Introduction
There is no really straightforward or simple formula that can be adapted to treat a child with diabetes mellitus (>99% of children with diabetes mellitus have type 1 diabetes). Definitely, a patient with type 1 diabetes mellitus who has overt hyperglycemia and glycosuria must be treated with insulin. Insulin acts directly to counteract the metabolic defects of insulin deficiency. Additionally, there is now evidence that early, aggressive treatment with insulin may have a beneficial effect on progression to total insulin deficiency in type 1 diabetes by decreasing islet cell antigenicity and autoimmune destruction of b cells1. For this reason, although temporary use of an oral hypoglycemic agent is possible in the early stages of type 1 diabetes, insulin is preferred.
It is pertinent to mention that newly diagnosed type 1 diabetics who receive insulin are at a greater risk of hypoglycemia and require small doses of insulin because some recovery of residual insulin secretion is possible. In the first few months of therapy, it is not unusual to see a decline, to very low levels, of the daily dose of insulin necessary for glycemic control, and in many cases, the use of insulin could be stopped temporarily. This remission is often called "Honeymoon Phase" and may last for a few weeks to several months. During this period it is advisable to continue therapy with insulin at a low dose rather than stop it entirely because intermittent use of insulin can increase its antigenicity and as mentioned above, uninterrupted insulin therapy may have a beneficial effect in preserving beta cell function.
Advantages and Disadvantages of Intensive Insulin Therapy
Table 2: Intensive Insulin Therapy
ADVANTAGES:
- More rational control of blood glucose with the ability to adjust insulin doses to change in diet, activity and illness
- Delay in onset of and progression of: Retinopathy, Nephropathy (proteinuria and microalbuminuria), Neuropathy
- Reduced risk of hypercholesterolemia and hypertriglyceridemia
- Improved sense of well being
- Decreased fetal and maternal morbidity during pregnancy
DISADVANTAGES:
- Increased episodes of severe hypoglycemia
- Hypoglycemia unawares
- Increased weight gain
- Transient exacerbation of pre-existing retinopathy (rare in children)
- Increased time, effort and cost
- In pump patients, increased ketoacidosis, and infection at infusion site
- Less suitable in young children (<7 years)
The major disadvantage of intensive insulin therapy is that there is a distinct increase in the incidence of clinically significant hypoglycemia. Furthermore, it may be associated with hypoglycemia unawareness. These effects may be attributable to an increased threshold of the secretory response of adrenaline to hypoglycemia and enhanced suppression of glucogenesis in the liver by insulin5,6. Intensive insulin regimens are associated with a significantly greater weight gain than standard therapy7 and these children may be at a significant risk of developing obesity. Careful attention has thus to be given to diet and caloric restriction in these patients. Transient exacerbation of retinopathy has been reported with intensive insulin therapy but this is rarely if ever, encountered in the pediatric age group. That small subset of patients who are on insulin pump therapy(continuous subcutaneous insulin infusion-CSII) may face the likelihood of increased risk of diabetic ketoacidosis due to pump failure and an increased incidence of infection at the pump infusion site8. As great vigilance and frequent adjustment of doses are required for this type of regime, it is not generally recommended for very small children. Furthermore, intensive insulin regimes require a great deal of time, motivation, and effort on the part of the patient. They have added to be supported by a dedicated diabetes team especially to prevent frequent episodes of hypoglycemia. Notwithstanding the above, intensive insulin regimes have come to stay and are perhaps the ideal mode of therapy for juvenile diabetics as they are the ones who will inevitably face both microvascular and macrovascular complications in their adult life.
Insulin Therapy - Therapeutic Choices
If the child presents to the doctor with extreme hyperglycemia or diabetic ketoacidosis, the initial treatment is with human regular insulin infused intravenously. Once the blood glucose levels and electrolyte levels are stabilized and ketones are cleared from the circulation, they can be switched to a daily regime of subcutaneous insulin injections. Patients, who present with a lesser degree of hyperglycemia not associated with significant ketosis, can initially be treated with insulin directly as outpatients. Care must be taken at this time to ensure that patients receive a thorough education of how to use insulin, understand the rules for adjustment of insulin doses, learn to recognize and treat hypoglycemia, understand the relation of insulin to diet and exercise, are instructed on sick day guidelines and are familiarized with other aspects of diabetes such as foot care, dietary guidelines and hygiene (Table 3). Children, adolescents, those with acute illness, or learning problems are better hospitalized initially. This can help in familiarizing the child and the parents with insulin injection techniques and intensive education can be given over several days as well as insulin dosage adjustments can be made under the supervision of a physician.
