Harvinder K Palaha1, Prerna Mewawalla2, Vidhyachandra Gandhi3, Anaita Hegde1, Ritu Kakkar4, Aabha Nagral2.
1Department of Pediatrics, Jaslok Hospital & Research Centre, Mumbai,
2Department of Gastroenterology, Jaslok Hospital & Research Centre, Mumbai,
3Department of Gastrointestinal Surgery, Jaslok Hospital & Research Centre, Mumbai,
4Department of Radiology, Jaslok Hospital & Research Centre, Mumbai.
ADDRESS FOR CORRESPONDENCE
Dr. Aabha Nagral, 7, Snehasagar, Prabhanagar, Prabhadevi, Mumbai 400 025, India.
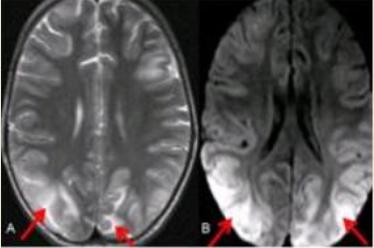
Email: aabhanagral@gmail.com | | Abstract | | We report a 6 year old liver transplant recipient who developed seizures, altered behavior and cortical blindness (Posterior Reversible Encephalopathy Syndrome) on the tenth day after transplantation. This was related to tacrolimus and hypertension, which completely reversed with reduction of the tacrolimus dose and controlling the hypertension. | | | | Keywords | | India, tacrolimus, biliary atresia | | | | Introduction | | A wide spectrum of neurological complications can occur in patients undergoing transplantation. We report a 6 year old child presenting with multiple neurological symptoms. | | | | Case Report | A 6-year-old girl underwent Living related Liver Transplantation for secondary biliary cirrhosis. She had undergone a Kasai procedure for biliary atresia at the age of 2 months. In the immediate postoperative period after transplantation, she received antibiotics (piperacillin and tazobactum) and antifungals (fluconazole). She was immunosuppressed with tacrolimus and methylprednisolone. In the early postoperative period, she developed small bowel perforation, which required exploration and closure. On postoperative day 10 of the transplant, the child was found to be restless and had an arterial blood pressure of 172/72 (mean BP 82mm of Hg), which was above 90th centile for her age. She was started on amlodipine and investigated for the cause of hypertension. The serum tacrolimus trough level was 22 mg/dl (desirable range 10-12 mg/dl) and serum magnesium level was 1.1 mg/dl (normal 1.3-2.1 mg/dl). On the next day she developed twitching at the right angle of mouth and psychosis like symptoms. Usually a quiet and withdrawn child, she suddenly became boisterous, talking irrelevantly and excessively. She also developed complete blindness with no perception of light. In view of the hypertension, convulsions and abnormal behavior, an magnetic resonance imaging (MRI) of the brain was advised. The MRI scan revealed T2W and FLAIR hyperintensities in bilateral posterior parietal and occipital cortex (figure 1 A) and subcortical white matter with restricted diffusion in these areas (figure 1 B). This confirmed the diagnosis of Posterior Reversible Encephalopathy Syndrome. (PRES). The child was managed with reduction of the dose of tacrolimus to maintain a serum level between 5-7 mg/dl, magnesium correction and control of her blood pressure. She recovered completely within 4 to5 days and regained normal vision. She continues to remain asymptomatic on follow up.
Figure 1. A - Axial T2 Weighted (WI) MR image showing hyperintensities in bilateral posterior parieto occipital cortex and subcortical white matter (red arrows). B-Axial diffusion T2 WI MR image showing restriction of diffusion in bilateral posterior parieto occipital cortex and subcortical white matter (red arrows)
 | | | | Discussion | Tacrolimus is now the most commonly used immunosuppressive drug to prevent rejection in the solid organ and hematopoietic stem cell transplantation. Neurotoxicity is a significant complication with the use of tacrolimus and is well described in solid organ transplant recipients (1). It causes headache, tremors, insomnia and paresthesia. One of the rare neurological complications is posterior reversible encephalopathy syndrome, which occurs in 1.6% of patients receiving tacrolimus (2). PRES has been recognized in setting of severe hypertension after solid organ transplantation (3). Patients typically present with altered mental status, headache, focal neurological deficits, visual disturbances, and seizures. Our patient received tacrolimus after liver transplantation, following which she developed visual disturbances, hypertension and psychosis like symptoms. These findings correlated with a high tacrolimus level. After liver transplantation, tacrolimus level is usually maintained between 10-12 mg/dl. tacrolimus also causes hypertension and low magnesium levels, which was also observed in our patient. It has been suggested that hypomagnesemia may augment PRES (4). Our patient had hypertension, which could have been caused due to tacrolimus/methylprednisolone, and it also would have acted as an individual precipitating factor of PRES. In addition, the low magnesium levels could also be a result of high tacrolimus levels and augment PRES.
