Andreas Chiabi1, Walter Dobgima Pisoh2, Flavila Tchoupou Tsayim1, Moses Samje1, Evelin Feuldi3, Frida Sunjo3, Daniel Armand Kago Tague4, Denis Nsame Nforniwe3.
1Faculty of Health Sciences, University of Bamenda, Northwest Region, Cameroon,
2Faculty of Health Sciences, University of Bamenda/Bamenda Regional Hospital, Cameroon,
3Bamenda Regional Hospital, Cameroon,
4Faculty of Medicine and Biomedical Sciences, University of Yaoundé I, Cameroon.
ADDRESS FOR CORRESPONDENCE
Prof Chiabi Andreas, Faculty of Health Sciences, University of Bamenda, Cameroon.
Email: andy_chiabi@yahoo.co.uk | | Abstract | Aim: To assess the incidence of perinatal asphyxia, its risk factors and outcome at a single tertiary referral center in Cameroon.
Methods: This was a hospital-based prospective case-control study on term neonates delivered at tertiary referral center over three months. Cases were defined as neonates with an Apgar score less than 7 at the 5th minute. Controls were neonates with an Apgar score greater than or equal to 7 at the 5th minute, matched to cases for gestational age and sex. Maternal socio-demographic, antenatal, intrapartum, and neonatal variables including delivery outcome, Sarnat's score, time of death if it occurred, and duration of admission were noted. Risk factors for perinatal asphyxia were determined.
Results: Of 740 live births during the study period, 72 (9.7%) neonates had perinatal asphyxia and most of the asphyxiated neonates were males (sex ratio 1.4), 86.1% weighed between 2500-3999g, and 42 (58.3%) mothers were in the age group between 18-29 years. Following multivariate analysis, risk factors for birth asphyxia included: were single mothers [AOR=3.9993;95% CI: 1.1176-14.3110) p=0.0331], mothers who had urinary tract infection in pregnancy [AOR=15.6325;95% CI: 2.3083-105.8688) p=0.0048], prolonged rupture of membranes [AOR=6.2537;95% CI: 1.1744-33.3013) p=0.0317], prolonged labor [AOR=3.6521;95% CI: 1.0940-12.1918) p=0.0352] and mothers who delivered by emergency caesarian section [OR=8.0169;95% CI: 2.3669-27.1537), p=0.0008]. Among the asphyxiated neonates, 15 died with a case fatality of 20.8%. Fourteen (93.3%) of those who died had severe asphyxia. Eighty-eight percent of asphyxiated neonates developed hypoxic ischemic encephalopathy, of which, 54.7% were Sarnat’s stage I, 15.6% stage II and 29.7% stage III.
Conclusion: The incidence and case fatality of perinatal asphyxia are high. Early diagnosis, prompt management of pathological disorders during pregnancy and labor, and neonatal resuscitation could largely prevent perinatal asphyxia. | | | | Keywords | | Perinatal asphyxia, incidence, associated risk factors, Outcome. | | | | Introduction | Perinatal asphyxia is defined by the World Health Organization (WHO) as the failure to initiate and sustain breathing at birth.1 It is a common and serious neonatal problem globally and it significantly contributes to both neonatal morbidity and mortality.2 It was caused by maternal and fetal factors.3 Perinatal asphyxia occurs when an inadequate amount of oxygen is delivered to the fetus, usually during labor and childbirth.4 The maternal factors can further be divided into antepartum and intrapartum factors. Some antepartum risk factors are increasing or decreasing maternal age, place of antenatal visit, booking status, pre-eclampsia, malaria during pregnancy and primigravida.3,4 Intrapartum risk factors are breech presentation, prolonged labor, stationary labor, term prolonged rupture of membranes, caesarean section, home delivery and maternal fever and fetal risk factors are resuscitation of child, preterm babies, fetal distress, and low birth weight.3,4 In Cameroon in general and in our context, there is not much data available on the incidence, risk factors and hospital outcome of perinatal asphyxia despite its high contribution to neonatal morbidity and mortality, hence we undertook this study to evaluate its incidence, risk factors and hospital outcome so that interventions and management strategies could be undertaken.
