Joana Pinho1, Beatriz Vieira2, Vilma Lopes1, Conceição Costa3.
1Paediatrics Department, Centro Hospitalar Vila Nova de Gaia/ Espinho, Portugal,
2Paediatrics Department, Centro Hospitalar da Póvoa de Varzim/Vila do Conde, Póvoa de Varzim, Portugal,
3Neonatal Intensive Care Unit, Paediatrics Department, Centro Hospitalar Vila Nova de Gaia/Espinho, Portugal.
ADDRESS FOR CORRESPONDENCE
Joana Pinho , Centro Hospitalar Vila Nova de Gaia/Espinho R. Conceição Fernandes S/N, 4434-502 Vila Nova de Gaia.
Email: joana.pinho.92@gmail.com | | Abstract | Macroglossia is a condition characterized by an abnormally enlarged tongue, which may cause several complications. Beckwith-Wiedemann syndrome (BWS) is one of the causes of macroglossia and may also be associated with macrosomia and omphalocele. This syndrome may also be associated with congenital malformations and embryonal tumours.
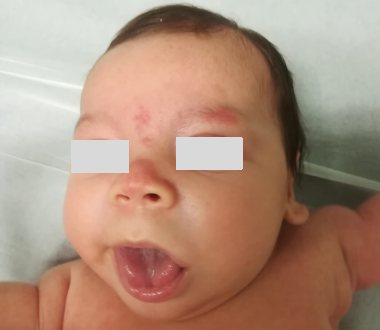
We present a clinical case of a male infant with macroglossia since birth. Genetic testing of the 11p15.5 region confirmed the diagnosis of BWS with loss of methylation in the region corresponding to domain 2 (DMR2. The methylation pattern change observed in the DMR2 region, as seen in our patient, is less common than changes in the DMR1 region and is associated with a lower risk of tumour development. Tumour screening protocols are modified according to molecular test results and geographical location. This personalised approach reduces unnecessary invasive procedures and associated anxiety and allows for more effective and efficient surveillance. | | | | Keywords | | macroglossia, Beckwith-Wiedemann Syndrome, genetic testing. | | | | Case Report | We report the case of a male infant with macroglossia and persistent tongue protrusion since birth. (Figure 1) The pregnancy resulted from intracytoplasmic sperm injection and developed with appropriate prenatal care and without complications (serology and ultrasound without changes). He was born by uncomplicated caesarean section with a birth weight of 3965 g (63rd percentile), length of 49.5 cm (14th percentile) and head circumference of 36 cm (64th percentile) according to the World Health Organization growth charts. There was no evidence of hypoglycaemia or other postnatal complications. At one month of age, the macroglossia persists without any other objective changes. (Figure 2) There is no evidence of respiratory distress or obstruction. Family history is negative for genetic disorders or hereditary metabolic disorders. Urine and blood tests excluded some pathologies such as hypothyroidism, mucopolysaccharidosis and Pompe disease. Genetic testing of the 11p15.5 region confirmed the diagnosis of SBW with loss of methylation in the region corresponding to domain 2 (DMR2), more precisely in KCNQ1 overlapping transcript 1 (KCNQ1OT1). No alterations were found in the domain 1 (DMR1) region.
Figure 1. Male infant with macroglossia at birth.
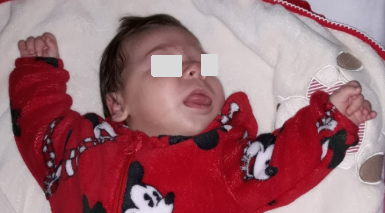
Figure 2. One-month-old infant with persistent macroglossia.
