Dr Tripti Pensi.
Professor of Pediatrics, Dr.R.M.L Hospital, New Delhi. | The major modes of transmission of HIV include sexual route, transfusion of infected blood and blood products, intravenous drug abuse and mother to child transmission. Another mode of transmission, though largely undocumented, is transmission of infection by needle stick or other sharp injury in a hospital or a health care setting. Although, avoiding occupational blood exposures is the primary means of preventing HIV transmission in health care settings, appropriate post exposure management guidelines form an important element of work place safety. These guidelines were first formulated in 1990 and were last revised by US Public Health Services in 1998. Since then, several new anti-retroviral agents have been approved by FDA, more information is available regarding use and safety of these drugs, possibility of transmission of drug resistant virus and concern has arisen about use of PEP when it is not warranted. Hence, these guidelines have been revised and published by US Public Health Service Department again in June 2001. Post-exposure prophylaxis following exposure to HIV is most commonly used after an occupational exposure but can also be used following non-occupational exposure particularly in cases of sexual assault.
Post exposure prophylaxis following occupational exposure to HIV:
Health Care Personnel (HCP) is normally at a very low risk of acquiring HIV infection during management of an HIV infected patient. However, the absence of a vaccine or effective-curative treatment following an accidental occupational exposure makes the health care personnel apprehensive. So, it is very necessary to have a comprehensive programme in place to deal with any accidental exposure to blood or body fluids of an HIV infected person.
Some of vital issues dealing with transmission of HIV to HCP are:- Who is at risk?
- How much is the risk?
- What factors influence this risk?
- How to reduce the risk?
- What is role of antiretroviral agents in reducing this risk?
- Issues about safety of PEP drugs and their use in pregnancy
- Need to develop a comprehensive programme for PEP implementation
Who is at Risk?
All categories of Health Care Personnel (HCP) are at a potential risk of acquiring HIV infection during care of an HIV infected patient.
Definition of "Health Care Personnel" & Exposure
"Health Care Personnel (HCP)" are defined as "any person (e.g. employees, students, clinicians, public safety workers or volunteers) whose activities involve contact with patients or with blood or other body fluids from patients in a health care or laboratory setting".
An "exposure" that may place an HCP at risk of HIV infection is defined as a percutaneous injury (e.g. needle-stick or cut with a sharp instrument), contact of mucous membrane or non-intact skin (esp. when the exposed skin is chapped, abraded, or afflicted with dermatitis), or contact with intact skin when the duration of contact is prolonged (e.g. several minutes or more) with blood or other potentially infectious body fluids. Body fluids that are potentially infectious include - blood, semen, vaginal secretions, cerebrospinal, synovial, pleural, peritoneal, pericardial and amniotic fluids or other body fluids contaminated with visible blood. Exposure to tears, sweat, urine, faeces, saliva of an HIV infected person is normally not considered as an "exposure" unless these secretions contain visible blood.
Professionals with frequent blood exposures :- Surgeons
- Emergency care providers
- Dentist
- Nurses
- Labour and delivery personnel
- Laboratory technicians
What is the average risk of acquiring HIV infection after an occupational exposure?
The risk is normally very low, but it is not zero. The average risk of acquiring HIV infection after different types of occupational exposure is given in Table.
| Average risk of HIV infection after an Occupational Exposure | - Small amount of blood on intact skin
| No risk | | - Risk with damaged skin / prolonged exposure
| 1 in 1000 | (0.01%) | - Exposure on eyes, nose or mouth
| 1 in 1000 | (0.01%) | - Needle Stick / other sharp injuries
| 1 in 300 | (0.3%) | | (Risk from all "exposures" increases further if exposure involves a large volume of blood or a higher amount of HIV in patient's blood as in patients with acute HIV infection or patient near death.) |
As is evident, the average risk of acquiring HIV infection after a needle stick injury is 0.3%, while the comparable risk for HBV is 9 - 30% and 1 - 10% for HCV.
How many health care personnel have been infected occupationally?
