Nivedita Sharma, Piyush Gautam, RS Jaswal, Naveen Kumar.
Department of Pediatrics, Dr Rajendra Prasad Medical College and Hospital Tanda at Kangra, Himachal Pradesh, India.
ADDRESS FOR CORRESPONDENCE
Dr Nivedita Sharma, Senior Resident, Department of Pediatrics, Dr Rajendra Prasad Medical College and Hospital, Tanda at Kangra, Himachal Pradesh, India.
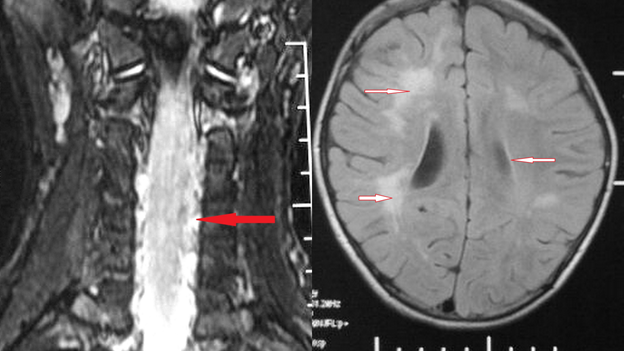
Email: niveditasharma2@yahoo.com | | Abstract | | Neuromyelitis optica (NMO) is an uncommon clinical syndrome in pediatric age group associated with unilateral or bilateral optic neuritis and transverse myelitis. NMO-IgG antibody estimation has broadened the spectrum of the disease. We report an 11 year old female with neuromyelitis optica following a flu-like syndrome with significant brain involvement at presentation. | | | | Keywords | | Devics, Neuromyelitis Optica, spectrum | | | | Introduction | | Neuromyelitis optica (NMO) is a devastating myelitis with an acute unilateral or bilateral optic neuropathy, no clinical involvement beyond the spinal cord or optic nerves, and a monophasic, rarely a multiphasic illness. (1,2) In most cases, involvement of the spinal cord and optic nerves occurs within 3 months of each other, although some authors have included patients with 2 or more years between these occurrences. (2) It is also called as Devic’s disease and is associated with good visual and neurological prognosis, without long term recurrence or sequelae. Once believed to be a variant of multiple sclerosis, newer diagnostic criteria make it a distinct syndrome. Detection of an antibody, NMO-IgG against aquaporin 4 protein helps in the diagnosis of disease. (3) | | | | Case Report | An 11 years old female presented with pain in the neck and limbs (lower limbs more affected than upper limbs), inability to stand and walk, urinary retention and dribbling for one day. There was a preceding history of fever and cough, one week prior to these complaints. There was history of headache associated with two episodes of projectile vomiting. There was no history of altered sensorium, any visual complaint or seizures. There was no history of trauma, recent vaccination. On examination, the child was fully conscious with stable vitals. On neurological examination, higher functions were normal. On cranial nerve examination, there was involvement of the left sided optic nerve, with only light perception being present. The direct pupillary reflex was absent while the consensual reflex was present (Marcus Gunn pupil). There was generalized hypotonia, power was grade 2 in both lower limbs, right upper limb and grade 3 in left upper limb. Deep tendon reflexes were not elicitable in the upper limbs while they were brisk in the lower limbs along with ankle clonus. Plantar reflex was extensor bilaterally. Abdominal reflex was absent. There was complete sensory loss below the umbilicus, with a band of hyperaesthesia at this level. Fundus examination showed hyperemic disc on the right side while there was optic atrophy on the left side. Terminal neck rigidity was present . Other systems were normal. A clinical impression of acute transverse myelitis, Devic's disease or multiple sclerosis was kept. Laboratory investigations revealed a normal hemogram and erythrocyte sedimentation rate (ESR). Chest X-ray was normal. Cerebrospinal fluid (CSF) examination showed 225 cells/cumm ( lymphocytes 85%, neutrophils 15%), proteins 200 mg%, sugar 43 mg% with simultaneous blood sugar of 128 mg% and no microorganisms on gram stain. MRI brain and spine (Figure 1) showed asymmetric T2/ Flair hyperintensities in bilateral periventricular white matter, centrum semiovale, deep white matter ( right more than left) with diffuse expansion from cervico-medullary junction to C7, 8.5 cm long with signal alteration in cervical cord with syrinx formation in dorsal part of the cervical cord. Expanded cord showed signal alteration in both central and peripheral parts. A central linear hyperintensity was also seen along cervical as well as thoracic cord upto D4 level . Antinuclear antibody was negative while NMO (anti-aquaporin 4) antibody was positive . The child was started on intravenous methylprednisolone, (30mg/kg/day for 5 days) followed by oral prednisolone. The child gradually started improving, her muscle tone improved and power increased to 3/5 on the right side and 4/5 on the left side by day 40. Her urinary incontinence improved gradually and she regained full bladder control by day 60 of illness. On follow-up, the child had one episode of generalized seizure, for which she was put on levetiracitam. After 6 months, there was blindness of the left eye, however there was no motor deficit. Bladder control was normal but there was high residual urine on ultrasonography, leading to recurrent urinary tract infection.
