas Padjadjaran, Bandung, Indonesia,
2Department of Child Health, Faculty of Medicine, Universitas Padjadjaran, Dr. Hasan Sadikin General Hospital, Bandung, Indonesia.
ADDRESS FOR CORRESPONDENCE
Gartika Sapartini, Buah Batu Regency Blok D4 No.6 , Bandung, Indonesia, 40287.
Email: gartika.sapartini@unpad.ac.id | | Abstract | Aim
To understand the comparison of MexSLEDAI in pediatric lupus nephritis treatment with CYC and mmf after induction period.
Methods
This was a cohort retrospective study on pediatric patients with lupus nephritis treated with CYC and mmf. The study was conducted at the Dr. Hasan Sadikin General Hospital, Bandung, with retrospective data collection of pediatric lupus nephritis patients for the period 2017 to 2021. Characteristics, clinical manifestations, MexSLEDAI scores and treatment protocol were taken from medical record just before the start of protocol administration (0 months) and after the induction period (≥6 months). Statistical analysis using Man-Whitney U test and chi-squared tests to compare MexSLEDAI scores with mid-clinical parameters of pediatric lupus nephritis patients between CYC and mmf after induction period.
Results
Of 33 pediatric lupus nephritis patients, 25 received CYC and 8 received mmf. Statistically, no significant difference in clinical parameters was found between the CYC and mmf after the induction phase (all p > 0.05). Both protocol groups showed a decreased in median MexSLEDAI scores after the induction phase, 4 (1.5–7) for CYC and 6.5 (1.75–9.25) for mmf, but there was no significant difference between the two protocol groups (p = 0.39).
Conclusion
There were no significant differences in MexSLEDAI scores between CYC and mmf during the induction phase between the two protocol groups. | | | | Keywords | Cyclophosphamide, Lupus Nephritis, MexSLEDAI, Mycophenolate Mofetil.
Abbreviations
Cyclophosphamide (CYC), Disease Activity Index (DAI), Lupus Nephritis (LN), Mexican Systemic Lupus Erythematosus Disease Activity Index (MexSLEDAI), Mycophenolate Mofetil (MMF), Systemic Lupus Erythematosus (SLE), Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) | | | | Introduction | The incidence of systemic lupus erythematosus (SLE) generally occurs in girls aged 13 years and above. Renal involvement occurs in 3 out of 4 pediatric SLE patients and more than 90% develop lupus nephritis (LN) within 2 years after the child has been diagnosed with SLE.1,2 In assessing disease activity, determining treatment plans and response to therapy in SLE patients, a score called the disease activity index (DAI) is needed. Management of SLE and LN patients in Indonesia is guided by the DAI based on SLEDAI/MexSLEDAI.1
In this study, MexSLEDAI has been used because this parameter has several advantages, such as simple scoring and that it can be used in retrospective research.3 Determination of the treatment plan in pediatric SLE and LN patients is based on the results of the evaluation of the DAI. The options are the CYC and the mmf protocols. These protocols were administered for 36 months. The MexSLEDAI scoring was performed at early and mid-therapy stages. Early assessment was conducted before the patient had been given the protocol. The mid-assessment was carried out at ≥6 months, which was the induction phase, to determine the success of the protocol.4,5,6
Until now, no comparison study has been conducted in Indonesia to establish the effects of MexSLEDAI in pediatric lupus nephritis treatment with CYC and mmf after induction period. Therefore, this study was carried out to add new information regarding the comparison of MexSLEDAI in pediatric lupus nephritis treated with CYC and mmf after induction period.
