The target joint in Severe Hemophilia A
Govind Kendre, Sunil Hilalpure, Shruti Mantri, Suraj Goyanka, Leo Prince, Murlidharan C
Department of Clinical Hematology, Seth G.S. Medical College, KEM Hospital, Mumbai, India
Address for Correspondence: Dr. Govind Kendre, Department of Clinical Haematology, Seth G.S. Medical College, KEM Hospital, Parel, Mumbai, India.
Email: govindken143@gmail.com
Keywords: Hemophilia A, factor VIII, prophylaxis, target joint
Clinical Problem :
A 12-years-old-boy presented with recurrent left knee joint swelling for 6 months. He was diagnosed to have severe hemophilia A at the age of one year when he was evaluated for multiple ecchymoses over the body. Since then, he has been receiving Factor VIII concentrates on an episodic basis for various problems such as hemarthrosis, muscle hematoma, and gastrointestinal bleeding. He developed a left knee joint swelling 6-months ago following a trivial trauma which was associated with pain and restriction of joint movement. He developed four more spontaneous episodes of left knee joint swelling over a 6-month period. Each time his symptoms resolved with a single dose of Factor VIII concentrate (20 U/Kg). The current episode was treated with RICE (rest, ice, compression, and elevation) measures and a single dose of Factor VIII concentrate (20 U/Kg). His pain resolved and joint movement improved but there was residual joint swelling. On further evaluation, ultrasonography showed synovitis of the left knee joint. After this episode, he was put on secondary prophylaxis with FVIII concentrate (20 U/Kg twice a week). He is on prophylaxis for 6 months and has not developed any further episodes of joint bleed.
Figure 1: The Target Joint
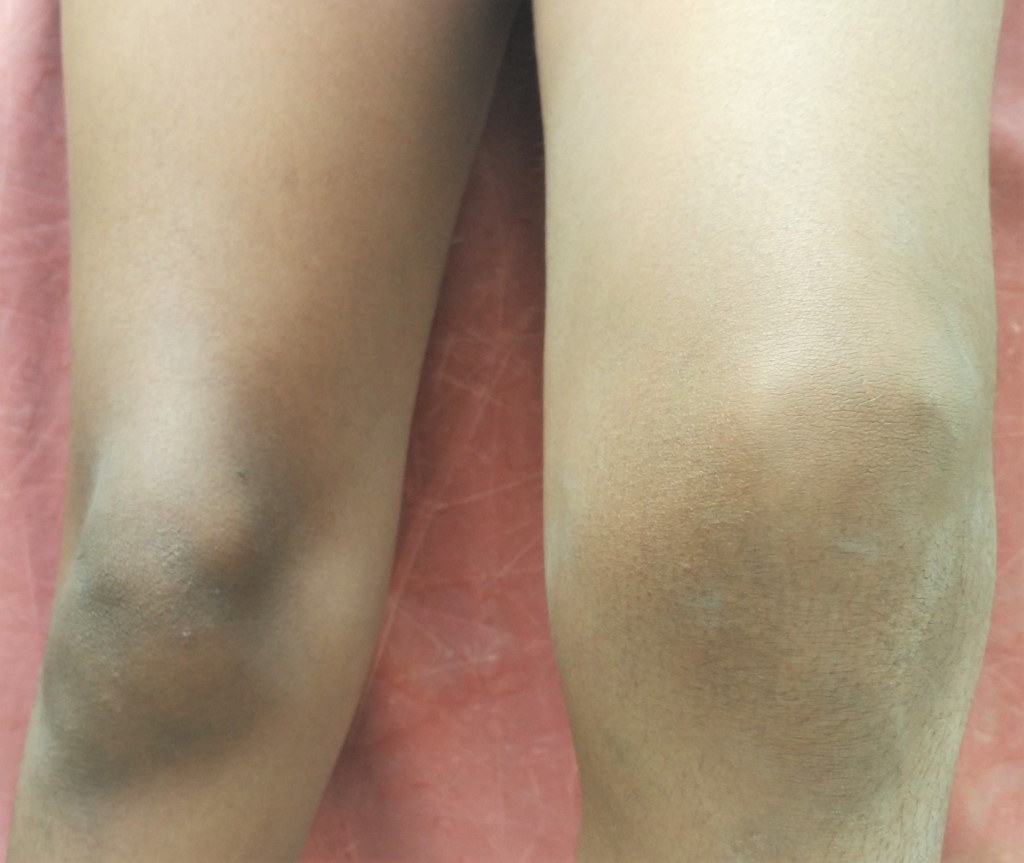
|
What is the target joint and how it should be managed?