Figure 1a: MRI Coronal section of cervical spine showing diffuse expansion from cervico-medullar junction to C7 with signal alteration in cervical cord with syrinx formation. 1b: MRI axial section of brain showing T-2 FLAIR diffuse and confluent hyperintensities in periventricular region and in deep white matter (right > left).
| | | | Discussion | | The age of onset of neuromyelitis optica ranges from childhood to late adulthood. (4) Multiple sclerosis (MS) and neuromyelitis optica are rare in children, especially in India. (5) NMO-IgG seropositivity in India has been found to be low. (6) The Mayo Clinic revised criteria for diagnosis of neuromyelitis optica in 2006 requires two absolute plus at least two of three supportive criteria. (7) Absolute criteria include optic neuritis and acute myelitis. Supportive criteria include 1) Brain MRI not meeting criteria for MS at disease onset, 2) Spinal cord MRI with contiguous T2-weighted signal abnormality extending over three or more vertebral segments, indicating a relatively large lesion in the spinal cord, 3) NMO-IgG seropositivity. Our patient fulfilled all the criteria but she had brain MRI changes similar to MS. At presentation, she had already developed bilateral optic nerve involvement with visual loss on the left side. Because of pediatric NMO's frequent association with a preceding infection, monophasic course and generally good outcome, some authors consider pediatric NMO to be a variant of acute disseminated encephalomyelitis (ADEM ). (8) Our patient also had a viral prodrome one week prior. The response to treatment in our patient was slow and 6 months after the attack, she had blindness of the left eye and residual bladder involvement, which is unusual in pediatric NMO. Early and accurate diagnosis of NMO and differentiation from MS is important because NMO has a poorer prognosis than MS and generally accepted treatment approaches differ. (6) In a recent case series, CSF cells more than 50 cells/mm3, predominantly polymorphonuclear cells were suggestive of NMO. (4) Neuroimaging has an important role in diagnosis. (3,9) Brain MRI findings are generally either negative or nonspecific in NMO but our patient had periventricular brain lesions, as has been described in multiple sclerosis in bilateral hemispheres. In MS, white matter lesions are typically located juxtacortically and periventricular or callosal. Spinal cord MRI is the most discriminative imaging test for NMO, having T1 hypointense lesions located in the central part of the spinal cord, extending over 3 or more vertebral segments while MS plaques do not extend over more than two segments, are T1 isointense and located posterolaterally in the spinal cord. (9) Our patient also had involvement of more than three contiguous areas along with involvement of both central and peripheral parts. Multiple sclerosis like lesions on brain MRI have been documented in childhood NMO in different case series, but during the later stage of the illness. (10) Our patient had similar lesions at presentation. | | | | Conclusion | | Symptomatic brain lesions, similar to those seen in multiple sclerosis can occur in childhood NMO even at presentation. Optic nerve involvement can go unnoticed in children and the patient can have seizures during the disease course. Unusual manifestations of NMO can be present in children and a sharp watch should be kept for the same. | | | | Contributor Statement | | NS, RSJ, NV managed the case. NS did the literature search. NS, PG prepared the draft of the manuscript. All authors edited and checked the final manuscript. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Mandler RN, Davis LE, Jeffery DR, Kornfeld M. Devic's neuromyelitis optica: a clinicopathological study of 8 patients. Ann Neurol 1993; 34:162–8. [CrossRef]
- Wingerchuck DM, Hongcamp WF, O'Brien PC, Weinshenker BG. The clinical course of neuromyelitis optica. Neurology. 1999; 53:1107-14. [CrossRef]
- Vande Vyver V, Lemmerling M, De Potter R , Verstraete K. Devic's neuromyelitis optica: clinical and imaging findings. JBR–BTR 2007; 90: 284-7.
- Cree B A.C, Goodin DS, Hauser SL. Neuromyelitis Optica. Semin Neurol 2002; 22:105-22. [CrossRef]
- Madan S, Aneja S, Tripathi RP, Batra A, Seth A, Taluja V. Acute disseminated encephalomyelitis: A case series. Indian Pediatr 2005;42:367–71. [PubMed]
- Lekha P. Neuromyelitis optica antibody (NMO-IgG) status in Indian patients with multiple sclerosis and allied demyelinating disorders. Neurology Asia. 2008;13:175-8.
- Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG. Revised diagnostic criteria for neuromyelitis optica. Neurology. 2006; 66:1485–9. [CrossRef]
- Jeffery AR, Buncic JR. Pediatric Devic's neuromyelitis optica. J Pediatr Ophthalmol Strabismus 1996;33:223-9. [PubMed]
- Filippi M, Rocca MA, Moiola L. MRI and magnetization transfer imaging changes in the brain and cervical cord of patients with Devic's neuromyelitis optica. Neurology 1999; 53:1705-10. [CrossRef]
- Cheng C, Jiang Y, Chen X, Dia Y, Kang Z, Lu Z et al. Clinical, radiographic and immunomodulating Changes in Neuromyelitis Optica with extensive brain lesions. BMC Neurol 2013;13:72. [CrossRef] [PMC free article]
DOI: https://doi.org/10.7199/ped.oncall.2015.10
|
| Cite this article as: | | Sharma N, Gautam P, Jaswal R, Kumar N. Wider Spectrum of Neuromyelitis Optica. Pediatr Oncall J. 2015;12: 16-18. doi: 10.7199/ped.oncall.2015.10 |
|