Ramya Madhavan, Maheswari Srinivasan, John Matthai.
Masonic Hospital for children, Coimbatore, India.
ADDRESS FOR CORRESPONDENCE
Dr.Ramya Madhavan, 232, Race Course Road, Coimbatore - 641018, India.
Email: drramyavijay82@gmail.com | | Abstract | Objective: To determine the incidence, risk factors, and severity of oral antibiotic-associated diarrhoea (AAD) in outpatient children aged 1 month to 5 years.
Methods: This prospective observational study was conducted from March 2024 to March 2025 in Masonic Hospital for Children. All Children who were started on oral antibiotics and fulfilled the inclusion criteria were followed up for 10 weeks via phone. WHO criteria and the Bristol stool chart were used.
Results: 1039 children completed the study follow-up. AAD occurred in 55 children (5.3%), with the highest incidence in infants under 1 year (10.9). Most cases were mild and self-limiting. Mean duration was 1.87 days. Onset and duration of AAD, along with antibiotic duration, showed no significant variation across groups. Only One child had severe diarrhoea requiring hospitalization.
Conclusion: AAD is generally mild and often subsides despite continued antibiotic use. Younger age was a risk factor. These findings can guide caregiver counselling to promote antibiotic adherence and unnecessary probiotic usage.
| | | | Keywords | Antibiotic-associated diarrhoea, Aminopenicillins, Cephalosporins, Macrolides, Outpatient, Oral antibiotics.
Abbreviations: AAD - Antibiotic-Associated Diarrhoea.
| | | | Introduction | Antibiotics are essential in treating children with bacterial infections. Antibiotic-associated diarrhoea (AAD) is a known consequence of such treatment.1,2 AAD is defined as the passage of three or more liquid or watery stools per day, or more frequent passage than is normal for the individual, associated with antibiotic exposure, either during treatment or up to eight weeks after antibiotics have been discontinued.3,8
The reported incidence of AAD in paediatric populations varies widely (5-30%), influenced by factors like antibiotic type, patient age, and diagnostic criteria.2,5 While often self-limiting, AAD causes discomfort, parental anxiety, and may necessitate changes in therapy.6 Young children, particularly infants, have been reported to be at higher risk due to their developing gut microbiota, being vulnerable to antibiotic-induced dysbiosis.9,10 This has led to combinations of oral antibiotics with probiotics. But AAD can also result from a decrease in metabolism of carbohydrates and bile acid.2 A Cochrane review found that probiotics are protective against Clostridioides difficile–associated diarrhoea; however, C. difficile causes only a small proportion of AAD cases.7,14
However comprehensive data on various aspects of AAD in children in India is limited.4,5,6 Such data are crucial for formulating clinical guidelines and antibiotic stewardship. This prospective study, conducted at a tertiary care childrens’ hospital aimed to determine the incidence of AAD, identify associated risk factors (age, antibiotic type, duration), and characterize its clinical course and severity in children aged 1 month to 5 years receiving oral antibiotics in an outpatient setting.
| | | | Methods & Materials | This was a prospective observational study conducted at the outpatient department (OPD) of Masonic Hospital for Children from March 2024 to March 2025. A total of 1746 children were initially screened. Exclusion at enrolment (n=603) included children with antibiotic use in the past 2 months, chronic gastrointestinal illness, history of gastrointestinal surgery, long-term drug therapy, immunocompromised status, or gastrointestinal symptoms at presentation. Following this, 1143 children met the inclusion criteria (aged 1 month–5 years). Informed consent was obtained and oral antibiotics were started. The type and duration of antibiotics was decided by the treating paediatrician as per hospital protocol. Caregivers were educated on Bristol Stool Chart (BSC). Follow-up was done via phone calls on days 2, 3, 7 and once in weeks 2, 4, 8, 10. During follow-up,if the child had diarrhoea for 3 or more days,he/she had rotaviral antigen testing and stool culture done and were excluded if positive.Those who received second course of antibiotics during the follow up period were also excluded. 104 were excluded for above reasons. The final study population comprised 1039 children and assessment included onset, duration, and severity of diarrhoea. Probiotics were not prescribed for any patient in our study. Antibiotics were continued in all except in 6 children with diarrhoea.