Table 3: Patient Education at First Visit/Initial Hospitalization
- Insulin injection technique and sites
- Insulin storage, instructions during travel and while at school
- Recognition and treatment of hypoglycemia
- Rules for adjusting insulin dosages, especially in relation to exercise.
- Diet counseling. Importance of meal timings and snacks.
- Sick day guidelines
- Foot care and hygiene
Types Of Insulin
With the advent of human insulins, bovine or porcine insulins have hardly any role to play in the management of type 1 diabetes in children. Avoiding the use of bovine and porcine insulins can successfully obviate the attendant problems of insulin resistance, insulin antibodies, and lipoatrophy.
The introduction of rapid-acting insulin analogs, Lispro, and Aspart insulin has further facilitated intensive glycemic control by their use in the MDI regimes16-18 (Fig 2). Further, substituting regular insulin with these analogs can solve many a problem encountered in achieving adequate glycemic control. The foremost among these is a marked reduction in nocturnal hypoglycemic episodes. They also have a special role to play in cases of Somogyi phenomena, Dawn phenomena, and brittle diabetes.
Ultralente insulin (bovine) was the closest we had to good basal insulin. However, it was still woefully inadequate. The introduction of Glargine insulin has shown great promise as it provides the near-ideal, once a day, peakless, basal insulin.
Pulmonary aerosol (inhalant) insulin is the next major blessing for young diabetics19. With its introduction, giving a single bedtime dose of Glargine insulin and metered doses of inhaled insulin prior to each meal/snack may further facilitate intensive control. [Fig 2: Mean serum insulin concentration in non - diabetic subjects after subcutaneous injection (0.2 U/kg) of human insulin Aspart (*) and regular human insulin (o ) at various sites. A. Abdomen B. Deltoid C. Thigh. (20)
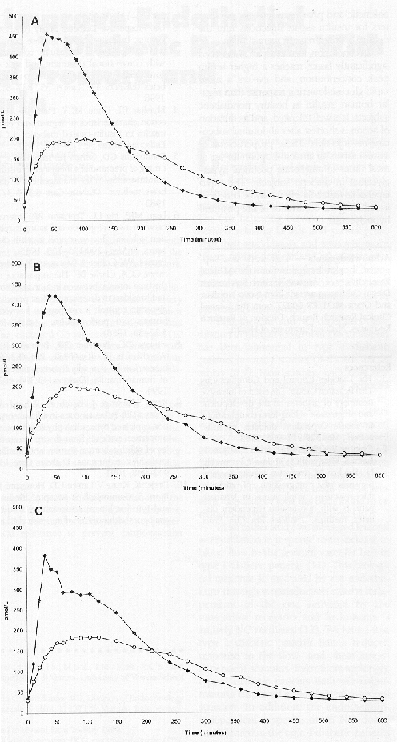 ]
]
Goals Of Therapy
The major aim of therapy in type 1 diabetic children in the prevention of symptoms of hyperglycemia (osmotic and other symptoms) and prevention of diabetic ketoacidosis as well as hyperosmolar coma (Table 1). Inevitably if diabetes has been of some duration, most children will exhibit retardation in growth parameters (height, weight, and height velocity). Adequate insulin therapy will ensure catch-up growth so that these children can achieve meaningful growth and development rates, similar to their peers. Along with this, there should be a concurrent increase in their exercise capacity and a decrease in the incidence and severity of common infections. Associated with these significant changes is a sense of well being. Until recently, it was debated whether intensive insulin therapy that aims at bringing blood glucose levels to near the normal range is safe and whether it can ameliorate or prevent the development of long term microvascular and macrovascular complications of diabetes mellitus. However, with the publication of a few landmark studies like DCCT2, Kumamoto3, and UKPDS4, this debate has been resolved. In the DCCT study, there was a significant reduction in all components of microvascular disease with good glycemic control (retinopathy-76%, proteinuria-56%, microalbuminuria-46%, neuropathy-61%). However, the results regarding macrovascular disease were not so dramatic and did not reach statistical significance.