Methods
This study was a hospital-based case control study with prospective collection of data carried out from the 1st of November 2017 to the 30th of April 2018 at the Delivery room and Neonatology unit of the Bamenda Regional Hospital (BRH), involving all neonates born at term at BRH. This hospital serves as a reference health facility for mother and child health care. Ethical clearance was obtained from the ethical committee of the Faculty of Health Sciences of the University of Bamenda. Administrative authorization was obtained from the Regional Delegation of public health of the Northwest Region and the Director of the Bamenda Regional Hospital for carrying out this study.
The Kelsey and Fleiss formula was used to calculate the sample size taking into consideration meconium-stained amniotic fluid to be an important risk factor for birth asphyxia from a study by Chiabi et al in 2013.4 The minimum acceptable sample size for our study was 46 cases and 46 controls. The study involved 72 cases and 72 controls after written informed consent. Cases included all term neonates with an Apgar score <7 while controls (matched for gestational age and sex) involved neonates with an Apgar score ≥7.
Term pregnant women admitted at the delivery room of the BRH were recruited once they accepted to participate in our study. The aim, objectives, procedures, and constraints of the study were explained to the women and their doubts clarified before signing the consent forms. Participants were interviewed and their responses noted on a structured data collection sheet. Data were collected on a wide range of risk factors including sociodemographic (maternal age, marital status, educational level and monthly allowance), antepartum history (parity, number of antenatal care (ANC) visits, place of ANC visit and maternal pathologies), intrapartum history (fetal presentation, premature rupture of membranes, prolonged rupture of membrane, color of amniotic fluid, prolonged active phase of labor, prolonged second stage of labor, place of delivery and method of delivery) and fetal factors (Apgar score, sex and weight of baby). Maternal occupation was classified as unemployed (housewife, student), salaried (civil servants), or non-salaried (hairdressers, tailors, traders). Parity was classified as primiparous (had delivered one time), multiparous (had delivered two to four times) and grand-multiparous (had delivered greater than or equal to five times). Prolonged labor was defined as labor lasting more than 12 hours in primiparous women or more than 8 hours in multiparous women.6 Prolonged second stage of labor was defined as delivery after more than 2 hours of full dilatation.7 Premature rupture of membranes (ROM) was defined as rupture of the membrane of the amniotic sac and chorion that occurred more than one hour before the onset of labor.8 Prolonged ROM was defined as when the duration is more than 18 hours prior to delivery.9 After delivery, an Apgar score of 0-3 was classified as severe birth asphyxia and a score 4-6 was classified as moderate asphyxia.3 These neonates were examined taking into consideration 6 clinical categories; level of consciousness, activity, neuromuscular control, presence of primitive reflexes (Moro and sucking), autonomic function (pupils and heart rate) and seizures, to classify the neonates under the modified Sarnat’s classification.5 This classification helped to know if the baby developed hypoxic ischemic encephalopathy (HIE) which is one of the early complications of perinatal asphyxia. A neonate was classified as having severe HIE if he/she had any of the following: coma or stupor, absent activity, absent reflexes, flaccid muscle tone, and generalized seizures particularly of early onset.5 He/she was classified as having moderate HIE if he/she had any of the following: lethargy or obtundation, decreased activity, mild hypotonia, multifocal seizures and weak reflexes.5 This assessment was done on each neonate before 24 hours after birth and every day until discharge.
Statistical Analysis
All data was entered and analyzed using Epi info version 7.2. Frequency and percentages were calculated for categorical data. Risk factors for perinatal asphyxia were grouped into antepartum, intrapartum, and fetal variables. Odds ratio (OR) and confidence interval at 95% were used to evaluate the degree of association between these variables and the risk of asphyxia. Multivariate analysis with logistic regression was carried out, to look for independent association. Threshold of significance was set at p<0.05.
| | | | Results | Seven hundred and sixty-two neonates were born in the maternity during the study period. Twenty-two of them were stillbirths and 740 were live births including 89 premature deliveries. Out of these, 72 were asphyxiated neonates giving an incidence of 9.7% (Figure 1).
Figure 1. Participants flow diagram.
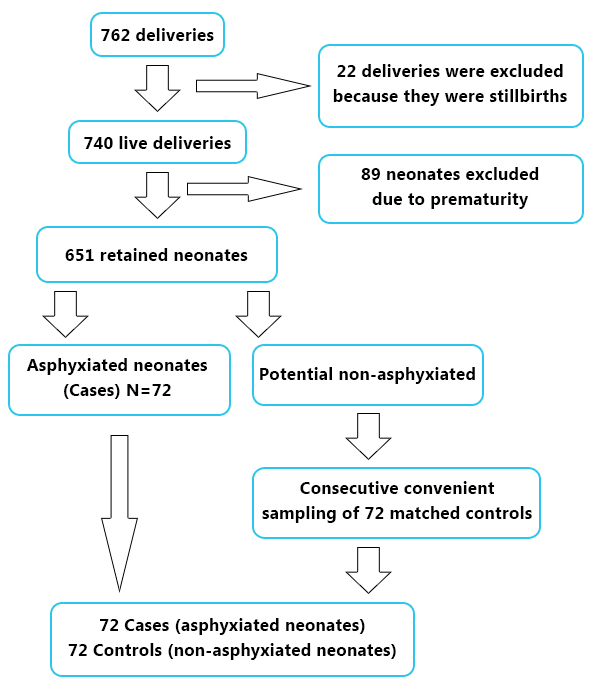
Out of the 72 asphyxiated neonates, 42 (58.3%) were males and 30 (41.7) were females with a male to female ratio of 1.4:1. Most neonates weighed between 2500 g- 3999 g (86.1%), while 9.7% weighed >4000 g and 4.2% weighed <2500 g. We noted that 38 (52.8%) of the neonates were born at gestational ages between 37 and 40 weeks. Maternal sociodemographic data, antepartum and intrapartum details are shown on table 1.
After bivariate analysis the factors that were with the occurrence of asphyxiated neonates were mothers who were; unemployed [OR 2.4462 (1.1944-5.008), p=0.0072], primiparous [OR 2.0800 (1.0388-4.1648) p=0.0198], grand multiparous [OR 2.4462 (1.194-5.008) p=0.007], who attended ANC at a health center [OR 2.5385 (1.252-5.126) p=0.0048], attended <4 ANC visits [OR 4.1814 (1.846-9.468) p=0.0002]. It also included the cases that came to the hospital directly (not referred) [OR 3.9565 (1.6945-9.2383) p=0.0005], had malaria during pregnancy [OR 3.9053 (1.933-7.887) p=0.0000] and had urogenital infections during pregnancy [OR 10.818 (2.396-48.831) p=0.0006]. It also included mothers who had; prolonged labor [OR 5.479 (2.596-11.567) p=0.0000], prolonged second stage of labor [4.688 (2.076-10.587) p=0.0000], prolonged ROM [OR 3.400 (1.254-9.216) p=0.0007] and delivered by emergency caesarean [OR 5.588 (2.578-12,111) p=0.0000]. (Table 1)
After bivariate analysis the factors that were against occurrence of asphyxiated neonates were mothers who were salaried [OR 0.4375 (0.714-1.099), p=0.0397], had a university level of education [OR 0.4088 (0.199-0.837), p=0.007], multiparous[OR 0.5077 (0.2610-0.9875) p=0.0237], had attended 4-7 ANC visits [OR 0.2800 (0.128-0.609) p=0.0005] and who had a vaginal delivery [OR 0.1402 (0.065-0.301) p=0.0000]. This also included neonates with birth weight 2500-4000 g [OR 0.3766 (0.1359–1.0438), p=0.029]. (Table 1)
Table 1. Distribution of risk of asphyxia according to sociodemographic characteristics, antepartum factors, and maternal pathologies.
| Variables |
Asphyxiated
(N=72) |
Non asphyxiated
(N=72) |
Odd’s ratio 95% CI |
P value |
| Number (%) |
Number (%) |
| Maternal socio-demographic characteristics |
| Maternal age (years) |
|
|
|
|
| <18 |
9 (12.5) |
0 (0) |
Undefined |
0.00 |
| 18-29 |
42 (58.3) |
51 (70.8) |
0.5765 [0.288-1.150] |
0.60 |
| 30-39 |
21 (29.2) |
21 (29.2) |
1.0000 [0.487-2.052] |
0.500 |
| Occupation |
|
|
|
|
| Unemployed |
17 (23.6) |
31 (43.1) |
2.4462 [1.1944-5.008] |
0.0072 |
| Salaried |
8 (11.1) |
16 (22.2) |
0.4375 [0.714-1.099] |
0.0397 |
| Non-salaried |
45 (62.5) |
37 (51.4) |
1.5766 [0.811-3.063] |
0.0920 |
| Level of education |
|
|
|
|
| Primary |
10 (13.9) |
4 (5.6) |
2.7419 [0.818-9.190] |
0.050 |
| Secondary |
45 (62.5) |
37 (51.4) |
1.5766 [0.811-3.063] |
0.0920 |
| University |
17 (23.6) |
31 (43.1) |
0.4088 [0.199-0.837] |
0.007 |
| Antepartum characteristics |
| Parity |
|
|
|
|
| Primiparous |
32 (44.4) |
20 (27.8) |
2.0800 [1.0388-4.1648] |
0.0198 |
| Multiparous |
33 (45.8) |
45 (62.5) |
0.5077 [0.2610-0.9875] |
0.0237 |
| Grand multiparous |
17 (23.6) |
31 (43.1) |
2.4462 [1.194-5.008] |
0.007 |
| Place of ANC |
|
|
|
|
| Health center |
54 (75.0) |
39 (54.2) |
2.5385 [1.252-5.126] |
0.0048 |
| Regional hospital |
18 (25.0) |
33 (45.8) |
|
|
| Type of pregnancy |
|
|
|
|
| Singleton |
63 (87.5) |
67 (93.1) |
1.9143 [0.608-6.022] |
0.1393 |
| Multiple |
9 (13.5) |
5 (6.9) |
|
|
| Number of ANC visits |
|
|
|
|
| <4 |
29 (40.3) |
10 (13.9) |
4.1814 [1.846-9.468] |
0.0002 |
| 4-7 |
42 (58.3) |
60 (83.3) |
0.2800 [0.128-0.609] |
0.0005 |
| =8 |
1 (1.4) |
2 (2.8) |
2.028 [0.179-22.883] |
1.0000 |
| Mode of admission |
|
|
|
|
| Elective |
46 (63.9) |
63 (87.5) |
3.9565 [1.6945-9.2383] |
0.0005 |
| Referral |
26 (36.1) |
9 (12.5) |
|
|
| Maternal pathologies |
|
|
|
|
| Malaria |
42 (58.3) |
19 (23.4) |
3.9053 [1.933-7.887] |
0.0000 |
| Pre-eclampsia |
3 (4.2) |
2 (2.8) |
1.5217 [0.246-9.390] |
1.001 |
| Urogenital infections |
17 (23.6) |
2 (2.8) |
10.818 [2.396-48.831] |
0.0006 |
| Intrapartum factors |
| Prolonged labor |
41 (56.9) |
14 (19.4) |
5.479 [2.596-11.567] |
0.0000 |
| Prolonged second stage |
33 (45.8) |
10 (13.9) |
4.688 [2.076-10.587] |
0.0000 |
| Premature ROM |
17 (23.6) |
10 (13.9) |
1.9164 [0.809-4.534] |
0.0715 |
| Prolonged ROM |
17 (23.6) |
6 (8.3) |
3.400 [1.254-9.216] |
0.0007 |
| Mode of delivery |
|
|
|
|
| Emergency caesarean |
38 (52.8) |
12 (16.7) |
5.588 [2.578-12,111] |
0.0000 |
| Vaginal |
28 (38.9) |
59 (81.9) |
0.1402 [0.065-0.301] |
0.0000 |
| Instrumental delivery |
6 (8.3) |
1 (1.4) |
0.1549 [0.018-1,321] |
0.121 |
| Neonatal characteristics |
| Birth weight (kg) |
|
|
|
|
| <2500 |
5 (6.9) |
1 (1.4) |
0.6032-46.5392 |
0.2109 |
| 2500-4000 |
58 (80.6) |
66 (91.7) |
0.3766 [0.1359-1.0438] |
0.029 |
| >4000 |
9 (12.5) |
5 (6.9) |
0.5224 [0.1661-1.6433] |
0.1393 |
| Presentation |
|
|
|
|
| Cephalic |
65 (92.3) |
69 (95.8) |
0.403 [0.1001-1.6279] |
0.325 |
| Breech |
5 (6.9) |
2 (2.8) |
2.6119 [0.489-13.926] |
0.4383 |
| Transverse |
2 (2.8) |
1 (1.4) |
0.4930 [0.0437-5.5609] |
1.000 |
Note: ANC- antenatal center, ROM - rupture of membranes
After multivariate analysis with logistic regression the risk factors for birth asphyxia were single mothers [AOR=3.9993;95% CI: 1.1176-14.3110) p=0.0331], mothers who had; urinary tract infection in pregnancy [AOR=15.6325;95% CI: 2.3083-105.8688) p=0.0048], prolonged ROM [AOR=6.2537;95% CI: 1.1744-33.3013) p=0.0317], prolonged labor [AOR=3.6521;95% CI: 1.0940-12.1918) p=0.0352] and mothers who delivered by emergency caesarian section [OR=8.0169;95% CI: 2.3669-27.1537), p=0.0008].