Statistical Analysis
Data was coded in Microsoft excel, analysed using SPSS 27. Kolmogorov-Smirnov(K-S) test was used to check the normality of the data. Descriptive statistics, including means and standard deviations (SD)for continuous variables and frequency /percentages for categorical variables, where used to summarise participants characteristics and outcomes. Chi-square test (for categorical variables), and t test for (continuous variables) was used to find out the association between those with AAD and without. Analysis of variance (ANOVA) was employed to compare the mean onset and duration of AAD across different antibiotics types. A p-value of <0.05 was considered statistically significant.
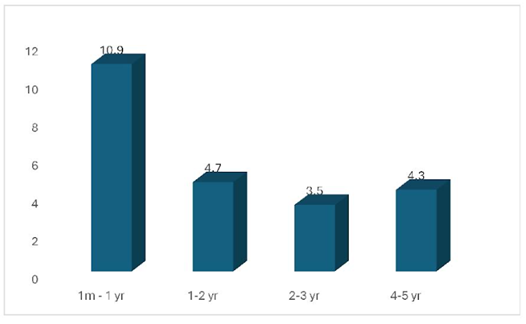
Figure 1. Incidence of AAD in 1 month to 5 years.
 | | | | Results | A total of 1039 participants completed the 10-week follow-up of whom 566 (54.5%) were boys. The overall incidence of oral AAD in our outpatient paediatric population was 5.3% (55 out of 1039 enrolled children). There was no statistically significant difference in AAD between genders.
The incidence of AAD varied by age group: 1 month to 1 year: 10.9% (18/164), 1-2 years: 4.7% (10/211), 2-3 years: 3.5% (7/2 00), 3-5 years: 4.3% (20/464).
Most common indication for antibiotic was acute respiratory tract infection (337/1039;36.2%) followed by other infection like AOM, UTI, skin and soft tissue infections.
Antibiotic classes prescribed were aminopenicillins (Amoxicillin, amoxicillin-clavulanate - 671), macrolides (Azithromycin, erythromycin-266), and cephalosporins (cefixime, cephalexin, cefpodoxime-102). The AAD incidence associated with each major antibiotic type was: Aminopenicillins: 5.3% (36/671), Cephalosporins: 4.9% (5/102), Macrolides: 4.8% (13/266). The mean duration of antibiotic therapy was comparable between children who developed AAD (5. 69 days; SD-1.53 days) and those who did not (5.71days; SD-1.43 days).44 (80%) out of 55, had onset of loose stools within 1 week of starting antibiotics of whom 30(68%) had onset within 2 days,10(22.7%) between 3-5 days and 4(9) between 5-7 days. 5.4% developed diarrhoea in the second week, and 14.5% between third and tenth week. 40 children had loose stools while on antibiotics, and 15 had diarrhoea onset after stopping antibiotics.
The mean duration of AAD was 1.87 days (SD = 1.23 days). Loose stools lasted for 1 day in 29 (52.7%) children and 1-3 days in 21 (38.1%) children. Only 5 cases lasted longer than 3 days (9%) (3 cases for 4 days, 1 case for 5 days, and 1 case for 7 days).
There was no statistically significant difference in the mean onset of AAD across different antibiotic types (F = 0.450, P = 0.811). Similarly, there was no statistically significant difference in the mean duration of AAD across different antibiotic types (F = 1.782, P = 0.134). Antibiotics were discontinued due to AAD in 6 children, of whom only 1 (a 2-year-old toddler on azithromycin) required hospitalization; rotavirus antigen and stool culture in this case were negative. | | | | