Dr. Harish G V, Dr. Sayyed John Pasha.
Department of Pediatrics, Prathima Institute of Medical Sciences, Karimnagar, Telangana, India.
ADDRESS FOR CORRESPONDENCE
Dr. Harish GV, Associate Professor, Department of Pediatrics, Prathima Institute of Medical Sciences, Karimnagar, Telangana, 505417, India.
Email: drharish82@gmail.com | | Abstract | Aim: To determine liver dysfunction and growth in transfusion-dependent beta-thalassemia children of age 2-16 years with iron overload state.
Methods: Forty-one children aged 2-16 years with transfusion-dependent beta-thalassemia who were on chelation therapy with oral iron chelator deferasirox for at least 1 year were included in the study. Serum ferritin, growth assessment and liver function tests (LFT) were measured and correlated. Stunting was defined as low height for age (HFA). According to the World Health Organization (WHO), HFA between -2SD to -3SD was defined as moderate stunting and HFA less than -3SD as severe stunting. Spearmen correlation coefficient was used for finding out a correlation among different variables. Multiple linear regression was carried out to estimate the relationship between serum ferritin level, transfusion rate per year, age, sex and HFA status of thalassemia children.
Results: Out of 41 children, 25 (60%) patients were in the age group of 5-10 years and the overall median age of the patients was 8 years. The overall median rate of blood transfusion was 12.3 per year and median ferritin levels were 3863 ng/ml (95% CI 2781-4860). There was a significant relationship between ferritin level and transfusion rate per year (p=0.048). The median aspartate aminotransferase (AST) was 51 IU/L (95% CI 43-66), alanine aminotransferase (ALT) 45 IU/L (95% CI 31-58). There was a significant relationship between ferritin level and AST, ALT levels (p<0.005) with correlation coefficient 0.31 and 0.55 respectively. AST and ALT levels were rising as the serum ferritin was increasing and after the level of 3000 ng/ml, there was a steep rise. Moderate stunting was seen in 9 (23%) patients with median ferritin level of 3452 ng/ml (95% CI 370–4992 ), severe stunting was present in 14 (34%) patients with median ferritin level of 4432 ng/ml (95% CI 2781 -4860 ) and normal height was seen in 18 (44%) cases with median ferritin level of 3992 ng/ml (95% CI 1904–4862) (p=0.4).
Conclusion: High serum ferritin levels in beta-thalassemia are associated with hepatic dysfunction. Hence liver functions should be monitored frequently in patients with beta-thalassemia major. In our study, we found many children with stunting, but stunting is not affected by iron overload states. | | | | Keywords | | thalassemia major, serum ferritin, liver enzymes, anthropometry. | | | | Introduction | Thalassemia major is the most common genetically transmitted disease in the world. Two related features contribute to the sequelae of thalassemia major; inadequate beta-globin gene production leading to a decreased level of normal hemoglobin (HbA) and unbalanced alpha and beta-globin chain production.1 Depending on mutation and degree of fetal hemoglobin production, transfusions in beta-thalassemia major are necessary beginning in the second month to the second year of life, but rarely later.1 Chronic transfusion therapy improves the quality of life and reduces complications.1 Transfusion induced hemosiderosis becomes the major complication and hemosiderosis induced morbidity can be prevented by adequate iron chelation therapy.1 Frequent blood transfusion causes progressive iron overload which is a major complication of treatment.2 Iron toxicity is one of the major complications with consequential end-organ damage and stunted growth. Iron is initially deposited in the liver and then in the endocrine system and then in heart.1 This leads to iron-induced liver injury, growth hormone deficiency, hypothyroidism, hypoparathyroidism, diabetes mellitus and cardiac dysfunction.1 The liver is the earliest site of iron deposition in transfusion-dependent thalassemia children and iron-induced liver injury is the commonest cause of morbidity. Iron overload occurs both in hepatocytes and reticuloendothelial cells.2 Growth retardation in thalassemia is because of chronic tissue hypoxia, iron toxicity, nutritional deficiency (zinc, folate) and growth hormone failure.3 With introduction of iron chelators, survival rates have improved4 but endocrine complications have become more frequent and significantly affect the quality of life.5 We did this study to correlate liver dysfunction and growth in beta-thalassemia major children aged between 2 to 16 years with iron overload states.