Out of the 72 cases of asphyxiated neonates, 57 (79.2%) were discharged alive, with 46 (63.9%) having no complications, and 11 (15.3%) with persistent neurologic deficits, while 15 (20.8%) died. Out of the 15 deaths, 14 (93.3%) had an Apgar score of <3. Among those who had an Apgar score between 4 and 6, 42 (97.7%) were discharged alive. Fifty (69.4%) of the asphyxiated neonates, stayed less than 7 days in the hospital, and 18 (25.0%) between 7-10 days, and 4 (5.6%) more than 10 days. Amongst the 72 cases of asphyxia observed in our study, 64 (88.9%) neonates developed HIE among which, 35 (54.7%) had mild HIE, 10 (15.6%) had moderate HIE and 19 (29.7%) had severe HIE. | | | | Discussion | In our study, incidence of perinatal asphyxia was 9.73% (97.29‰). This incidence was slightly lower than the 112‰ obtained by Douba6 in 2001 but higher than the 86.9‰ obtained by Tchouankeu.7 It was also higher than 80.6‰ obtained by Chiabi et al4 in 2013 and 82.96‰ obtained by Zukou8 in 2017. Other researchers in Africa have reported lower rates 4.7‰ by Padayachee et al9 and 33.2‰ by Ugwu et al.10 Other studies reported incidences higher than ours: 100.2‰ by Ogunlesi et al11 and 206‰ by Drama et al.12 Out of Africa, the incidence observed in our study is very high compared to 9.7‰ obtained by Lee et al13 in 2008 in Southern Nepal and 5.4‰14 obtained in Sweden in 2008. Variations could be due to the context of the studies and methodologies used.
Majority (58.33%) of the asphyxiated neonates were males and the sex ratio was 1.4. Douba6, Monebenimp et al5 and Padayachee et al9 (58.9%) have also described this. Shankaran16 established that the male sex (57.1%) was statistically significant compared to the female sex. Another study rather found a female predominance without a statistically significant difference; 59.6% for Ilah et al.17 According to Hill et al18, the sex discrepancy may be due to sex differences in the preferred mechanisms of apoptosis, the presence of sex specific hormones or the protective effect of a family of proteins called inhibitors of apoptosis on the caspase-dependent apoptotic pathway. In addition, according to Johnston et al.19 female sex hormones (estrogens) enhance the protection against anoxo-ischemic lesions.
Majority (80.6%) of the asphyxiated neonates weighed between 2500-3999g. This is similar to 83.93% observed by Zukou8 but higher than 77.80% observed by Chiabi et al4 in the same country. Our result is also similar to the 80.3% observed by Ibrahim et.al.20 Most of the asphyxiated neonates had gestational ages between 37 and 42 weeks. Same observation was made by Zukou8 and Chiabi et al4 in Cameroon and Dramé et al in Senegal.12 The reason why this group was more affected remains unknown.