Table 1: Goals of Insulin Therapy
- Elimination of hyperglycemia symptoms
- Prevention of diabetic ketoacidosis and hyperosmolar coma.
- Restoration of lean body mass, height velocity and weight.
- Improvement in exercise capacity and work performance.
- Reduction in frequency of infections (improvement in immunological defense).
- Delay, arrest or prevention of microvascular and macrovascular complications of diabetes mellitus.
Single Dose Regimes
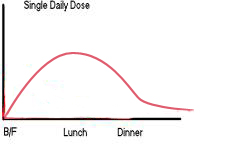
This is mentioned as it is frequently used, but rarely, if ever, is it possible to achieve glycemic control with this. The starting dose is usually in the range of 0.2-0.3 U/Kg per day. Most patients will require much larger doses of approximately 0.5-1.0 U/Kg per day as they have no endogenous insulin secretion, but it is advisable to start with lower doses as the patient may develop hypoglycemia and because the patient may have some endogenous insulin secretion intact. The choice of insulin is usually a combination of regular insulin (approximately 30%) with intermediate-acting insulin (NPH or Lente) given about 30-45 min prior to breakfast (Fig 1A). The insulin dosages are gradually adjusted as per self-monitoring of blood glucose (before breakfast, late afternoon [post lunch], and pre-dinner). This regime may be of some use in those patients who have some endogenous insulin secretion, or those that are in their honeymoon phase, but as mentioned, more often than not the patient has to be shifted to twice daily or multiple daily injection regimes to achieve glycemic control9,10 FIGURE 1: A- Single Dosage Regime, B- Twice Daily Regime, C- Multiple Daily Regime, D- Normal Physiological Insulin Secretion in Non Diabetic Individual.
Twice Daily Regimes
Patients on a single daily injection regimen often have high fasting glucose but low or normal glucose levels in the afternoon and evening. This is because both NPH and Lente insulin has a duration of action lasting between 8-18 hours. In such a situation the second injection of intermediate-acting insulin can either be given singly at bedtime or in combination with regular insulin before dinner (Split mix regime). Many diabetologists initiate insulin therapy with a minimum of two injections (Fig 1B). Although this does not mimic the physiological secretion of insulin, yet it is more likely to achieve a fair glycemic control than with a single injection. The disadvantage of this regime is that it may increase the risk of hypoglycemia, especially during the middle of the night or at 3 am in response to the action of the evening dose of the intermediate-acting plus regular insulin.
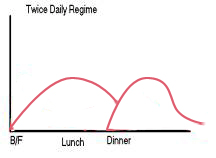
Twice daily insulin regimes are commonly used because of their convenience and simplicity for the patient. However, this advantage is often offset by the lack of flexibility with just two doses of insulin, adjustments for a change in diet and physical activity can sometimes be difficult. This is especially applicable to the school going children and adolescents, where often they have no control over either their meal timings or an exercise schedule. The hypoglycemic effect of the intermediate-acting insulin mandates scheduling lunch and dinner at fairly precise times, in addition to breakfast, mid-morning and mid-afternoon snacks. The dietary context of each meal, especially of carbohydrates must be kept fairly constant in order to avoid hypoglycemia. Unscheduled physical activity or exercise can make such children especially prone to hypoglycemia.
Quite often when NPH or Lente insulin is given before dinner, the peak action and duration are too short to last throughout the night, resulting in hypoglycemia at about 3 am and hyperglycemia in the early morning11. In addition, hepatic glucose output in the early morning hours increases in association with an increase in the basal insulin requirement, which is thought to be the consequence of pulses of growth hormone secreted a few hours after onset of sleep. The problem often teemed as "DAWN PHENOMENON", results in elevated fasting blood glucose. The situation can be partially remedied by giving the intermediate-acting insulin at bedtime, rather than before dinner. Alternatively, ultra Lente insulin can be given before dinner. However, this often complicates the issue as the patient has to use these different types of insulin in a day.