As on June 2000, in USA, there are 56 reported cases of HCP with documented HIV seroconversion associated with an occupational exposure. Another 138 cases are considered possible occupational exposure as these had history of occupational exposure to HIV, no other risk factors for HIV but seroconversion was not documented. However, as per reported data globally there are 98 confirmed and 194 possible cases of HCP getting infected occupationally. In our country there is only one documented case reported to NACO of a HCP acquiring HIV due to occupational exposure.
| Type of Exposure of 55 documented cases | Type of fluid involved in exposure | | Needle stick / cuts/sharp injuries | 48 | Blood | 50 | | Eye, Nose, Mouth, Skin exposure | 5 | Conc.virus in lab. | 3 | | Both injury & mucous membrane exposure | 1 | Visibly bloody fluid | 1 | | Unknown | 1 | Unspecified fluid | 1 |
What factors influences this risk?
Various epidemiological and laboratory studies have shown that the risk of infection varies with type of exposure factors such as- Type of needle (hollow bore Vs. solid)
- Device visibly contaminated with patient's blood
- Depth of injury
- The amount of blood involved in the exposure
- The amount of virus in patient's blood at the time of exposure.
- Whether Post Exposure Prophylaxis (PEP) was taken within the recommended time
How to reduce this risk?
Before we discuss the post exposure prophylaxis, it must be emphasised that prevention remains the mainstay and universal precautions must be followed by all health care workers at all the times. Also all the patients must be seen as potentially infectious because it may not be practical or desirable to test all the patients for HIV. The standard (universal) precautions are well known to the medical personnel but they are not followed in usual practice, may be due to lack of facilities particularly in district hospitals and primary health centre. But sometimes even when the facilities are available, there is a certain degree of complacency among doctors and other health care workers. This complacency has to be removed and concept of universal precautions be put to actual use once again in this era of emerging and re-emerging infections. One must remember, "Prevention is the main stay of strategy to avoid occupational exposure to the HIV".
Protective barriers
Whenever exposure to blood/other potentially infected fluid is anticipated, protective barriers must be used.
- Latex or vinyl gloves must be worn while carrying out any procedure and be decontaminated after each use. Gloves with holes/tears should not be used. Double gloves are not preferred as these are not more protective than single glove & these may be more clumsy also. Heavy-duty rubber gloves should used for cleaning instruments, handling solid linen or spills of blood/body fluids. These can be washed & reused.
- Gloves and aprons protect one from splashes of blood or body fluids e.g. during surgery/delivery. One may wear a waterproof gown or a sterile cloth with a plastic apron underneath. Protective eyewear may be used to prevent transmission by splash of fluids to the mucous membrane.
Safe handling of sharps :- Careful handling of hollow bore needle is very essential as it may lead to deep injuries.
- The needles should never be recapped. In situations where recapping is essential, single hand method should be used.
- Needles should never be bent/broken by hand.
- Needles should not be left on trolleys & beds and must be disposed of immediately.
- Never pass used sharps from one person to another directly.
- Use forceps instead of fingers for guiding sutures.
- The sharps should be disposed off in a puncture resistant container containing bleach.
What should be done on exposure to HIV infected blood / body fluids?
Immediately following an exposure :- Needle stick injuries and cuts should be washed with soap and water
- Pricked finger should not be put into mouth reflexly.
- Splashes to the nose, mouth or skin, should be flushed with plenty of water
- Eyes should be irrigated with clean water; saline, or sterile irrigants
Note: No scientific evidence exists as to the fact that the use of antiseptics for wound care or squeezing the wound will reduce the risk of transmission of HIV. However, use of antiseptics is not contraindicated. The use of a caustic agent such as bleach is not recommended.
One must report an "exposure" immediately to the appropriate authorities and condition must be treated as an emergency. Prompt reporting is essential because in some cases, HIV post exposure prophylaxis (PEP) may be recommended and it should be started as soon as possible, preferably within a few hours. Initiating treatment after 72 hours of exposure is usually not recommended.
Is there some treatment available after an occupational exposure to HIV?
Various animal studies have been done over the years and these have provided encouraging evidence of post exposure chemo prophylactic efficacy. It has also been shown that delaying initiation, shortening the duration or decreasing the anti retroviral dose of PEP, individually or in combination, decreased its prophylactic efficacy
However, little information exists about efficacy of PEP in humans as seroconversion is infrequent following an occupational exposure to HIV infected blood, it would need thousands of HCP to enroll in prospective study to achieve the statistically significant results on efficacy of PEP
However, in a retrospective case control study of HCP, it was demonstrated that use of zidovudine as PEP was associated with a reduction in the risk of HIV infection by approximately 81%.