Most of the mothers of the asphyxiated neonates were aged between 18 and 30 years. The same observation was made by Chiabi et.al4 and Zukou in 2017 in Cameroon8, Ogunlesi et al11, Ibrahim et.al20 and Dramé et al.12 The possible explanations for this could be due to pelvic disproportion which is more common in women of this age group, and it will make deliveries more difficult and prolonged.20
Those that were single were four times more at risk to give birth to an asphyxiated child than those who were living as a couple. This result corroborates with those obtained by Raatikainen et al in Finland21, Torres-munoz et al in Colombia22 and Chiabi et al4 while other authors did not find an association between single marital status and perinatal asphyxia.3,23,24 A possible explanation for this could be that single women generally lack financial and emotional support, hence do not promptly and regularly attend antenatal consultations.
Women with urinary tract infection (UTI) during pregnancy had a fifteen-time risk to bear an asphyxiated neonate. Our result corroborates with what was obtained by Hall et al25, Kaye24, Dramé et al12 and Saeed Zaman et.al.26 This could be explained by the fact that UTI activate the host monocyte-macrophage system and the cytokines released during this process signal the initiation of contractions by stimulating prostaglandin biosynthesis by intrauterine tissues27, these contractions will decrease the amount of oxygen to the fetus and thus will lead to asphyxia.
Prolonged ROM increased the risk to be asphyxiated by six. This same observation was made by Zukou8 and Chiabi et al4 in Cameroon, Lee et.al in Nepal13, and Kardana et.al in Indonesia28 while Kaye in Uganda24 and Dramé et al in Senegal12 instead had premature rupture of membranes as a risk factor of perinatal asphyxia. The most common complications of prolonged rupture of membrane are amnionitis and endometritis, both put the fetus at a high risk of developing an over whelming infection in the bloodstream (sepsis) which will lead to hypoxia and thus perinatal asphyxia.28
Women with prolonged labor were four times at risk to bear an asphyxiated neonate than the others. Ugwu et.al in 201210, Chiabi et.al4, Tabassum et al29, Ilah et.al in 201517 and Kardana et al28 also had prolonged labor to be associated with perinatal asphyxia. Prolonged labor is mostly caused by inadequate contractions. Inadequate contractions are usually treated with oxytocin and misoprostol. These drugs can cause excessive contractions, which will prevent the placenta to supply oxygen adequately to the fetus30, this could lead to fetal hypoxia and thus perinatal asphyxia.
Most (89.47%) of the caesarean sections we had in our study were emergency caesarean sections. Neonates born by caesarean section were eight times more at risk to be asphyxiated than those born through the vagina were. Kaye24, Chiabi4, Ilah17 and Dramé et al12 also had a significant association between caesarean section and birth asphyxia. This could be explained by the fact that most of the indications for emergency cesarean sections were due to conditions that compromise adequate oxygen delivery to the fetus as acute fetal distress, cephalo-pelvic disproportions, and prolonged labor.4
In our study, out of the 72 asphyxiated neonates, 15 died giving a case fatality of 20.8%. Ninety-three per cent of those who died had severe asphyxia. This correlates with the fact that there is a direct relationship between the severity of asphyxia and death.31 This case fatality is similar to 16.07% observed by Zukou in Cameroon8 but however higher than other studies carried out in the same country; Douba6, had 4.2%; Monebenimp et al.15 had 5.4%, Chiabi et al4 had 6.7% and Tchouankeu et.al7 had 14%. Out of Cameroon, this case fatality was higher than 9.7 per 1000 observed by Lee et al13 but it was lower than 24.7% and 25.5% observed by24 and Ilah et al17 respectively.