| | | | Methods & Materials | The research was conducted at Dr. Hasan Sadikin General Hospital, Bandung, with retrospective data collection, using secondary data obtained from medical records of patients who reported to the Division of Allergy and Immunology. This research was conducted from October to December 2021. The study used analytical research methods with research subjects in the form of medical record data for the years 2017 to 2021. Characteristics, clinical manifestations, MexSLEDAI scores and treatment protocol were taken from medical record data just before the start of protocol administration (0 months) and after induction period (≥6 months). MexSLEDAI scores ranging from 0-32. There are several parameters used in MexSLEDAI with the score such as neurological problems (8), kidney disorders (6), vasculitis (4), haemolysis/thrombocytopenia (3), myositits (3), arthritis (2), mucocutaneous disorders (2), serositis (2), fever/fatigue (1), leucopenia/lymphopenia (1). To assess the degree of disease in systemic lupus erythematosus (SLE) over the past 10 days, clinicians can use the Mexican Systemic Lupus Erythematosus Disease Activity Index (MEX-SLEDAI). The total MEX-SLEDAI score ranges from 0 to 32. Scores of 0-1 indicate remission or inactive lupus, 2–5 reflect mild disease activity, 6–9 indicate moderate activity, 10–13 represent severe activity, and scores of ≥14 indicate very severe lupus activity.1 Laboratory testing included routine blood tests and urinalysis, but no renal biospies done.
The cyclophosphamide protocol is administered over a 36-month period. It begins with monthly pulses of intravenous methylprednisolone at a dose of 30 mg/kg/day for three consecutive days, given every month for the first 6 months. This is followed by daily oral corticosteroids throughout the initial 6-month period, and then continued daily for every 3-month interval up to month 36. In addition, cyclophosphamide is administered at a dose of 500–750 mg/m² once monthly for the first 6 months. Afterward, the dosage schedule is adjusted to 500–750 mg/m² once every 3 months until the 36th month of therapy.4
The mycophenolate mofetil (MMF) protocol is administered over a 36-month treatment period. Therapy begins with monthly intravenous methylprednisolone pulses at a dose of 0.5–1 g/m²/day for three consecutive days during the first 6 months. This is followed by daily oral prednisone at 0.5–1 mg/kg/day throughout the initial 6 months, which is then tapered to <0.5 mg/kg/day. The reduced daily prednisone dose (<0.5 mg/kg/day) is continued until the 36th month of therapy. Concurrently, oral mmf is given at a dose of 1,200 mg/m²/day during the first 6 months. After this induction phase, the dose is reduced to 600 mg/m²/day, continued daily through month 36 as the maintenance regimen.5,6
Statistical analysis using the Man Whitney non-parametric comparison test method for median (interquartile range) and chi-squared for frequency (percentage) were used to compare MexSLEDAI scores and mid-clinical parameters of pediatric lupus nephritis patients between CYC and mmf after induction period. | | | | Results | Characteristics of pediatric lupus nephritis patients (Table 1), results of the comparison study of clinical parameters (Table 2), and MexSLEDAI scores (Table 3) in pediatric lupus nephritis patients treated between CYC and mmf protocols can be seen in full in the table provided.
Table 1. Early Characteristics of Pediatric Lupus Nephritis Patients.
| Variable |
CYC |
MMF |
P-value |
| (n=25) |
(n=8) |
| Age (year) |
14.3 (11.9-15.9) |
12.5 (9.5-14.5) |
0.18 |
| Height (cm) |
151 (144.5-155.3) |
142.5 (128.1-148.6) |
0.04 |
| Weight (kg) |
49.4 (45-58.5) |
41 (29.2-52.7) |
0.03 |
| Gender, n(%) |
|
|
|
| Male |
1 (4) |
1 (12.5) |
0.38 |
| Female |
24 (96) |
7 (87.5) |
|
| MEXSLEDAI score, n(%) |
|
|
|
| 0-1 |
1 (4) |
0 (0) |
0.37 |
| 2-5 |
3 (12) |
0 (0) |
|
| 6-9 |
5 (20) |
4 (50) |
|
| 10-13 |
13 (52) |
4 (50) |
|
| =14 |
3 (12) |
0 (0) |
|
Table 2. Comparison of Early and Mid-therapy Clinical Parameters.