Discussion :
Hemophilia is an inherited X-linked recessive disorder caused by a deficiency of coagulation Factor VIII (hemophilia A) or Factor IX (hemophilia B). It is subclassified based on the factor activity as follows: severe [<1 IU/dL (<0.01 IU/mL) or <1% of normal], moderate [1–5 IU/dL (0.01–0.05 IU/mL) or 1%–5% of normal], and mild [5–40 IU/dL (0.05–0.40 IU/mL) or 5% to <40% of normal]. People with severe hemophilia usually bleed frequently into their muscles or joints either spontaneously or related to trauma. A person with moderate hemophilia will rarely experience spontaneous bleeding. A major complication in hemophilia is hemophilic arthropathy caused by repeated intra-articular bleeding, which accounts for 65%–80% of all bleeding episodes. 1 The definition of target joint varies, but commonly a target joint is a joint in which 3 or more spontaneous bleeds have occurred within a consecutive 6-month period. The most commonly affected joints are the knees, ankles, and elbows, although other joints including the hip, wrist, and shoulder are also involved. The process of chronic hemophilic arthropathy is set in motion by the immediate effects of blood on the articular cartilage during hemarthrosis 2,3 and reinforced by persistent chronic synovitis and recurrent hemarthroses, resulting in irreversible damage. With advancing cartilage loss, a progressive arthritic condition develops that includes secondary soft tissue contractures, muscle atrophy, and angular deformities. Failure to manage acute synovitis can result in repeated hemarthroses. 2,3 The radiographic features of chronic haemophilic arthropathy depend on the stage of involvement. Radiographs will only show late osteochondral changes. Ultrasound or MRI examination will show early soft tissue and osteochondral changes. The goal of treatment is to deactivate the synovium as quickly as possible and preserve joint function. Prophylaxis is the treatment by intravenous injection of a factor concentrate a few times per week to prevent anticipated bleeding. Prophylaxis prevents bleeding and joint destruction and should be the goal of therapy to preserve normal musculoskeletal function. The effects have been reported particularly in patients with severe and moderate hemophilia. Doses for prophylactic replacement of factor concentrates vary among centers. Commonly-used dosage for prophylactic factor replacement is 25-40 IU/kg 2-3 times weekly in countries with fewer resource constraints. 4,5 In situations where there are greater constraints on the supply of factor concentrates, prophylaxis may be initiated with lower doses of 10-20 IU/kg 2-3 times per week. 6 Prophylaxis is advisable prior to engaging in activities with a higher risk of injury. Adequate levels of clotting factors are required; a level of >5% is standard for a normal level of daily activity, whereas a factor level of >15% is recommended prior to sports activities. 7
Our case highlights the importance of factor concentrate prophylaxis for the prevention and treatment of the target joint.
Acknowledgment
We thank Dr. Chandrakala S (HOD clinical hematology) and Dr. Farah Jijina for their valuable inputs. | References : | - Gomis M, Querol F, Gallach JE, González LM, Aznar JA. Exercise and sport in the treatment of haemophilic patients: a systematic review. Haemophilia. 2009;15:43–54.
- Llinás A. Haemophilic arthropathy. Haemophilia 2010;16(Suppl 5):121
- Rodriguez-Merchan EC. Musculoskeletal complications of hemophilia. HSSJ. 2010; 6: 37-42.
- Astermark J, Petrini P, Tengborn L, Schulman S, Ljung R, Berntorp E. Primary prophylaxis in severe haemophilia should be started at an early age but can be individualized. Br J Haematol. 1999;105(4):1109-13.
- Blanchette VS. Prophylaxis in the haemophilia population. Haemophilia. 2010;16(Suppl 5):181-8.
- Wu R, Luke KH, Poon MC, Wu X, Zhang N, Zhao L, Su Y, Zhang J. Low dose secondary prophylaxis reduces joint bleeding in severe and moderate haemophilic children: a pilot study in China. Haemophilia. 2011;17:70-4.
- Rodriguez-Merchan EC. Aspects of current management: orthopaedic surgery in haemophilia. Haemophilia 2012;18:8-16.
|
| Correct Answers : |  100% 100% |
|
|
|
|
|
|