Sara Completo1, Joana Gomes Vieira1, Patrícia Lipari Pinto1,2, Alexandra Dias2, Anna Sokolova3.
1Child and Youth Department, Hospital Prof. Doutor Fernando Fonseca, E.P.E., Amadora, Portugal,
2Pediatrics Department, Hospital de Santa Maria, Centro Hospitalar de Lisboa Norte, E.P.E., CAML, Lisbon, Portugal,
3Pneumology Department, Hospital Prof. Doutor Fernando Fonseca, E.P.E., Amadora, Portugal.
ADDRESS FOR CORRESPONDENCE
Sara Completo, Hospital Prof. Doutor Fernando Fonseca E.P.E . IC 19, 2720-276 Amadora.
Email: completo.sara@gmail.com | | Abstract | Biological drugs are becoming the standard of care in severe refractory allergic diseases. In the case of omalizumab, it is indicated for severe asthma. Some biological drugs have unexpected effects related to hematotoxicity.
Hereditary spherocytosis is a common cause of hereditary anemia, with heterogeneous manifestations influenced by multiple factors, as exposure to drugs. Our aim was to evaluate possible hematological effect of omalizumab in a case of hereditary spherocytosis.
A 10-year-old boy with hereditary spherocytosis and severe asthma was started on omalizumab due to several exacerbations and frequent need of oral corticosteroids, along with a decreased respiratory function. After six months, clinical improvement was observed. Simultaneously, there were no changes in hematological parameters.
We report the first case of treatment with omalizumab in a child with severe asthma and hereditary spherocytosis. In this study, this therapy appears to be safe in hemolytic anemia. | | | | Keywords | | Anemia, hemolytic, Asthma, Biological therapy, Hemolysis, omalizumab. | | | | Introduction | Omalizumab is a recombinant DNA-derived humanized monoclonal antibody indicated in Step 5 treatment of asthma.1,2,3,4,5,6 This biological drug has a specific connection to the constant region present on the surface of the molecule of Immunoglobulin E (IgE) - Cɛ3, which will block the binding to its receptors (both high and low affinity), decreasing IgE levels in the bloodstream. This process will reduce the expression of the specific IgE receptors on inflammatory cells, such as mast cells and basophils, decreasing the release of inflammatory and allergic mediators. omalizumab will act on dendritic cells as well, limiting the process of allergen presentation to T cells and consequently, the cascade of the allergic reaction.1,2
Some biological therapies have shown unexpected effects related to hematotoxicity, such as thrombocytopenia, de novo immune hemolysis or even the worsening of the previous anemia.7
Hereditary spherocytosis is a congenital, non-immune, hemolytic anemia, characterized by modifications of the red cell membrane. It is a common cause of hereditary anemia in children. Its clinical expression is heterogeneous, varying from silent chronic hemolysis to severe, transfusion-dependent forms. A broad range of factors can influence a worsening of the anemia, namely: infections, certain foods, deficiencies (vitamin B12, iron, folate) and oxidative drugs.8
There are no previous published data on the use of omalizumab in patients with hemolytic hereditary anemias.
This paper aimed to evaluate the possible effect of omalizumab therapy, directed to the treatment of severe asthma, in a child with concomitant hereditary spherocytosis, and to ascertain any effects on hemolysis, given the underlying hemolytic anemia of the patient.
For that, the authors collected clinical and laboratory data and compared them, before and after the periodic administration of omalizumab.
| | | | Case Report | A ten-year-old boy (weight 30 kg, height 137 cm), with spherocytosis and asthma. His hemolytic anemia was previously stable, with compensated hemolysis, moderate reticulocytosis and slightly increased bilirubin, without significat anemia [Hemoglobin 12.8 g/dL, hematocrit 35%, reticulocytes 239 000/uL (5.3%), total bilirubin 0.5 mg/dL, lactate dehydrogenase (LDH) 264 U/L, haptoglobin <10 mg/dL]. He did not have splenomegaly neither gallstones.
By the age of nine, he had been diagnosed with severe asthma (stage 4), with persistent symptoms and multiple exacerbations, despite being medicated with high dose of inhaled corticosteroids therapy and long-acting beta-agonist combination (budesonide 160 ug/dose and formoterol 4.5ug/dose, twice daily), plus inhaled budesonide (200 ug/dose) twice daily and nasal mometasone (50 ug/dose). He was also medicated with oral corticosteroid therapy during crises (maximum of 2mg/kg/day), which were becoming more frequent and more severe over time. By then, he had a reported forced expiratory volume in the first minute (FEV1) of 1.14L (63%) and forced vital capacity (FVC) of 1.6L (74%). He had several exacerbations in a short period of time, with need to resort to salbutamol several times in a week, and, therefore, he was started on omalizumab therapy. During the first six months, omalizumab was administered in the scheme of 300 mg intramuscular, bimonthly. Afterwards, it passed to 300 mg intramuscular each three weeks for a period of four months and then to 300 mg intramuscular with four weeks intervals, staying controlled with this dosage at the present time.
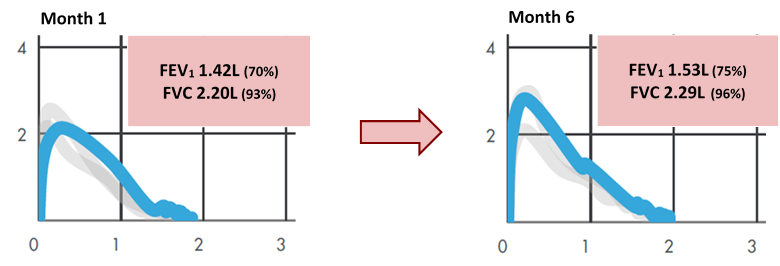
After only six months of treatment, there was a significant improvement of his respiratory symptoms and parameters in respiratory function tests, with FEV1 increasing at that time to 1.53L (75%) and FVC to 2.29L (96%), as can be observed in figure 1.