| | | | Discussion | Macroglossia is a rare anatomical condition often associated with abnormal growth of the tongue, sometimes with protrusion.1 It can cause airway obstruction, speech changes, feeding difficulties and deformities of the oral cavity. It rarely occurs alone and is often associated with other conditions. When present from birth, it is classified as congenital and occurs in conditions such as Beckwith-Wiedemann syndrome (BWS), congenital hypothyroidism, mucopolysaccharidosis, Pompe disease, haemangiomas, lymphangiomas and others. It may also occur in association with infectious/inflammatory diseases (allergic or radiation reactions, trauma, syphilis, tuberculosis, glossitis, etc.), tumours (haemangioma, lipoma, etc.), endocrine diseases (hypothyroidism, acromegaly, diabetes mellitus, etc.) or metabolic diseases.1
BWS is a rare genetic disorder with an estimated prevalence of 1 in 10300-13700 births, with no sex difference.2,3 Most cases occur spontaneously, but hereditary transmission is present in 15% of cases. Reproductive techniques are a risk factor as they are associated with an increase in imprinting disorders.4
Although the phenotype is very variable due to the presence of macroglossia, macrosomia and omphalocele, these are the common clinical manifestations of this syndrome.3,5 Of these three features, only macroglossia was present in our patient.
SBW may also be associated with the development of congenital malformations and the development of embryonal tumours such as Wilms' tumour, hepatoblastoma, neuroblastoma and rhabdomyosarcoma.6 The risk of neoplasia is approximately 7.5%, with a higher incidence in the first 8 years of life.
The majority of BWS cases are mainly due to genetic or epigenetic defects within the 11p15.5 region (~80%).3 This gene has two functionally independent domains: Imprinting centre 1 (IC1) regulates the expression of insulin-like growth factor 2 (IGF2) and H19 (non-translated RNA that may function as a tumour suppressor) in domain 1; and imprinting centre 2 (IC2) regulates the expression of cyclin-dependent kinase inhibitor 1C (CDKN1C), potassium channel voltage-gated KQT-like subfamily member (KCNQ1) and KCNQ1 overlapping transcript 1 (KCNQ1OT1) in domains 2.5 IC1 and IC2 are sometimes referred to as differentially methylated regions DMR1 and DMR2, respectively and in this clinical case a change in the methylation pattern of DMR2, more specifically in KCNQ1OT1, was identified.5
Alterations in the methylation pattern of the DMR2 region (as seen in our patient) are rarer than alterations in the DMR1 region and are associated with a lower risk of developing tumours such as Wilms' tumour and hepatoblastoma (2.6% vs. 16-30%).5
Tumour screening is modified according to molecular test results and geographical location. The tumour screening guidelines developed by the American Association for Cancer Research Childhood Cancer Predisposition Workshop include abdominal ultrasound and serum alpha-fetoprotein at diagnosis for all patients, then every 3 months until 4 years of age. Thereafter, ultrasound screening alone should continue every 3 to 4 months until the age of 7 years.3,7 According to protocols developed for European experts, abdominal ultrasound is not recommended for surveillance in patients with genetic alterations associated with a lower risk of tumour development, such as loss of methylation in IC2.3,5,7,8 Given the fact that our patient has this genetic alteration, there is no formal indication to carry out additional screening tests. If genetic alterations are associated with an increased risk of abdominal tumours or if the genetic test doesn't show any alteration, surveillance by abdominal ultrasound should be performed at regular intervals from the age of 3 to 7 years.5
With this clinical description, we want to highlight the importance of aetiological investigation of examination findings such as macroglossia and their respective orientation. The result of the genetic test allowed the identification of the affected region and this information is of fundamental importance for the prognosis and follow-up of the patient. In this case, once the region has been identified as having a low risk of tumour development, the patient should continue to be followed, but it's not necessary to have regular abdominal ultrasound scans.
Learning points:
● Beckwith-Wiedemann syndrome is a rare genetic disorder associated with macroglossia, macrosomia and involving a predisposition to tumour development.
● Alterations in the methylation pattern of the DMR2 region are associated with a lower risk of developing tumours such as Wilms' tumour and hepatoblastoma.
● Tumour screening is modified according to molecular test results and geographical location.