Sixty-four out of 72 asphyxiated neonates developed HIE resulting in 88.88% rate of HIE. Most of them (54.69%) were in Sarnat’s stage I, 15.63% were in stage II and 29.69% were in stage III. Our incidence is similar to 71.73% obtained by Zukou in 2017.8 Other studies done in Africa: Cameroon4, South Africa9 and out of Africa: Sweden14 and India32 found lower rates of HIE 48.8%, 3.6 per 1000, 1.8 per 1000 and 1.9% respectively. This high rate observed in our study could be because BRH has a pediatrician, technical facilities and is the only tertiary hospital in the Northwest Region and as such receives a large number of high-risk referral cases.
| | | | Conclusion | | Perinatal asphyxia is high at the BRH as in other parts of the country. Risk factors were single mothers, prolonged rupture of membranes, prolonged labor, and urinary tract infection during pregnancy. We thus recommend that information, education, and communication to all pregnant women on the importance of antenatal visits should be reinforced to reduce the risk factors incriminated. Refresher courses for midwives and all health personnel working at the delivery room and in the neonatology unit on neonatal resuscitation should be initiated and reinforced to reduce the mortality from perinatal asphyxia. | | | | Acknowledgement | The authors are grateful to the entire staff of the maternity and nursery of the Bamenda Regional Hospital as well as the mothers who accepted to participate in this study.
Thanks to Drs Donfack Lorianne, Nini Yesih Ngwengi and Meten Elvis for reading and revising the manuscript. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Maternal and Newborn Health/Safe Motherhood Unit. Division of Reproductive Health (Technical Support). Basic Newborn Resuscitation: A Practical Guide, vol. 4. Geneva: World Health Organization; 1997. Accessed from: https://www.who.int/maternal_child_adolescent/
- UNICEF. Levels & Trends in Child Mortality. Report 2015. Available from: https://www.unicef.org/media/files/IGME_Report_Final2.pdf2015:1-34. Accessed 15 November 2019.
- Aslam HM, Saleem S, Afzal R, Iqbal U, Saleem SM, Shaikh MW, et al. Risk factors of birth asphyxia. Ital J Pediatr. 2014 Dec; 40:94. [CrossRef]
- Chiabi A, Nguefack S, Mah E, Nodem S, Mbuagbaw L, Mbonda E, et al. Risk factors for birth asphyxia in an urban health facility in Cameroon. Iran J Child Neurol. 2013;7:46-54.
- Shankaran, S. Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Fanaroff AA et al. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med. 2005; 353:1574-1584. [CrossRef]
- Douba EC. Souffrance cérébrale asphyxique du nouveau-né à terme au Centre Mère-Enfant de Yaoundé, Faculté de Médecine et de Sciences Biomédicales, Université de Yaoundé .2007.
- Tchouankeu FK. Asphyxie néonatale au Centre Mere et Enfant de la fondation Chantal Biya: aspects épidémiologiques, cliniques et évolutifs. [Medicinea Doctorae thesis]: Faculty of Medicine and Biomedical Sciences, University of Yaoundé 1. 2015.
- Zukou KV. Birth Asphyxia at the Laquintinie Hospital: Prevalence, Factors Associated and Early Outcome. [Medecinae Doctorae thesis]. University of Health Sciences, University of Buea, 2017.
- Padayachee N, Ballot DE. Outcomes of neonates with perinatal asphyxia at a tertiary academic hospital in Johannesburg, South Africa. SAJCH 2013; 7:89-94. [CrossRef]
- Ugwu GIM, Abedi HO, Ugwu EN. Incidence of Birth Asphyxia as Seen in Central Hospital and GN Children's Clinic both in Warri Niger Delta of Nigeria: An Eight Year Retrospective Review. Glob J Health Sci. 2012; 4: 140-146. [CrossRef]
- Ogunlesi T, Oseni S. Severe Birth Asphyxia in Wesley Guild Hospital, lesa: A persistent plague! Niger Med Pract. 2008;53 :40-43. [CrossRef]
- Dramé A, Iz C, Sylla A. Asphyxie périnatale au service de néonatologie de l'hôpital La Paix de Ziguinchor (Senegal). European Scientific Journal. 2017;13:217-226. [CrossRef]
- Lee AC, Mullany LC, Tielsch JM, Katz J, Khatry SK, LeClerq SC, et al. Risk factors for neonatal mortality due to birth asphyxia in southern Nepal: a prospective, community-based cohort study. Pediatrics. 2008; 121:e1381-1390. [CrossRef]
- Thornberg E, Thiringer K, Odeback A, Milsom I. Birth asphyxia incidence, clinical course and outcome in a Swedish population. Acta Paediatr. 1995;84:927-932. [CrossRef]
- Monebenimp F, Tietche F, Eteki N. Asphyxie néonatale au centre hospitalier universitaire de Yaoundé. Clin Mother Child Health. 2005;2:335-338.