Also our experience in HIV infected patients has shown that combination of different anti retroviral agent is superior to monotherapy regimen, so a combination of two or three drugs in PEP regimen should be more beneficial than single drug. One needs to consider toxicity of a combination regimens vis-a-vis risk of transmission. NRTI combinations being considered for PEP includes zidovudine (ZDV) & lamivudine (3TC), 3TC & stavudine (d4T), and didanosine (ddI) & d4T. In previous regimens ZDV & 3 TC was considered first regimen but emerging resistance to ZDV & 3TC in certain geographical areas might demand different initial combinations. The addition of a third drug for PEP in high risk exposures is based on their demonstrated effectiveness in reducing viral burden in HIV infected persons. Previously indinavir (IDV) or nelfinavir (NFV) were recommended as first choice agents in "expanded PEP regimens". But now with chances of PI resistance particularly on exposure to patients who are on triple therapy, efavirenz (FFV) and abacavir (ABC) are being considered as alternatives. However, nevirapine (NVP) has not been recommended for use in PEP regimen.
Failure of PEP to prevent HIV infection in HCP has been reported in at least 21 cases (16 on ZDV, two on ZDV and ddI and three on triple drug combination).
How does PEP works?
PEP is recommended because it has been observed that systemic infection does not occur immediately after an exposure and there is a brief window period of opportunity available to contain or eradicate HIV from body before wide spread cellular infection occur. After a needle-stick injury, immune cells (dendritic) in skin present the HIV to T-lymphocytes and natural killer cells. It is seen that in monkey models, Simian immunodeficiency virus (SIV) when injected into vagina, virus is engulfed by immune cells in the vaginal lining. Within 2 days SIV is found in inguinal lymph nodes. Within 5 days SIV infected T-cells and free SIV can be detected in blood stream 9. Migration of SIV after rectal or penile tip exposure follows a similar time frame. PEP can stop viral replication during this early period before it gains a 'foothold' in the body.
Is post-exposure prophylaxis recommended for all types of occupational exposures to HIV?
No, because most occupational exposures do not lead to HIV infection. The chances of possible serious side effects (toxicity) from the drugs used for PEP may be much greater than the chance of HIV infection from such exposures. Both risk of infection and possible side effects of drugs should be carefully considered when deciding whether to give post-exposure prophylaxis or not.
The decision to start PEP is made on the basis of degree of exposure to HIV (Exposure Code) and the HIV status of the source (HIV Status Code) from whom the exposure has occurred. The Exposure Code and Status Code are then correlated to arrive at a decision regarding initiation of PEP.
Determination of the Exposure Code (EC):
Exposure code can be defined as per the flow chart given below. It may be classified into three categories, EC-1, EC-2 and EC-3, depending upon the severity of exposure.
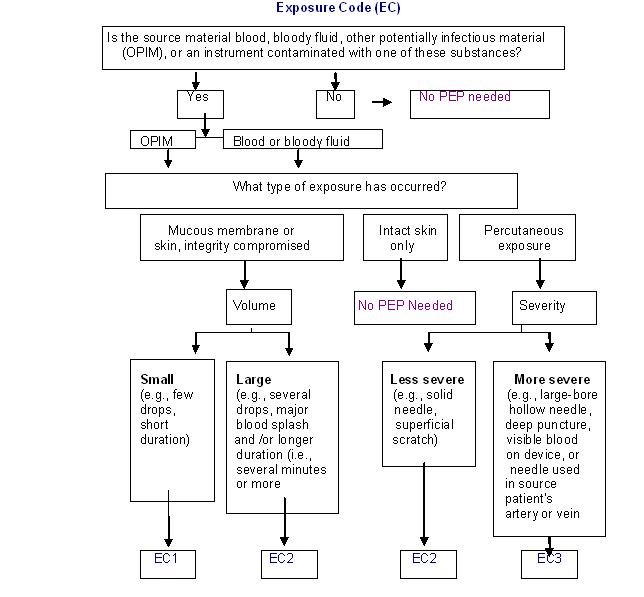
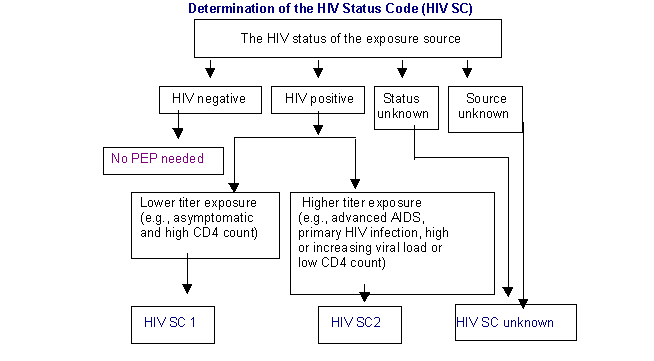
Determine the PEP recommendation
| EC | HIV SC | | PEP recommendation | | 1 | 1 | | PEP may not be warranted | | 1 | 2 | | Considerbasic regimen.