● In Europe, genetic alterations that are associated with a lower risk of tumour development, such as loss of methylation in IC2, don't have an indication for a subsequent screening test such as abdominal ultrasound.
● Genetic testing is very important for predicting prognosis and monitoring patients.
| | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Núñez-Martínez PM, García-Delgado C, Morán-Barroso VF, Jasso-Gutiérrez L. Macroglosia congénita: características clínicas y estrategias de tratamiento en la edad pediátrica [Congenital macroglossia: clinical features and therapeutic strategies in paediatric patients]. Bol Med Hosp Infant Mex. 2016 May-Jun;73(3):212-216. Spanish. doi: 10.1016/j.bmhimx.2016.03.003. Epub 2016 Jun 4. PMID: 29421209. [CrossRef] [PubMed]
- Mussa A, Russo S, De Crescenzo A, Chiesa N, Molinatto C, Selicorni A,et al. Prevalence of Beckwith-Wiedemann syndrome in North West of Italy. Am J Med Genet A. 2013 Oct;161A(10):2481-6. doi: 10.1002/ajmg.a.36080. Epub 2013 Aug 5. PMID: 23918458. [CrossRef] [PubMed]
- Wang KH, Kupa J, Duffy KA, Kalish JM. Diagnosis and Management of Beckwith-Wiedemann Syndrome. Front Pediatr. 2020 Jan 21;7:562. doi: 10.3389/fped.2019.00562. PMID: 32039119; PMCID: PMC6990127. [CrossRef] [PubMed] [PMC free article]
- Cortessis VK, Azadian M, Buxbaum J, Sanogo F, Song AY, Sriprasert I, et al. Comprehensive meta-analysis reveals association between multiple imprinting disorders and conception by assisted reproductive technology. J Assist Reprod Genet. 2018 Jun;35(6):943-952. doi: 10.1007/s10815-018-1173-x. Epub 2018 Apr 25. PMID: 29696471; PMCID: PMC6030010. [CrossRef] [PubMed] [PMC free article]
- Brioude F, Kalish JM, Mussa A, Foster AC, Bliek J, Ferrero GB, et al. Expert consensus document: Clinical and molecular diagnosis, screening and management of Beckwith-Wiedemann syndrome: an international consensus statement. Nat Rev Endocrinol. 2018 Apr;14(4):229-249. doi: 10.1038/nrendo.2017.166. Epub 2018 Jan 29. PMID: 29377879; PMCID: PMC6022848. [CrossRef] [PubMed] [PMC free article]
- Cohen MM Jr. Beckwith-Wiedemann syndrome: historical, clinicopathological, and etiopathogenetic perspectives. Pediatr Dev Pathol. 2005 May-Jun;8(3):287-304. doi:10.1007/s10024-005-1154-9. Epub 2005 Jul 14. PMID: 16010495. [CrossRef] [PubMed]
- Kalish JM, Deardorff MA. Tumor screening in Beckwith-Wiedemann syndrome-To screen or not to screen? Am J Med Genet A. 2016 Sep;170(9):2261-4. doi: 10.1002/ajmg.a.37881. PMID: 27518916; PMCID: PMC5930355. [CrossRef] [PubMed] [PMC free article]
- Kalish JM, Doros L, Helman LJ, Hennekam RC, Kuiper RP, Maas SM, et al. Surveillance recommendations for children with overgrowth syndromes and predisposition to wilms tumors and hepatoblastoma. Clin Cancer Res. 2017 Jul 1;23(13):e115-e122. doi: 10.1158/1078-0432.CCR-17-0710. PMID: 28674120; PMCID: PMC5538793. [CrossRef] [PubMed] [PMC free article]
DOI: https://doi.org/10.7199/ped.oncall.2024.23
|
| Cite this article as: | | Pinho J, Vieira B, Lopes V, Costa C. Macroglossia: the importance of genetic testing for prognosis. Pediatr Oncall J. 2024;21: 65-67. doi: 10.7199/ped.oncall.2024.23 |
|