| | | | Methods & Materials | Our institution has a center of excellence for sickle cell anemia and thalassemia where blood transfusions and iron chelation therapy are provided. We enrolled the children diagnosed with thalassemia major from January 2019 to March 2019. A total of 45 children presented to the center, but 4 of them did not give consent to participate in the study. Thus, 41 children aged 2-16 years with transfusion-dependent beta-thalassemia who were registered in our center and who were on chelation therapy with oral iron chelator deferasirox for at least 1 year were enrolled in the study. In these children, serum ferritin levels, liver function tests (LFT) and anthropometric measurements (height, weight, body mass index (BMI), height for age, weight for age) were measured. Body mass index (BMI) was calculated using the formula of weight in kg/height in sqm. Growth parameters were calculated based on the World Health Organization (WHO) growth charts. Stunting was defined as low height for age (HFA). According to WHO, HFA between -2SD TO -3SD was moderate stunting and HFA less than -3SD as severe stunting.6 According to Indian Academy of Pediatrics (IAP) classification, weight for age (WFA) >80% was considered as normal, between as 71-80% as grade 1 protein-energy malnutrition (PEM), between 61-70% as grade 2 PEM, between 51-50% as grade 3 PEM and <50% as grade 4 PEM.7
Demographic details were recorded with semi-structured questionnaires. Serum ferritin levels were measured by enzyme-linked fluorescent assay method. Serum ferritin levels >250 ng/ml, aspartate aminotransferase (AST) >38 IU/L, alanine aminotransferase (ALT) >40 IU/L and alkaline phosphatase (ALP) >96 IU/L were considered abnormal. Normal range of total protein were considered at 6.7 to 8.6 gm/dl, albumin at 4-5 gm/dl, total bilirubin at 0.3-1.3 mg/dl, direct bilirubin at 0.1-0.4 mg/dl.8 Serum ferritin levels were correlated with LFT and anthropometric measurements. The transfusion requirement per year was calculated.
Statistical Analysis: Data were entered in MS Excel 2007. Median with 95% Confidence Interval (CI) was calculated for quantitative data such as age, serum ferritin, and LFT. Few patients had extremely high or low values for serum ferritin and LFT, so mean and its standard deviation (SD) would be highly skewed. Hence, the median with 95% CI were analyzed in the study. Qualitative data such as gender, nutritional status was presented as a percentage with its 95% CI. Spearmen correlation coefficient was used for finding out a correlation among different variables. Kruskal Wallis test was used to see the difference in median serum ferritin levels among different grades of growth parameters (HFA and WFA). Multiple linear regression was carried out to estimate the relationship between serum ferritin level, transfusion rate per year, age, sex and HFA status of thalassemia children. All statistical analysis was done using IBM SPSS version 20. | | | | Results | Male: female ratio was 16: 25. Twenty-five (60.9%) patients were in the age group of 5-10 years, 5 (12%) were <5 years of age and 11 (27%) were in the age group of 11 to 16 years. The overall median age was 8 years. The median rate of blood transfusion was 12.3 per year (in males 12.1 per year, in females 12.9 per year). The median serum ferritin level was 3863 mg/dl (95% CI 2781-4860 ng/ml). Median serum ferritin levels in males were 4122 mg/dl and in females 3475 mg/dl. The median duration of the disease was 87 months (89 months in males, 85 months in females). Multiple linear regression the relationship between serum ferritin level, transfusion rate per year, age, sex and HFA status showed R2 value of 25.4% (p=0.028) indicating 25.4% of the variation in serum ferritin level can be explained by the model containing age, sex, HFA status and transfusion rate per year. There was a statistically significant relationship between serum ferritin level and transfusion rate per year (p=0.048) keeping all other factors constant in this model. With one transfusion per year, the mean increase in serum ferritin level was by 149.1 (95% CI 1.3-297.6).
