Sivalingam Pavithra, Srinivasan Kavitha, Annamalai Odayappan.
Glaucoma Services, Aravind Eye Hospital, Pondicherry, India.
ADDRESS FOR CORRESPONDENCE
Dr. Srinivasan Kavitha, Head of the Department, Glaucoma services, Aravind Eye Hospital, Cuddalore main road, Thavalakuppam, Pondicherry- 605007
Email: skavitha.shree@gmail.com | | Abstract | | An asymptomatic 6-year-old boy was identified in our school screening camp with defective vision in both eyes and referred to us for refractory error evaluation. He was found to have corneal edema as a consequence of very high intraocular pressure (IOP) in both the eyes. On further evaluation, no congenital ocular anomalies which could lead to IOP elevation were found. He had earlier been diagnosed with acute lymphoblastic leukemia at 3 years of age and was on repeated cycles of systemic steroid therapy. Consequent to this, the child had developed raised IOP and steroid-induced secondary open-angle glaucoma with irreversible damage of the optic nerve in both eyes. He was started on topical anti-glaucoma medications and his IOP was brought under control with no worsening of optic nerve damage. This report highlights the possibility of steroid-induced glaucoma in the pediatric age group due to systemic steroid therapy and the necessity for timely referral to the ophthalmologist to avoid needless and irreversible blindness due to glaucoma. | | | | Keywords | | glaucoma, steroids, raised intraocular pressure | | | | Introduction | Glaucoma is a chronic, progressive, multifactorial optic neuropathy which damages the optic nerve with resultant loss of visual function. Worldwide, glaucoma is the second major cause of blindness after cataract with more than 60 million affected individuals.1 It is one of the most common causes of irreversible blindness. The most important modifiable risk factor for glaucoma is raised intraocular pressure (IOP). In the early stage of the disease, the peripheral field of vision is affected where the patient is asymptomatic. The central field of vision gets involved in the advanced stage, the patient becomes symptomatic and appreciates visual disturbance. At this stage, by controlling the IOP we can only prevent the progression of the disease, but the impaired visual component can never be restored.
Steroid-induced glaucoma is secondary glaucoma which can occur in any age group following corticosteroid administration by any route (topical, peri-ocular or systemic). However, the onset of rise in IOP can vary from within a few hours of steroid administration to many years of chronic steroid use. Unmonitored IOP elevation can lead to optic neuropathy resulting in steroid-induced glaucoma. It is reported that about 30% of the population will demonstrate a rise in IOP of 6−15 mmHg following 4−6 weeks of topical steroid administration and in 5% of the population IOP may rise by more than 16 mmHg.2 Studies describing systemic steroid-induced glaucoma have mostly focused on adults with only a few reports in pediatric patients3,4,5, although it constitutes one-fourth of all acquired glaucoma in children.6
Our report emphasizes the need for periodic ocular evaluation when a higher dose or longer duration of systemic corticosteroids are warranted in treating life-endangering conditions like acute lymphoblastic leukemia, to prevent irreversible blindness.
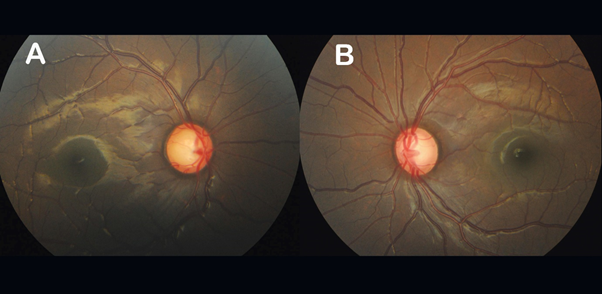
| | | | Case Report | A 6-year-old boy was identified at one of our school screening ophthalmic camps with defective vision in both eyes. He was referred to our tertiary eye hospital for further evaluation. His uncorrected visual acuity was 6/9 in the right eye, 6/12 in the left eye. General examination showed good build, nourishment for age, he weighed 23 kg. Facial puffiness was noted. Ocular examination showed corneal haze due to edema, normal anterior chamber depth with a clear lens in both eyes and an ill-sustained pupillary reaction in the left eye. IOP was 58 mmHg in the right eye and 64 mmHg in the left eye (Normal range: 10-21 mmHg). Gonioscopy, which is the study of the outflow pathway of aqueous humour, revealed open angles in both eyes. Fundus examination showed damaged optic nerve head in both eyes with an increased cup to disc ratio (CDR) of 0.8 (Normal CDR <0.6) with superior neuro-retinal rim thinning in the right eye and a CDR of 0.7 with bipolar neuro-retinal rim thinning in the left eye (Figure A & B respectively). Optical coherence tomography showed a marked decrease in the average retinal nerve fibre layer thickness in both eyes. Based on the above findings, he was diagnosed to have open-angle glaucoma in both eyes.