(Exposure type poses a negligible risk for HIV transmission). | | 2 | 1 | | Recommend basic regimen
(Most HIV exposures are in this category; no increased risk for HIV transmission has been observed but use of PEP is appropriate). | | 2 | 2 | | Recommend expanded regimen.
(Exposure type represents an increased HIV transmission risk.) | | 3 | 1 or 2 | | Recommend expanded regimen.
(Exposure type represents an increased HIV transmission risk.) | | | UNKNOWN | If the source, (in the case of an unknown source), the setting where the exposure occurred suggests a possible risk for HIV exposure and the EC is 2 or 3, consider PEP basic regimen. |
What specific drugs are recommended for PEP?
Basic Regimen:
(4 weeks therapy) | - | Zidovudine (AZT/ZDV) - 300mg twice a day is used for all types of exposure
+
Lamivudine (3TC) - 150 mg twice a day is added to increase the effectiveness of ZDV and to prevent resistance to ZDV | Expanded Regimen:
(4 weeks therapy) | - | Basic Regimen (AZT/ZDV+3TC)
+
Indinavir - 800 mg/thrice a day or any other protease inhibitor is added for highest risk categories. |
These are the guidelines recommended at present by National AIDS Control Organization, Government of India. However, with the use of anti retroviral therapy for HIV infected patients, the issue of transmission of drug resistant virus (particularly in developed world where large number of infected persons are on triple therapy) is emerging as a serious consideration in choosing drugs for PEP. Detailed guidelines in this regard can be accessed from updated USPHS guidelines issued in June 2001.
However, due to relatively lesser number of HIV infected individuals on triple therapy and an overall low prevalence of HIV in our country, presently above guidelines are being followed. The Physicians dealing with HIV medicines may modify these guidelines depending on the source, risk and their experience.
If the HIV status of the source person is unknown
If the source individual cannot be identified or tested, decisions regarding follow up should be based on the exposure risk and whether the source is likely to be a person who is HIV positive. Follow up HIV testing should be available to all workers who are concerned about possible HIV infection through occupational exposure.
Testing & counselling following exposure
The exposed HCP is tested for HIV immediately following exposure, at six weeks following exposure and again at twelve weeks after the exposure. On all the occasions, HCP must be provided with a pre-test and post-test counselling. HIV testing should be carried out on three ERS (ELISA/Rapid/Simple) test kits or antigen preparations. A complete blood count renal function and liver function tests should be done 2 weeks after starting treatment HCP should be advised to report any flu like illness, fever, rash, muscle aches, swollen glands etc He/She should be advised to refrain from donating blood, semen or organs/tissues and abstain from sexual intercourse. In the case sexual intercourse is undertaken a latex condom be used consistently. In addition, women HCP should not breast-feed their infants during the follow up period.
How soon after exposure to HIV should treatment start?
Treatment should be started promptly, preferably within 1 - 2 hours after the exposure or as soon as possible. Although animal studies suggest that treatment is substantially less effective when started more than 24 - 36 hours after exposure, it is not known if this time frame is the same for humans. Starting treatment after a long period (for example, 1-2 weeks) may be considered for the highest risk exposures; even if HIV infection is not prevented, early treatment of initial HIV infection may lessen the severity of symptoms and delay the onset of AIDS.
How long do the drugs need to be taken?
The optimal course of treatment is unknown; since it has been observed that 4 weeks of ZDV appears to provide protection against HIV infection, if tolerated, treatment should probably be taken for 4 weeks.
How safe are these drugs?