Figure 1. Respiratory function tests, before and after six months of Omalizumab therapy (adapted from "Aria: Spirometry & Lung Health, NuvoAir Home®").
He did not suffer any further significant exacerbations nor did he need systemic corticotherapy again. Moreover, both the child and his parents reported an improvement in physical capacity and tolerance in daily activities. There was no mention of weight gain besides the normal growth for the age.
Simultaneously, we observed no significant effect in either studied hemolytic parameters, even after corticotherapy was discontinued, with only minor modifications, as observed in table 1. Reticulocytes had a variation of 40%, which is considered as not significant. Every other parameters remained stable.
Table 1. Variation of hemolytic parameters before and after therapy (minimum and maximum values).
| Parameter |
Pre-therapy |
Post-therapy (minimum) |
Post-therapy (maximum) |
| Hemoglobin (gm/dl) |
12.8 |
12.5 |
13.2 |
| Reticulocyts [U/uL (%)] |
2,39,000 |
1,96,000 |
2,80,000 |
| Platelets [U/uL] |
3,25,000 |
2,76,000 |
3,59,000 |
| Haptoglobin [mg/dL] |
<10 |
<10 |
<10 |
| LDH (U/L) |
264 |
259 |
270 |
| | | | Discussion | The use of biological therapy has been increasing exponentially and it is an important weapon in the prevention and treatment of diseases refractory to standard treatments.5,6 In this case, consistently with literature, omalizumab showed to be extremely effective in improving severe asthma symptoms, even after a short period.1,2,3,5 There was a drastic reduction of asthma exacerbations and it allowed the reduction in the use of systemic corticotherapy as well.
As previously mentioned, some studies reported a possible hematotoxic effect of omalizumab.7 Our findings do not support this, as it did not appear to have any substantial effects on hemolysis in this case. This was documented and proved in laboratory analysis of hemolytic parameters, before and after this biological drug was administered. The child did not have any repercussions in his previous hematologic disease, maintaining the asymptomatic form of spherocytosis, with compensated hemolysis. Also, corticosteroid therapy withdrawal produced no effect on hemolytic parameters and, still, there was an improvement in respiratory symptoms.
| | | | Conclusion | This is the report of only one patient and more studies with larger samples are needed, in order to draw broader conclusions. Nevertheless, there are no other cases described of a patient suffering from spherocytosis, or other hemolytic hereditary anemias, treated with omalizumab. Additionally, it would be important to analyze the impact of omalizumab in a longer period.
To conclude, to the best knowledge of the authors, this is the first reported case of treatment with omalizumab in a child with hereditary spherocytosis and severe asthma. This study highlights omalizumab efficacy and safety in asthma control, and most of all, demonstrates no influence in hemolysis, opening the possibility that it could be safe in patients with hemolytic anemias. | | | | Acknowledgement | A written consent from the mother of the patient was obtained.
The authors declare no conflicts of interest. There was no funding for the study. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Licari A, Marseglia A, Caimmi S, Castagnoli R, Foiadelli T, Barberi S, et al. Omalizumab in Children. Pediatr Drugs. 2014; 16(6):491-502. [CrossRef] [PubMed] [PMC free article]
- Tadrous M, Khuu W, Lebovic G, Stanbrook M, Martins D, Paterson M, et al. Real-world health care utilization and effectiveness of omalizumab for the treatment of severe asthma. Ann Allergy, Asthma Immunol. 2018; 120(1):59-65. [CrossRef] [PubMed]
- Odajima H, Ebisawa M, Nagakura T, Fujisawa T, Akasawa A, Ito K, et al. Omalizumab in Japanese children with severe allergic asthma uncontrolled with standard therapy. Allergol Int. 2015; 64(4):364-370. [CrossRef] [PubMed]
- Odajima H, Ebisawa M, Nagakura, T, Fujisawa T, Akasawa A, Ito K, et al. Long-term safety, efficacy, pharmacokinetics, and pharmacodynamics of omalizumab in children with severe uncontrolled asthma. Allergol Int. 2017; 66(1):106-115. [CrossRef] [PubMed]
- Saji J, Yamamoto T, Arai M, Mineshita M, Miyazawa T. Efficacy of long-term omalizumab therapy in patients with severe asthma. Respir Investig. 2017; 55(2):114-120. [CrossRef] [PubMed]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, 2020, at www.ginasthma.org.
- Everds NE, Tarrant JM. Unexpected hematologic effects of biotherapeutics in nonclinical species and in humans. Toxicol Pathol. 2013; 41(2):280-302. [CrossRef] [PubMed]
- Konca Ç, Söker M, Taş MA, Yıldırım R. Hereditary Spherocytosis: Evaluation of 68 Children. Indian J Hematol Blood Transfus. 2015; 31(1):127-132. [CrossRef] [PubMed] [PMC free article]
DOI: https://doi.org/10.7199/ped.oncall.2022.44
|
| Cite this article as: | | Completo S, Vieira J G, Pinto P L, Dias A, Sokolova A. Omalizumab use in a patient with asthma and hereditary spherocytosis. Pediatr Oncall J. 2022;19: 131-133. doi: 10.7199/ped.oncall.2022.44 |
|