Looking at the unusual facial puffiness when probed for more details about his past medical history, it was found that the boy was diagnosed with B-cell acute lymphoblastic leukemia (ALL) at 3 years of age. He was on chemotherapy (UK-ALL MRC PROTOCOL REGIMEN - B) which he had been taking for 2 years and 4 months along with systemic steroids in repeated cycles, from the beginning of his treatment. At the time of presentation to our eye institute, he was on maintenance chemotherapy which included oral mercaptopurine 50 mg daily, oral methotrexate 15 mg per week, intravenous vincristine 1.2 mg once a month and oral prednisolone 30 mg per day for 1 week every month apart from vitamin supplementation and cotrimoxazole prophylaxis. He had also received intrathecal methotrexate during his induction remission phase and prophylactic cranial radiotherapy once in his course of treatment.
The child was started on maximal topical medical therapy - latanoprost 0.004% eye drops once at night and a fixed combination of brimonidine 0.1 % with brinzolamide 1% eye drops thrice a day in both eyes to bring down the IOP. On follow up, after 3 weeks of treatment with the above-mentioned anti-glaucoma medications and temporary withdrawal of steroids as per his routine chemotherapy protocol, the IOP reduced to 23 mmHg in the right eye and 20 mmHg in the left eye. Corneal edema had cleared and uncorrected visual acuity improved to 6/6 in both eyes. The treating oncologist was requested to consider the possibility of withdrawing steroid therapy in view of his high IOP and optic nerve damage. Following bone marrow aspiration study which showed a reversal of myeloid, erythroid ratio (1:2) with less than 5% blasts and cerebrospinal fluid cytology being negative for malignant cells, the treating oncologist then withdrew oral steroid therapy completely. His IOP was well under control in his subsequent follow-up visits with anti-glaucoma medications. On the last follow up almost 2 years after the initial presentation and 1 year after completion of chemotherapy, his IOP was 14 mmHg in the right eye and 18 mmHg in the left eye. His uncorrected visual acuity was 6/6 in both eyes. The fundus examination was stable with no worsening of optic nerve damage. At present, he is maintaining well on the same 3 topical anti-glaucoma medications (latanoprost, brimonidine, and brinzolamide eye drops) and being followed up at 4 monthly intervals.
Figure A. Damaged optic nerve head in right eye with a cup to disc ratio of 0.8 with superior neuro-retinal rim thinning. Figure B. Damaged optic nerve head in left eye with a cup to disc ratio of 0.7 with bipolar neuro-retinal rim thinning.
| | | | Discussion | Glucocorticoid is an essential component of ALL treatment in stabilizing the disease. Our case report highlights the importance of timely intervention in a child with steroid induced glaucoma, a potentially blinding disease. Glaucoma is a highly asymptomatic disease often referred as “silent thief of sight”, since the damaged nerve does not regenerate. Moreover, glaucoma has asymmetrical presentation in that the
advanced field defects of adversely affected eye can be overlapped by relatively normal field of less affected eye. The affected children can remain asymptomatic despite very high IOP till the late stage of disease, which necessitates periodical monitoring.
Liebreich in 1861 first described the ophthalmological signs in patients with leukemia as leukemic retinopathy.7 Typical ocular manifestations of leukemic retinopathy are round or flame-shaped hemorrhages with a white component, intra-retinal hemorrhages, and cotton wool spots. Most of the ocular manifestations occur secondary to direct leukemic infiltration, hematologic abnormalities and opportunistic infections.8 Rowan et al described that high IOP can occur in ALL patients due to infiltration of the trabecular meshwork9, but this is difficult to demonstrate in vivo due to its confounded anatomical position. In our patient, apart from raised IOP and optic nerve head cupping, the history of use of corticosteroids, absence of congenital ocular anomalies indicated it to be a steroid-induced glaucoma. In addition, the IOP spikes reduced when steroids were withdrawn. In our literature search, we could not find any clinical report on anti-cancer drugs like vincristine, mercaptopurine or methotrexate causing IOP spikes and open angle glaucoma. Reports of ALL patients presenting with ocular signs as the initial manifestation of the disease are rare since the systemic diagnosis is established prior to the ocular manifestation in most cases.10,11
Yamashita et al conducted a retrospective review of 5 paediatric patients with ALL who received chemotherapy with repetitive cycles of systemic corticosteroids. They were followed up until the final cycle of chemotherapy and symmetrical IOP rise higher than 21 mmHg was observed in all patients.3 When the IOP alone rises, they are labelled as “steroid responders”, but some children, as in our case, react adversely to such raised IOP and develop irreversible damage to the optic nerve and asymptomatic blinding.
High dose of systemic steroids is prescribed for a wide range of systemic disorders. Hayasaka et al reported that 20% of paediatric patients with nephritic syndrome on corticosteroids developed IOP higher than 22 mmHg.4 Tripathi et al reported increased IOP in paediatric patients with inflammatory bowel disease after oral corticosteroid treatment.5 These reports stress the importance of IOP monitoring in children whenever maintained on steroid therapy.