| Variable |
0 month (early) |
P-value |
>6 month (mid) |
P-value |
| CYC (n=25) |
MMF (n=8) |
CYC (n=25) |
MMF (n=8) |
| Oedema, n(%) |
5 (20) |
1 (12.5) |
0.63 |
0 (0) |
0 (0) |
N/A |
| Haemoglobin (g/dL) |
10.8 (9.7-12) |
11.1 (10-13.4) |
0.42 |
12 (10.9-12.9) |
11.7 (11-13) |
0.9 |
| Leukocytes (/mm³) |
12.2 (7.7-16.1) |
7.5 (4.9-12) |
0.08 |
8.3 (6.3-10.2) |
11.3 (5.8-14.1) |
0.29 |
| Platelets (/mm³) |
301 (213.5-395.5) |
284 (241.5-320.3) |
0.61 |
335 (230-411) |
321.5 (249-418) |
0.87 |
| Lymphocytes (/mm³) |
1.9 (1.1-2.7) |
1.5 (1.1-1.8) |
0.45 |
1.3 (0.8-2) |
1.5 (0.9-2.8) |
0.6 |
| Urinalysis |
|
|
|
|
|
|
| Leukocytes (per HPF) |
3 (1-7.5) |
1.5 (1-6) |
0.37 |
3 (1-5.5) |
1.5 (0-5.3) |
0.22 |
| RBC (per HPF) |
10 (3-40) |
37.5 (4-50) |
0.25 |
1 (0-7.5) |
6.5 (2.5-9.5) |
0.09 |
| Cylinder, n(%) |
10 (40) |
1 (12.5) |
0.15 |
0 (0) |
0 (0) |
N/A |
| Qualitative urine protein, n(%) |
|
|
|
|
|
|
| 0 |
5 (20) |
1 (12.5) |
0.41 |
17 (68) |
5 (62.5) |
0.61 |
| +1 |
4 (16) |
2 (25) |
|
3 (12) |
2 (25) |
|
| +2 |
3 (12) |
0 (0) |
|
3 (12) |
0 (0) |
|
| +3 |
10 (40) |
2 (25)) |
|
1 (4) |
1 (12.5) |
|
| +4 |
3 (12) |
3 (37.5) |
|
1 (4) |
0 (0) |
|
Table 3. MexSLEDAI Changes in Early and Mid-therapy Assessment.
| Time (month) |
CYC
(n=25) |
MMF
(n=8) |
P-value |
| MexSLEDAI |
|
|
|
| 0 (early) |
10 (7-12) |
8.5 (7-12) |
0.74 |
| >6 (mid) |
4 (1.5-7) |
6.5 (1.75-9.25) |
0.39 |
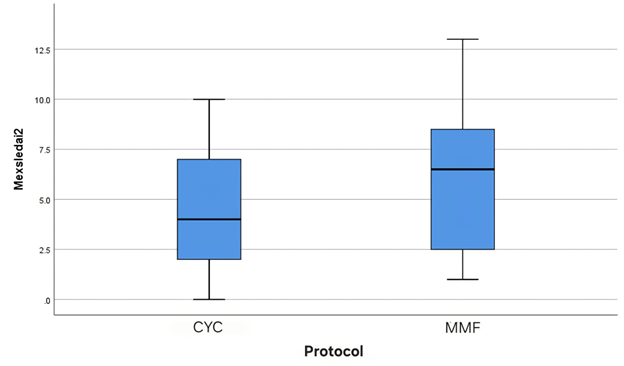
In this study, data were obtained from 33 pediatric lupus nephritis patients, 25 patients receiving the CYC protocol and 8 patients receiving the mmf protocol with ages ranging from 8.8 to 17.6 years. The ratio of women and men was 15.5:1 (Table 1). Among the 33 clinical parameter data of the pediatric lupus nephritis patients taken immediately before administration of the (0 month) CYC and mmf protocol, 5 (20%) and 1 (12.5%) patients had oedema and 10 (40%) and 1 (12.5%) patients, respectively, were detected as having cylindrical cells on urinalysis examination. By the middle of the CYC and mmf protocol, both complaints had disappeared 100%. The number of patients who developed proteinuria (+1 to +4) at the start of the CYC and mmf protocol was 20 (80%) and 7 (87.5%), respectively, which was then reduced to 8 (34%) and 3 (37.5%). Statistically, no significant differences in mid-clinical parameters were found in pediatric lupus nephritis patients between CYC and mmf (all p > 0.05, Table 2). Seen in the boxplot section, the median (interquartile range) MexSLEDAI 1 (0 month) score of pediatric lupus nephritis patients between CYC and mmf was 10 (7-12) and 8.5 (7-12) (Figure 1). When MexSLEDAI was calculated mid-protocol, the median (interquartile range) score decreased to 4 (1.5–7) in the CYC protocol and 6.5 (1.75–9.25) in the mmf protocol (Figure 2). Both protocol groups showed a decrease in MexSLEDAI scores in the induction phase, but when viewed statistically, there was no significant difference between the two protocol groups (p = 0.39, Table 3).