Multiple Daily Injections
For a tighter control of blood glucose, usually, a minimum of three daily injections is required (Fig 1C). Intensive therapy implies that not only are the symptoms of diabetes alleviated but also the average blood glucose values are maintained in a range as close as possible to the normal non-diabetic individual. For this to be achieved insulin must be given in a manner that duplicates the diurnal patterns of insulin secretion from the islets in the basal and postprandial states (Fig 1D). To achieve this the basal insulin levels have to be, maintained throughout the 24 hours by use of intermediate or long-acting insulin and postprandial peaks have to be provided by boluses of regular insulin or fast-acting insulin analogs. This can be achieved in a number of ways as follows:
- A mixture of intermediate and short-acting insulin before breakfast, regular insulin at lunch and again a mixture of short-acting and intermediate-acting insulin at dinnertime.
- Using regular insulin before each meal and intermediate-acting insulin at bedtime.
- Regular + intermediate insulin before breakfast, regular before Lunch and dinner, and intermediate at bedtime.
- Using regular insulin before each meal and long-acting insulin such as ultralente or insulin Glargine (Lantus), once a day.
The most commonly used regime is the mixture of intermediate-acting (NPH or Lente) with regular insulin before breakfast, regular insulin at lunch, and a similar mixture as used at breakfast before dinner. This is easily evolved from the split mix regime and is especially useful for patients having night hypoglycemia. Modifications involve the use of Lyspro or Aspart insulin analogs instead of regular insulin for a relatively better and fast postprandial control12.
The second regime, where regular or fast-acting insulin analogs are used prior to each meal and intermediate-acting insulin is given at bedtime enables the patient to vary their meal times and offers much more flexibility. The patients can also vary their doses to cover individual variations in carbohydrate content of their meals and exercise schedules.
Algorithms have been designed to determine the dose of regular insulin to be administered prior to the meal, depending on the premeal blood glucose value (Table 4). These are guidelines mainly for initiating the therapy. Sooner or later patients and their physicians learn to adapt to the individual needs and the final dosing may be well different from that shown in the algorithm. The availability of pen-type insulin injections has greatly facilitated the adoption of this regime.
Good glycemic control can also be achieved by using ultralente and now Glargine insulin (Lantus) instead of intermediate-acting insulin to cover the basal needs, with regular insulin or fast-acting insulin analogs given before each meal. The ultralente insulin may be given in a single dose, but is more effective if divided into two injections before breakfast and dinner. Insulin Glargine can be given as a single dose at bedtime as it has a flat curve and is effective over 24hrs13, 14. Approximately half the total dose of insulin is given in the long-acting form, but the proportion will vary from patient to patient.
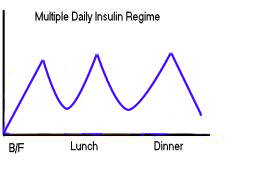
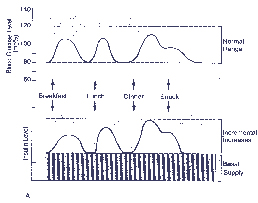
Table 4: Intensive Insulin Therapy Algorithm. Example of Four Injections a Day Regime
|
Regular Insulin Pre-meal Dose |
| Blood glucose(mg/dl) |
Breakfast units |
Lunch units |
Dinner units |
Bedtime units |
| 0-50 |
4 |
3 |
3 |
- |
| 51-100 |
6 |
5 |
5 |
- |
| 101-150 |
7 |
6 |
6 |
- |
| 151-200 |
8 |
7 |
7 |
- |
| 201-250 |
9 |
8 |
8 |
2 |
| 301-350 |
11 |
10 |
10 |
4 |
| 351-400 |
12 |
11 |
11 |
5 |
| >400 |
13 |
12 |
12 |
6 |
*These values are representative of what a patient with type 1 diabetes, weighing 50 kgs, may require. The actual insulin requirement, as well as gradation in the algorithm, will vary widely as per age, the weight of the child, and from patient to patient.