As the survival rate of childhood ALL reaches above 85% with current therapy12, there is increased concern on reducing treatment related toxicity as some of the consequences like steroid induced glaucoma could be irreversible, leading to total blindness with devastating impact on the quality of life in the survivor. In our case, with prompt intervention, although total blindness was prevented, the probable field defects due to the existing optic nerve damage were irreversible. Automated visual field testing which gives the magnitude of visual field defects, however, is not possible in very young children since it requires a good understanding and co-operation. We could not analyse the visual fields in our patient.
Glaucoma as a cause of childhood blindness is rare as compared to adults, but with increasing use of corticosteroids for various systemic conditions, quite a high prevalence of steroid induced ocular side effects like ocular hypertension, steroid induced glaucoma and cataract are widely being reported. It is an iatrogenic preventable disease which responds well to withdrawal of steroids and medical treatment. Generally, steroid-induced acute rise of IOP normalizes within 1 to 4 weeks of stopping steroids. But sometimes steroid-induced IOP rise may progress to glaucoma with permanent visual field loss, even after the cessation of steroid therapy though the IOP returns to normal range.13 In such cases and also when IOP remains persistently high, anti-glaucoma medications or surgery may become necessary.
| | | | Conclusion | | Although steroid induced glaucoma is a well-known reported entity, the number of periodical referrals for ocular examination by the treating physician is quite less. We emphasize that physicians should be aware of glaucoma as a possible sight threatening complication which can lead to total blindness when untreated following any form of steroid therapy. Unmonitored use of steroids should be discouraged and a switch over to alternative treatment is preferred whenever possible. But in warranted conditions where one cannot avoid steroid therapy, close monitoring of IOP and prompt intervention becomes mandatory irrespective of the nature of underlying disease since the ‘vision once lost is lost forever’ in glaucoma. | | | | Contributor Statement | PS conceptualized the study, drafted the initial manuscript. KS critically reviewed the manuscript for important intellectual content and approved the final version to be published. AO conceptualized the study, coordinated the study and revised the manuscript.
All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Saxena R, Singh D, Vashist P. Glaucoma: An emerging peril. Indian J Community Med. 2013;38:135-7 [CrossRef] [PubMed] [PMC free article]
- Armaly MF. Statistical attributes of the steroid hyper-tensive response in the clinically normal eye. Invest Ophthalmol. 1965;4:187-97
- Yamashita T, Kodama Y. Steroid-induced Glaucoma in Children with Acute Lymphoblastic Leukemia a possible complication. J Glaucoma. 2010;19:188-190 [CrossRef] [PubMed]
- Hayasaka Y, Hayasaka S, Matsukura H. Ocular findings in Japanese children with nephrotic syndrome receiving prolonged corticosteroid therapy. Ophthalmologica. 2006;220:181-5 [CrossRef] [PubMed]
- Tripathi RC, Kipp MA, Tripathi BJ, Kirschner BS, Borisuth NS, Shevell SK, Ernest JT. Ocular toxicity of prednisone in pediatric patients with inflammatory bowel disease. Lens Eye Toxic Res. 1992;9:469-82
- Kaur S, Dhiman I, Kaushik S, Raj S, Pandav SS. Outcome of ocular steroid hypertensive response in children. J Glaucoma. 2016;25:343-347 [CrossRef] [PubMed]
- Guyer DR, Schachat AP. Relationship between Fundus Lesions and Hematologic Parameters at Diagnosis. Ophthalmology. 1989;96:860-4. [CrossRef]
- Rosenthal AR. Ocular manifestations of leukemia: a review. Ophthalmology 1983;90:899-905 [CrossRef]
- Rowan PJ, Sloan JB. Iris and anterior chamber involvement in leukemia. Ann Ophthalmol. 1976;8:1081-5
- Reddy SC, Menon BS. A prospective study of ocular manifestations in childhood acute leukaemia. Acta Ophthalmol Scand. 1998;76:700-3 [CrossRef] [PubMed]
- Kim J, Chang W, Sagong M. Bilateral serous retinal detachment as a presenting sign of acute lymphoblastic leukemia. Korean J Ophthalmol. 2010;24:245-8 [CrossRef] [PubMed] [PMC free article]
- Pui CH, Campana D, Pei D, et al. Treating childhood acute lymphoblastic leukemia without cranial irradiation. N Engl J Med. 2009; 360:2730-2741 [CrossRef] [PubMed] [PMC free article]
- Sihota R, Konkal VL, Dada T, Agarwal HC, Singh R. Prospective, long-term evaluation of steroid-induced glaucoma. Eye (Lond). 2008;22:26-30. [CrossRef] [PubMed]
DOI: https://doi.org/10.7199/ped.oncall.2020.9
|
| Cite this article as: | | Pavithra S, Kavitha S, Odayappan A. Steroid Induced Glaucoma in a Child with Acute Lymphoblastic Leukemia – An Overlooked Complication. Pediatr Oncall J. 2020;17: 21-23. doi: 10.7199/ped.oncall.2020.9 |
|