Figure 1. Early MexSLEDAI Comparison.
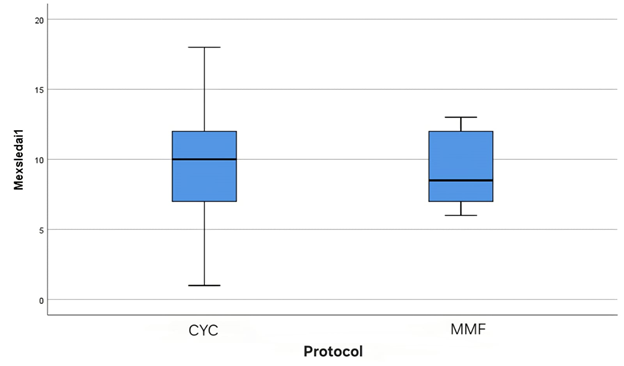
Figure 2. Mid-therapy MexSLEDAI Comparison.
 | | | | Discussion | This was a retrospective cohort study and aimed to compare the MexSLEDAI scores of pediatric lupus nephritis patients in Indonesia at induction period between CYC and mmf. In this study, improvements were found in MexSLEDAI scores and clinical parameters when compared at mid-treatment between the two protocol groups. The two groups showed similar effectiveness, but when compared statistically between them, there was no significant difference in the clinical parameters of the patients (all p >0.05). Tests on clinical parameters were carried out in addition and all clinical parameters checked affected the scoring process. This study did not include quantitative urine protein data, serum urea, serum creatinine or serum albumin. This happens because the data are only listed just before the administration of the protocol (0 month) which is the parameter for diagnosing SLE. In the comparison of MexSLEDAI scores in pediatric lupus nephritis patients, which was the main objective of this study, there was no significant difference in the scores between the CYC and the mmf groups (p = 0.39).
The use of MexSLEDAI in studies comparing the efficacy between the two protocol groups is very rare, in contrast to the SLEDAI parameter that is often found in several studies comparing the therapeutic efficacy of the protocol.7,8,9,10 In a study comparing low-dose CYC and mmf in India, improvements in SLEDAI scores and clinical parameters were found at 24 weeks of therapy.7 The median value of the SLEDAI score at week 24, found in the study of Manis Rathi et al. (2016), were 6 (4-8) and 4 (2-6) in the CYC and mmf groups, respectively.7 In our study, the median MexSLEDAI scores (≥6 months) were 4 (1.5-7) and 6.5 (1.75-9.25) in the CYC and mmf groups. When compared statistically, there was no significant difference between the two protocol groups after mid-therapy (≥6 months), between our study (p = 0.39) and that of Manis Rathi et al. (p = 0.17).7 The same result was also found in the study of Satish Mendonca et al. (2017) (p = 0.14) where there was no significant difference in SLEDAI scores ≥6 months between CYC and mmf.8
In our study, positive clinical parameter responses were found between the two protocol groups after induction period. Improvements in clinical parameters were reflected in the number of patients who were subjected to oedema and urine cylinder examination, which was 0 after the induction period for both protocol groups. Improvements were also found in the calculation of the number of mid-treatment patients who met the negative/0 quantitative proteinuria criteria, as shown in Table 2, 68% and 62.5% for CYC and mmf, respectively. The same findings were stated in several studies conducted in India by Satish Mendonca et al., Nepal by Arun Sedhain et al. (2018), and South Korea by Y. B. Joo et al. (2018), which was reflected in the improvement of quantitative urine protein (grams/24 hours) after the induction period.8,9,10
This is the first study conducted in Indonesia to compare MexSLEDAI scores in pediatric lupus nephritis patients with CYC and mmf protocol after the induction period. The study has several limitations, such as the number of pediatric lupus nephritis patients, the degree of patient's illness at the beginning, the duration of illness, patients who have received immunosuppressant drugs before being given CYC or mmf, and medical record data that are not centralised and therefore take a long time to collect. | | | | Conclusion | | In conclusion, from the results of this study, it can be seen that both protocols showed an improvement in the mid-MexSLEDAI score compared to the baseline. There was no statistically significant difference between the CYC and mmf protocols. In the results of the comparison of clinical parameters, there was also no significant difference between the two protocols. Both protocols showed the same efficacy in improving clinical parameters. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Indonesian Rheumatology Association. Diagnosis dan pengelolaan lupus eritematosus sistemik. Perhimpunan Reumatologi Indonesia.; 2019. pp. 1-129.
