|
Neurocutaneous Syndrome
Spoorthi Jagadish, Tanavi Ghadage.
Department of Pediatrics, Sir JJ Group of Hospitals, Mumbai, India.
ADDRESS FOR CORRESPONDENCE
Dr Spoorthi Jagadish, Thejas J B Lobo Road, Kodikal, Mangalore, Karnataka, India.
Email: spoojagadish@gmail.com
Figure 1a
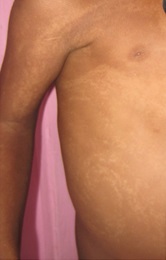
Figure 1b
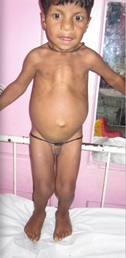
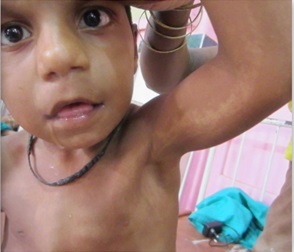
Figure 1c
|
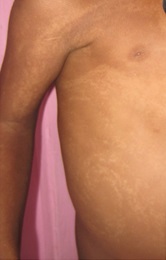
Figure 1a
|
What is the diagnosis?
Hypomelanosis of Ito. It is a rare neurocutaneous syndrome with hypopigmented whorls of skin along the Blaschko lines {1}, first described by Ito in 1951 {2} with a female: male ratio of 2:1 and no racial predilection. It was earlier also known as incontinent pigmenti achromians, because it appears as the negative image of incontinent pigmenti. The varied phenotype is probably due to genetic mosaicism. {3} Sporadic mutations also contribute to it. It is present at birth, or soon after and is classically described as hypochromic lesions in distinctive patterns {e.g. whorls, patches, streaks}. The lesions may be unilateral {46 percent} or bilateral and usually show a midline cutoff mostly on the ventral surface of the trunk and flexor surface of the limbs and are easily demonstrable by wood lamp examination. The skin lesions may later become pigmented and blend with the rest of the skin. It is associated with various other congenital abnormalities with mental retardation and seizures being the most commonly reported in literature. {4} Other abnormalities include hypertelorism, visual disturbances, cleft palate and lip, anodontia, renal anomalies, various musculoskeletal malformations like scoliosis, limb hypertrophy and syndactyly. Neuro imaging should be done to rule out associated structural abnormalities. There is no specific treatment and comprises of management of co-morbidities by a multidisciplinary approach. Traditional depigmenting agents {e.g. hydroquinone, corticosteroids, kojic acid} may be used. {5} |
| |
| Compliance with ethical standards |
|
Funding: None
|
|
|
Conflict of Interest: None
|
|
- Nehal KS, PeBenito R, Orlow SJ. Analysis of 54 cases of hypopigmentation and hyperpigmentation along the lines of Blaschko. Arch Dermatol. 1996;132:1167-1170 [CrossRef] [PubMed]
- Ito M. A singular case of naevus depigmentosus systematicus bilateralis. Jpn J Dermatol. 1951;61:31-32
- Gupta S, Shah S, Mcgaw A, Mercado T, Zaslav AL, Tegay D. Trisomy 2 mosaicism in hypomelanosis of Ito. Am J Med Genet A. 2007;143A:2466-2468 [CrossRef] [PubMed]
- Assogba K, Ferlazzo E, Striano P, Calarese T, Villeneuve N, Ivanov I, et al. Heterogeneous seizure manifestations in Hypomelanosis of Ito: report of four new cases and review of the literature. Neurol Sci. 2010;31:9-16 [CrossRef] [PubMed]
- Zhu W, Gao J. The use of botanical extracts as topical skin-lightening agents for the improvement of skin pigmentation disorders. J Investig Dermatol Symp Proc. 2008;13:20-24 [CrossRef] [PubMed]
|
|
DOI: https://doi.org/10.7199/ped.oncall.2014.44 |
| |
Cite this article as:
Jagadish S, Ghadage T. Neurocutaneous Syndrome. Pediatr Oncall J. 2014;11: 93. doi: 10.7199/ped.oncall.2014.44
|