- Shankaran S. Prevention, diagnosis, and treatment of cerebral palsy in near-term and term infants. Clin Obstet Gynecol. 2008;51:829-839. [CrossRef]
- Ilah BG, Aminu MS, Musa A, Adelakun MB, Adeniji AO, Kolawole T. Prevalence and Risk Factors for Perinatal Asphyxia as Seen at a Specialist Hospital in Gusau, Nigeria. Sub-Saharan Afr J Med. 2015;2:64-69. [CrossRef]
- Courtney A, Hill and Holly R. Sex differences in mechanisms and outcome of neonatal hypoxia-ischemia in rodent models: Implications for sex-specific neuroprotection in clinical neonatal practice. Neural Res Int. 2012; 2012:9. [CrossRef]
- Johnston MV, Hagberg H. Sex and the pathogenesis of cerebral palsy. Dev Med Child Neurol. 2007;49:74-78. [CrossRef]
- Ibrahim NA, Muhye A, Abdulie S. Prevalence of Birth Asphyxia and Associated Factors among Neonates Delivered in Dilchora Referral Hospital, in Dire Dawa, Eastern Ethiopia. Clin Mother Child Health.2017; 14: 1000279. [CrossRef]
- Raatikainen K, Heiskanen N, Heinonen S. Marriage still protects pregnancy. BJOG. 2005; 112:1411-1416. [CrossRef]
- Torres-Muñoz J, Rojas C, Mendoza-Urbano D, Marín-Cuero D, Orobio S, Echandía C. Risk factors associated with the development of perinatal asphyxia in neonates at the Hospital Universitario del Valle, Cali, Colombia, 2010-2011. Biomedica. 2017;37:51-56. [CrossRef]
- Lansac J, Body G, Berger C, Berland J, Bertrand J, Danan M, et al. Pratique de l'accouchement. Eur J Obstet Gynecol Reprod Biol.1992; 140-1.
- Kaye D. Antenatal and intrapartum risk factors for birth asphyxia among emergency obstetric referrals in Mulago Hospital, Kampala, Uganda. East Afr Med J. 2003;80:140-143. [CrossRef]
- Hall DR, Smith M, Smith J. Maternal factors contributing to asphyxia neonatorum. J Trop Pediatr. 1996;42:192-195. [CrossRef]
- Saeed Z, Sajid A, Shahid M, Sohail S, Misbah M, Ammara M. Birth Asphyxia in Resource Constrained Health Care Set Up. Pak Armed Forces Med J. 2017;67:971-975.
- Hurst J, Danchin A. Explanation of Benveniste. Nature. 2009:21402.
- Kardana IM. Risk factors of perinatal asphyxia in the term newborn at sanglah general hospital. Bali Med J. 2016;5:175-178. [CrossRef]
- Tabassum F, Rizvi A, Ariff S, Soofi S, Bhutta ZA. Risk Factors Associated with Birth Asphyxia in Rural District Matiari, Pakistan: A Case Control Study. Int J Clin Med. 2014; 5:1430-1441. [CrossRef]
- Vijai Anand Babu B, Shyamala Devi S, Kishore Kumar B. Birth asphyxia: incidence and immediate outcome in relation to risk factors and complications. Int J Res Heal Sci.2014; 2:1064-1071.
- Casey BM, McIntire DD, Leveno KJ. The continuing value of the Apgar score for the assessment of newborn infants. N Engl J Med. 2001;344:467-471. [CrossRef]
- Mundhra R, Agarwal M. Fetal outcome in meconium-stained deliveries. J Clin Diagn Res. 2013;7:2874-2876. [CrossRef]
DOI: https://doi.org/10.7199/ped.oncall.2021.45
|
| Cite this article as: | | Chiabi A, Pisoh W D, Tsayim F T, Samje M, Feuldi E, Sunjo F, Tague D A K, Nforniwe D N. Risk Factors of Perinatal Asphyxia and Neonatal Outcome. Pediatr Oncall J. 2021;18: 107-113. doi: 10.7199/ped.oncall.2021.45 |
|