- Pinheiro SV, Dias RF, Fabiano RC, Araujo SD. Pediatric lupus nephritis. Brazilian Journal of Nephrology. 2018 Nov 14;41:252-65. [CrossRef] [PubMed] [PMC free article]
- Ohmura K. Which is the best SLE activity index for clinical trials?. Modern Rheumatology. 2021 Jan 2;31(1):20-8. [CrossRef] [PubMed]
- Lehman TJ, Onel K. Intermittent intravenous cyclophosphamide arrests progression of the renal chronicity index in childhood systemic lupus erythematosus. The Journal of pediatrics. 2000 Feb 1;136(2):243-7. [CrossRef] [PubMed]
- Hahn BH, Mcmahon MA, Wilkinson A, Wallace WD, Daikh DI, Fitzgerald JD, Karpouzas GA, Merrill JT, Wallace DJ, Yazdany J, Ramsey‐Goldman R. American College of Rheumatology guidelines for screening, treatment, and management of lupus nephritis. Arthritis care & research. 2012 Jun;64(6):797-808. [CrossRef] [PubMed] [PMC free article]
- Hajizadeh N, Laijani FJ, Moghtaderi M, Ataei N, Assadi F. A treatment algorithm for children with lupus nephritis to prevent developing renal failure. International journal of preventive medicine. 2014 Mar;5(3):250.
- Rathi M, Goyal A, Jaryal A, Sharma A, Gupta PK, Ramachandran R, Kumar V, Kohli HS, Sakhuja V, Jha V, Gupta KL. Comparison of low-dose intravenous cyclophosphamide with oral mycophenolate mofetil in the treatment of lupus nephritis. Kidney international. 2016 Jan 1;89(1):235-42. [CrossRef] [PubMed]
- Mendonca S, Gupta D, Ali S, Gupta P. Mycophenolate mofetil or cyclophosphamide in indian patients with lupus nephritis: Which is better? A single-center experience. Saudi Journal of Kidney Diseases and Transplantation. 2017 Sep 1;28(5):1069. [CrossRef] [PubMed]
- Sedhain A, Hada R, Agrawal RK, Bhattarai GR, Baral A. Low dose mycophenolate mofetil versus cyclophosphamide in the induction therapy of lupus nephritis in Nepalese population: a randomized control trial. BMC nephrology. 2018 Dec;19(1):1-8. [CrossRef] [PubMed] [PMC free article]
- Joo YB, Kang YM, Kim HA, Suh CH, Kim TJ, Park YW, Lee J, Lee JH, Yoo DH, Bae SC, Lee HS. Outcome and predictors of renal survival in patients with lupus nephritis: Comparison between cyclophosphamide and mycophenolate mofetil. International journal of rheumatic diseases. 2018 May;21(5):1031-9. [CrossRef] [PubMed]
DOI: https://doi.org/10.7199/ped.oncall.2027.24
|
| Cite this article as: | | Immanuel K, Sapartini G, Ghrahani R, Setiabudiawan B. Comparison of Mexican Systemic Lupus Erythematosus Disease Activity Index pediatric lupus nephritis with Cyclophosphamid and Mycophenolate mofetil after induction period. Pediatr Oncall J. 2026 Mar 12. doi: 10.7199/ped.oncall.2027.24 |
|