While administering PEP, the important goal is completion of a 4 week PEP regimen. So the toxicity profile of drugs used is a relevant consideration. NRTIs are associated mainly with gastrointestinal side effects like nausea, diarrhoea; however ddI has been associated with fatal and non-fatal pancreatitis among HIV infected patients treated for more than 4 weeks. The use of PI has been shown to be associated with rise in blood sugar, triglycerides, IDV with nephrolithiasis (so more water intake), NFV with diarrhoea, NVP with hepatotoxicity and EFV with dizziness, insomnia etc. All these drugs have interactions with other drugs also.
It has been observed that 50 % of HCP experience adverse symptoms like nausea; malaise, anorexia and headache while taking PEP and approx. 33% stop taking PEP due to adverse signs and symptoms. The side effects are more among HCP taking three-drug regimen as compared to those on two-drug regimen. NVP has been associated with serious hepatic toxicity in a number of cases and is no longer recommended for PEP.
Should pregnant women take these drugs?
Based on limited information available, anti-retroviral therapy taken during 2nd and 3rd trimester of pregnancy have not caused serious side effects in mothers or infants. There is very little information on their safety in the 1st trimester. If the HCP is pregnant at the time of exposure to HIV, the designated authority/physician must be consulted about the use of the drugs for PEP. EFV is not used during pregnancy. D4T and ddI, IDV may be dangerous. However, zidovudine has been found to be quite "safe".
Availability of PEP
In most developing countries of world, PEP regimens are now widely known. However, there might be operational difficulties in early administration of drugs e.g. non availability of drugs resource person after "office hours" of hospital, lack of adequate testing/counselling services, monitoring of PEP toxicity and expert advice in difficult situations.
In our country, National AIDS Control Organisation, Government of India is providing drugs for PEP free of cost to all HCP in all Government Hospitals. An extensive plan has been developed whereby hospital ensures that drugs are accessible all 24 hours at a place in emergency room, more than one resource persons are identified to decide on administering PEP and adequate counselling and testing facilities are available to all categories of HCP. A proper "exposure report" is made in a pre designed form and full follow up of HCP for at least six months is ensured.
Following situations are anticipated wherein an"expert" consultation is sought for PEP:- Delayed exposure report (more than 24-36 hours)
- Unknown source (needle in sharp disposal container or laundry)
- Pregnant (or suspected pregnant) HCPLikely resistance of source virus to anti retroviral agents
- Toxicity of initial PEP regimen
Management of Sexual, Injecting Drug Use and other Non-occupational Exposure to HIV
The most effective methods for preventing HIV infection are the ones that protect against exposure to HIV. Preventive behaviours include sexual abstinence, sex only with an uninfected partner, consistent and correct condom use, abstinence from injecting drug use, and consistent use of sterile equipment by those unable to cease injecting drug use. Some health care providers have proposed offering anti-retroviral drugs to persons with unanticipated sexual or injecting drug use HIV exposure to prevent transmission. However, because no data exist regarding the efficacy of this therapy for persons with non-occupational HIV exposure, it should be considered an unproven clinical intervention. Health care providers and their patients may opt to consider using anti-retroviral drugs after non-occupational HIV exposure that carry a high risk for infection, but only after careful consideration of the potential risks and benefits and with a full awareness of the gaps in current knowledge.
Sexual activities associated with a risk for HIV transmission are also associated with risk for unintended pregnancy and STDs (e.g. syphilis, gonorrhoea, chlamydia, or Hepatitis B Virus). Treatment of STDs should follow the standard WHO/NACO guidelines for treatment of Sexually Transmitted Diseases, and victims of sexual assault should receive additional evaluation and counselling. Women at risk for unintended pregnancy should be offered emergency contraception. Persons with possible HIV exposure through percutaneous routes from sharing syringes or needles should be assessed for hepatitis B and hepatitis C virus infections and considered for hepatitis B vaccination.
Persons who report possible non-occupational HIV exposure should be evaluated for sexual and injecting drug use behaviour that might lead to recurrent exposure. In all situations, health care providers should offer confidential risk reduction counselling during initial and follow up visits. Persons who have been sexually assaulted can be referred to anonymous or confidential voluntary counselling and testing within 72 hours of exposure to establish their HIV status at the time of the assault.