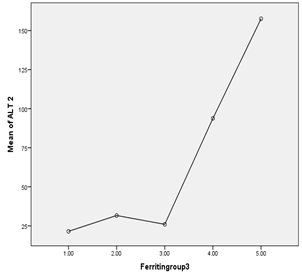
The median total protein was 7.2 gm/dl (95% CI 7-7.5), albumin 4.2 gm/dl (95% CI 4.2-4.3), globulin 2.9 gm/dl (95% CI 2.8-3.3), A/G ratio 1.4 (95% CI 1.4-1.5), total serum bilirubin 1.4 mg/dl (95% CI 1.1-1.7), direct bilirubin 0.6 mg/dl (95% CI 0.5-0.7), AST 51U/L (95% CI 43-66), ALT 45 U/L (95% CI 31-58) and alkaline phosphate 153 U/L (95% CI 133-190). There was a statistically significant relationship between serum ferritin levels and serum AST and ALT levels (p<0.05) with correlation coefficient 0.31 and 0.55 respectively. AST and ALT levels were rising as the serum ferritin was increasing and after the level of 3000 ng/ml, there was a steep rise in AST and ALT levels (Figures 1 and 2).
Figure 1. Association of serum ferritin and AST
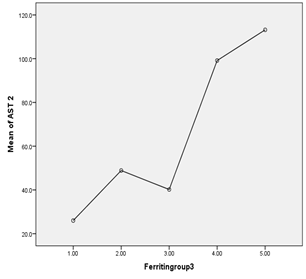
Figure 2. Association of serum ferritin and ALT
Moderate stunting was seen in 9 patients (males 6, females 3) with median serum ferritin level of 3452 ng/ml, severe stunting was seen in 14 patients (males 8, females 6) with median serum ferritin level of 4432 and normal height was seen in 18 patients (males 11, females 7) with median serum ferritin level of 3992 ng/ml (p=0.49). As per IAP classification, WFA was normal in 15 patients (males 12, females 3) with median serum ferritin level of 4122 ng/ml (95% CI 1838-6224), grade1 PEM was seen in 11 patients (males 5, females 6) with median serum ferritin level of 3319 ng/ml (95% CI 1996-4860), grade 2PEM was seen in 6 patients (males 3, females 3) with median serum ferritin level of 3731 ng/ml (95% CI 1095-7127) , grade 3 PEM was seen in 5 patients (males 2, females 3) with median serum ferritin level of 3088 ng/ml (95% CI 1256-8739), grade 4 PEM was seen in 4 patients (males 3, females 1) with median serum ferritin level of 4753 ng/ml (95% CI 370-7568) (p=0.861).
| | | | Discussion | The liver is the earliest organ affected by iron overload in thalassemia children and serum AST and ALT are raised due to peroxidase injury and direct toxic effect of iron on liver cells.2 As iron deposits in the liver, its functions are affected which are predicted by raised AST and ALT. In our study, AST and ALT levels were raised significantly especially in patients where serum ferritin was >3000 ng/ml and there was a positive correlation between serum ferritin and AST and ALT. A similar study was done by Suman et al where serum ferritin levels were correlated with liver enzymes and found a significant correlation between serum ferritin levels and liver enzymes.2 Asif et al did a study on serum ferritin level and LFT and found weak insignificant positive correlations between serum ALT (Pearson correlation 0.097; p=0.181), serum AST (Pearson correlation 0.045; p=0.335) and serum ALP (Pearson correlation 0.036; p=0.364) with serum ferritin levels.10
Despite major therapeutic progress, growth failure still is a significant challenge in beta-thalassemia major children. We found moderate stunting in 21%, severe stunting in 34% but stunting is not affected by iron overload states. Similarly, PEM was seen in 26 patients in our study but did not correlate with the iron-overload states. Pemde et al did a study on physical growth in children with transfusion-dependent thalassemia and found 33% of the patients had short stature which they found associated with increased serum ferritin which we did not find in our study.11 Moiz et al did a study on anthropometric measurements in children having transfusion-dependent beta-thalassemia and found that 65.4% children had stunted growth with a significant negative correlation between height for age z-score and serum ferritin levels.3 Elizabeth et al did a study on the correlation between serum ferritin level and growth disorders in children with thalassemia and found that the risk factors of short stature were low Insulin-like growth factor (IGF-1) level and low family income.12 Similarly