The mechanism by which tacrolimus causes this syndrome is not very clear. But it is widely believed that immunosuppressive therapy causes perturbation of blood brain barrier. It induces diffuse endothelial injury with systemic effects. Vasoconstriction develops (endothelin, sympathetic) with altered glomerular filtration, glomerular endothelial injury, proteinuria, and magnesium loss (resembling preeclampsia). A breakdown in cerebral autoregulation results in the leakage of fluid into the interstitium, which is detected as vasogenic edema (5).
The posterior part of the brain is more frequently involved and damaged since the circulation in this area is supplied by vertebro-basilar system, which has poor sympathetic innervation. MRI is the most sensitive imaging test to diagnose PRES. Typical lesions predominate in the posterior white matter, with some involvement of the overlying cortex. These lesions appear hyperintense on T2-weighted images; and are usually hypointense or isointense on diffusion-weighted images, with an increase of the apparent diffusion coefficient, indicating vasogenic edema (6). Our patient showed T2W and FLAIR hyperintensities in bilateral posterior parietal and occipital cortex and subcortical white matter with restricted diffusion in these areas.
Management includes prompt correction of the underlying precipitating factor like hypertension, by reducing the dose or withdrawal of the offending drug and correcting the magnesium level. Prompt recognition and treatment of PRES is important, as most patients will experience complete recovery. Our patient also responded well to these therapeutic interventions and recovered without any sequelae. When left untreated, the damage could be permanent. Hence it is vital, to diagnose it early and remove the offending agent. Previously reported cases of PRES in India included patients of acute lymphoblastic leukemia, nephrotic syndrome and pheochromocytoma (7-9).
We believe that this is the first documented report of PRES in a solid organ or hemopoetic cell transplantation in India. | | | | Financial Disclosure | | none | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest | | none | | |
- Eidelman BH, Abu-Elmagd K, Wilson J, Fung JJ, Alessiani M, Jain A et al. Neurologic complications of FK-506. Transplant Proc. 1991; 23: 3175-3178. [PubMed] [PMC free article]
- Wong R, Beguelin GZ, De Lima M, Giralt SA, Hosing C, Ippoliti C et al. Tacrolimus-associated posterior reversible encephalopathy syndrome after allogeneic haematopoietic stem cell transplantation. Br J Haematol. 2003; 122: 128-134. [CrossRef]
- Bartynski WS, Tan HP, Boardman JF, Shapiro R, Marsh JW. Posterior reversible encephalopathy syndrome after solid organ transplantation. AJNR Am J Neuroradiol. 2008; 29: 924-930. [CrossRef]
- Thompson CB, Sullivan KM, June CH, Thomas ED. Association between cyclosporine neurotoxicity and hypomagnesaemia. Lancet. 1984; 2: 1116-1120. [CrossRef]
- Bartynski WS. Posterior reversible encephalopathy syndrome, part 2: controversies surrounding pathophysiology of vasogenic edema. Am J Neuroradiol. 2008; 29: 1043-1049. [CrossRef]
- Lamy C, Oppenheim C, Meder JF, Mas JL Neuroimaging in Posterior Reversible Encephalopathy Syndrome. J Neuroimaging. 2004; 14: 89-94. [CrossRef]
- Gupta A, Swaroop C, Rastogi R, Garg R, Bakhshi S. Simultaneous occurrence of posterior reversible leukoencephalopathy syndrome in two cases of childhood acute lymphoblastic leukemia induction chemotherapy. Pediatr Hematol Oncol. 2008; 25: 351-358. [CrossRef]
- Archana B. Patel, Samiuddin Shaikh. Posterior reversible encephalopathy syndrome. Indian Pediatr. 2006; 43: 657-658.
- Sanjay KM, Partha PC. The posterior reversible encephalopathy syndrome. Indian J Pediatr. 2008; 75: 953-955. [CrossRef]
|
| Cite this article as: | | Palaha H K, Mewawalla P, Gandhi V, Hegde A, Kakkar R, Nagral A. Posterior Reversible Encephalopathy Syndrome after liver transplantation. Pediatr Oncall J. 2010;7: 73-74. |
|