Goals of Intensive Therapy
In all the regimes mentioned above, fasting or premeal blood glucose levels are used as a guide for adjustment of the dose of whatever the type of insulin is used. Fasting blood glucose levels should ideally be kept between 70-120 mg/dl for optimal control. In younger children, such rigid control may be associated with unacceptable episodic hypoglycemia and more liberal values between 80-140 mg/dl are acceptable. To avoid nocturnal hypoglycemia, patients should check the blood glucose levels at 3 am once a week or so and ensure that it is above 70 mg/dl. Patients should be willing to check their blood glucose levels four times a day, before each meal and at bedtime. Glucose levels before each meal should be generally within the same range as the fasting blood glucose levels. In addition, a 2hr postprandial blood glucose level should be monitored with a goal of maintaining it between 80-150 mg/dl. Careful adherence to the monitoring schedule with frequent communication with the physician and diabetic nurse is essential for the success of intensive insulin regimes.
Reverse Split Insulin
Certain patients tend to have high pre-dinner and post-dinner blood glucose values, with normal values at other times. In such cases, either intermediate-acting insulin may be given prior to lunch or a reverse split mixture with 30% intermediate-acting, and 70% regular insulin may be given at the same time. Such insulins, as mixtures of NPH and lyspro, are likely to be used in the future. Use of lyspro and aspart insulin results in good postprandial blood glucose regulation but poor inter-prandial glycemic control. Hence, 20-30% of intermediate-acting insulin with lyspro at each meal, particularly at breakfast and lunch, in the form of reverse split insulin will prevent inter-prandial blood glucose rise. The usually mixed insulin (30-50% regular and 70-50% NPH) is the more suitable insulin at a pre-dinner time.
Special Situations: Brittle Diabetes
Treating type 1 diabetes is beset with a number of problems. The major amongst these are brittle diabetes, where the patient has intermittent very highs and very lows. These patients either have marked and often incapacitating excursions of blood glucose levels on a daily basis or have frequent decompensation in their glycemic control often with recurrent ketoacidosis or hypoglycemia that significantly interferes with their lifestyles. Of particular concern is the apparent randomness of the blood glucose excursions, which do not show any diurnal pattern or predictable response to changes in diet or exercise. The problem has multiple causes. Insulin resistance with high titers of circulating insulin antibodies causing abnormalities in the kinetics of insulin action is one such cause. However, with the use of human insulin and insulin analogs such as Lispro insulin, this has been relatively uncommon. More common, however, are abnormalities in glucose counter-regulation such as Somogyi and Dawn phenomenon and hypoglycemia unawareness. Chronic undiagnosed infections, especially pulmonary tuberculosis is one such setting, and underlying hormonal disorders such as acromegaly, glucocorticoid therapy, thyroid dysfunction, isolated or multiple pituitary deficiency, and pheochromocytoma need exclusion. Gastroparesis can interfere with nutrient absorption and affect glycemic control. Poor injection technique and/or confusion by the patient as regards to the times of the dosages of insulin are often implicated. Of particular importance are psychosocial problems that lead to factitious disease (surreptitious administration of insulin, with no obvious gain or motive). Manipulative behavior is often seen in adolescents and poor glycemic control is often associated with eating disorders such as bulimia and anorexia nervosa. Patients concerned about weight gain in relation to insulin therapy discover that they can lose weight by ignoring their insulin injection
Brittle diabetes is best managed by a detailed history and careful physical examination by a physician who is familiar with these problems. Appropriate investigations, psychological evaluation, and patient's dietary and exercise habits have to be adequately evaluated. If necessary, the patient should be hospitalized for stabilization and investigations. Most such cases can be controlled with MDI regimes and the use of short-acting insulin analogs such as Lispro insulin, after treating the precipitating or aggravating factors. Continuous subcutaneous insulin infusion may be required in a subset of these patients. Behavioral therapy and psychotherapy may be of additional help.
Hypoglycemic Unawareness
Though this problem is infrequently encountered in the pediatric age group, one has still to be concerned as hypoglycemia can be particularly debilitating in the young child. Usually, this complication develops five to ten years after the diagnosis of type 1 diabetes and is due to impairment of the counter-regulatory response of noradrenaline, adrenaline, and glucagon. The prevalence of deficient adrenaline secretion in-patients with long-standing diabetes may be as high as 40% (15) and is usually due to associated autonomic neuropathy. This problem is most often encountered with intensive insulin therapy and often resolves when the intensive regimes are discontinued. In such patients, higher targets for blood glucose control should be set (fasting and premeal blood glucose levels up to 150 mg/dl).