Persons with non-occupational HIV exposure should receive medical evaluations, including HIV antibody tests at baseline and periodically for at least 6 months after exposure (e.g. at 4 weeks, 6 weeks, 12 weeks and 6 months). All persons evaluated for possible non-occupational HIV exposure should be counselled to initiate or resume protective behaviours to prevent additional exposure and to prevent possible secondary transmission if they become infected.
Considerations in initiating anti-retroviral therapy for PEP
Physicians considering the initiation of anti-retroviral therapy in an attempt to reduce the risk for HIV infection in an exposed person should take the following steps in consultation with an expert in the use of anti-retroviral agents:
- Evaluate the HIV status and risk behaviour history of the reported source of HIV exposure.
- Provide medical care, supportive counselling and prevention services to persons who are determined to be HIV infected when they seek care for a potential HIV exposure.
- Evaluate the risk for HIV transmission (if there is convincing evidence of HIV infection in the reported source). Physician should determine the specifics of the risk event (e.g. no condom, torn condom, whether receptive or insertive partner, injection before or after others, number of persons sharing injection equipment) and the presence or absence of factors that would modify risk (e.g. vaginal or anal tears or bleeding, visible genital ulcers or other evidence of an active STD, or bleach treatment of injection equipment).
- Determine the time elapsed between exposure and presentation for medical care. Although animal studies indicate that anti-retroviral agents are most effective within 1 - 2 hours of exposure and probably not effective when started later than 24 - 36 hours after exposure, the interval during which therapy can be beneficial for humans is unknown
- Evaluate the frequency of HIV exposure. Uninfected persons who request anti-retroviral agents should be evaluated for sexual, injecting drug use and other behaviours that might lead to recurrent HIV exposures. Anti-retroviral therapy is not a replacement for adherence to behaviours that reduce the risk of HIV exposure.
Provide counselling and obtain informed consent. Because post-exposure prophylaxis is an experimental therapy of unproven efficacy, informed consent should be obtained and recorded in the medical charts of all persons prescribed anti-retroviral agents following non-occupational exposure. Such consent should document the patient's understanding of
- The need to initiate or resume relevant HIV risk-reduction behaviours (e.g. condom use and /or drug treatment)
- The limited knowledge about the effectiveness and toxicity of anti-retroviral treatment for non-occupational exposure.
- The known side effects of the medications being prescribed.
- The name and phone number of a source for follow up medical care
- The frequency and timing of recommended follow up HIV testing
- The signs and symptoms associated with acute HIV seroconversion; and
- The need for adherence to prescribed medications to maximise efficacy and reduce the risk for infection with a drug resistant variant.
Persons younger than 16 years at the time of exposure should be evaluated (before therapy is initiated) by paediatricians, family physicians or other clinicians expert in the specific medical needs, consent issues and other factors involved in their treatment, including the use of anti-retroviral medicines for children and adolescents.
If anti-retroviral therapy is used, drug toxicity monitoring should include a complete blood count and renal and hepatic function tests when therapy is initiated and again 2 week after the patient begins to take the medications. It is possible that anti-retroviral therapy during early HIV infection could benefit the patient by reducing the initial level of viral replication (i.e. the set point) and decreasing the extent of lymph node infiltration. Thus, for patients with the highest risk exposures, health care providers may consider continuing therapy until HIV test results are received from a specimen drawn after 28 days of treatment. Patients should be monitored for signs and symptoms of acute HIV infection during therapy. If such conditions develop, the patient should be tested for HIV (p24 antigen, HIV viral load assays) during their 4-week course of therapy with confirmation by standard HIV antibody tests. Persons who become infected while taking anti-retroviral therapy should be advised to continue taking the medication pending transfer to a health care provider who specialises in long term HIV care.
There has been apprehension that availability of PEP may cause people to stop practising safe sex or stop safer needle use. HIV negative persons may wonder, "why bother with condoms or clean rig" when therapy to prevent HIV is available. HIV transmission could increase with these attitudes. So it must be emphasized through media campaign and counselling that no PEP therapy is 100% effective, it has its own side effects and it needs strict adherence. Hence the stress should be on preventive measures through safe sex and safer needle use. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
| Cite this article as: | | Pensi T. POST-EXPOSURE PROPHYLAXIS (PEP). Pediatr Oncall J. 2005;2. |
|