Saxena et al did a study on growth retardation in thalassemia major patients and found that thalassemic patients are short, have low rate of growth and BMI and have either delayed or absent pubertal spurt, which is related to low hemoglobin and high ferritin level and sub-optimal iron chelation therapy.13 In our study, ferritin levels did not statistically correlate with growth retardation. This may be due to a small sample size and extreme serum ferritin levels. Also, growth retardation is multifactorial (poor socio-economic background, low hemoglobin levels, nutrition). | | | | Conclusion | | High serum ferritin levels in beta-thalassemia are associated with hepatic dysfunction. Hence liver functions should be monitored frequently in patients with beta-thalassemia major. In our study, we found many children with stunting, but stunting is not affected by iron overload states. | | | | Acknowledgement | | Dr. Momin Kashif, Assistant Professor, Department of Community Medicine, Prathima Institute of Medical sciences, Karimnagar. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- BeBaun MR, Frei-Jones M, Vichinsky E. Thalassemia Syndromes. In: Kleigman RM et al, eds. Nelson Textbook of Pediatrics. 19th ed. Philadelphia, PA: Elsevier Saunders; 2011:1674-1677
- Suman RL, Sanadhya A, Meena P, Goyal S. Correlation of liver enzymes with serum ferritin levels in β-thalassemia major. Int J Res Med Sci 2016; 4:3271-4 [CrossRef]
- Moiz B, Habib A, Sawani S, Raheem A, Hasan B, Gangwani M. Anthropometric measurements in children having transfusion-dependent beta thalassemia. Hematology. 2018;23(4):248-252. [CrossRef] [PubMed]
- Telfer PT, Warburton F, Christou S, Hadjigavriel M, Sitarou M, Kolnagou A, Angastiniotis M. Improved survival in thalassemia major patients on switching from desferrioxamine to combined chelation therapy with desferrioxamine and deferiprone. Haematologica Dec 2009, 94 (12) 1777-1778 [CrossRef] [PubMed] [PMC free article]
- Italian Working Group on Endocrine Complications in Non-endocrine Diseases. Multicentre study on prevalence of endocrine complications in thalassaemia major. Clin Endocrinol (Oxf). 1995 Jun;42(6):581-6. [CrossRef] [PubMed]
- World Health Organization (WHO). The WHO Child Growth Standards. Available at URL: https://www.who.int/childgrowth/standards/en/. Accessed on 2nd Nov 2019
- Nutrition Sub-committee of Indian Academy of Pediatrics. Report. Indian Pediatr 1972; 9: 360.
- Rachmilewitz EA, Giardina PJ. How I treat thalassemia. Blood. 2011; 118: 3479-3488. [CrossRef] [PubMed]
- Rodwell VW, Bender DA, Botham KM, Kennelly PJ, Weil PA. Harpers illustrated biochemistry. New York: McGraw-Hill Education; 2018.
- Asif M, Manzoor Z, Farooq MS, Kanwal A, Shaheen U, Munawar SH, et al. Correlation between serum ferritin level and liver function tests in thalassemic patients receiving multiple blood transfusions. International Journal of Research in Medical Sciences. 2017;2: 988-994. [CrossRef]
- Pemde H, Jagdish Chandra J, Gupta D, Singh V, Sharma R, Dutta AK. Physical Growth in Children with Transfusion-Dependent Thalassemia. Pediatric Health, Medicine and Therapeutics 2011:2 13-19 [CrossRef]
- Elizabeth M, Fadlyana E, Reniarti L, Faisal F, Sukandar H, Rusmil K. Serum IGF-1 and short stature in adolescents with beta-thalassemia major. PI [Internet]. 16Jul.2018 [cited 5Nov.2019];58(4):151-. Available from: https://paediatricaindonesiana.org/index.php/paediatrica-indonesiana/article/view/1761. [CrossRef]
- Saxena A. Growth Retardation in Thalassemia Major Patients. Int J Hum Genet. 2003:3: 237-246 [CrossRef]
DOI: https://doi.org/10.7199/ped.oncall.2019.33
|
| Cite this article as: | | G V H, Pasha S J. Correlation of Serum Ferritin Levels with Liver Function Tests and Anthropometric Measurements in Transfusion Dependent Beta-Thalassemia Major Children: A Cross Sectional Study. Pediatr Oncall J. 2019;16: 101-104. doi: 10.7199/ped.oncall.2019.33 |
|