1. Shah SC, Malone JI, Simpson NE. A randomized trial of intensive insulin therapy in newly diagnosed insulin dependent diabetes mellitus. N Eng J Med 1989; 320-4.
2. The Diabetes Control and Complication Trial research group. The effect of intensive treatment of diabetes on the development of progression of long-term complications in insulin dependent diabetes mellitus. N Eng J Med 1993; 329; 977-86.
3. Ohkubo Y, Kishikawa H, Araki E et al. Intensive insulin therapy prevents progression of diabetic microvascular complications in Japanese patients with non-insulin dependent diabetes mellitus: a randomized prospective six year study. Diabetes Res Clin Pract 1995; 28; 103-17.
4. Turner R, Cull C, Holman R. United Kingdom Prospective Diabetes Study 17: A nine year update of a randomized control trial on the effect of improved metabolic control on complication in non insulin dependent diabetes mellitus. Ann Intern Med 1996; 33; 438-43.
5. The DCCT research group. Epidemiology of severe hypoglycemia in the diabetes control and complications trial. Am J Med 1991; 90; 450-9.
6. Amiel SA, Tamborlane WV, Simonson DC et al. Defective glucose counter regulation after strict glycemic control of insulin dependent diabetes mellitus. N Eng J Med 1987; 316; 1376-83.
7. The DCCT research group. Weight gain associated with intensive therapy in diabetes control and complication trial. Diabetes Care 1988; 11; 567-73.
8. Mecklenberg RS. Acute complications associated with the use of insulin infusion pumps. Endocrinologist 1991; 1; 19-24.
9. Chandalia H. B. Insulin therapy in Diabetes Mellitus. Bombay Hosp. Jour. 18 : 39, 1976.
10. Chandalia H. B.: Insulin Delivery Systems In Current concepts in Diabetes Mellitus. Ed : Sainani G. S., Indian College of Physicians, Bombay, 1993, p. 131-142.
11. Gale EAM, Tattersall RV. Unrecognized nocturnal hypoglycemia in insulin treated diabetics. Lancet 1979; 1; 1049-52.
12. Owens DR, Barnett A. Designer insulins: have they revolutionized insulin therapy? In Batterdge JD ed. Diabetes-current perspectives. London; Martin Dunilitz 1999.
13. Heinemann L, Linkeschova R, Rave K et al. Time action profile of the long acting analogue insulin Glargine (HOE 901) in comparison with those of NPH insulin and placebo. Diabetes Care 2000; 23; 644-9.
14. Dashora U, Dashora V. insulin Glargine. Int J Diabetes Developing Countries 2000; 20; 140-4.
15. Herold KC, Polonsky KS, Cohen RM et al. Variable deterioration in cortical function during insulin-induced hypoglycemia. Diabetes 1985; 34; 677-85.
16. Howey DC, Bowsher RR, Brunelle RL et al. [Lys (B28)Pro(B29)] Human insulin. A rapidly absorbed analogue of human insulin. Diabetes 1994; 43; 396-402.
17. Fineberg SE, Fineberg NS, Anderson JH et al. Insulin immune response to LYSPRO human insulin therapy in insulin native type 1 and type 2 patients. Diabetologia 1995; 38(Suppl 1): A78.
18. Uwe B, Ebraham S, Hirschberger S et al. Effect of rapid acting insulin analogue, insulin Aspart on quality of life and treatment satisfaction in type 1 diabetic patients. Diabetes 1999; 48 (Suppl 1); Abs 0481; A112.
19. Weiss SR, Berger S, Cheng S-L et al. Adjunctive therapy with inhaled human insulin in type 2 diabetic patients failing oral agents. A multicentric phase II trial. Diabetes 1999; 48 (Suppl 1) A 12.
20. Mudaliar SR, Strange P. Lindberg FA et al. Insulin Aspart : a fast acting analogue of human insulin. Diabetes Care 1999; 22; 1501-6.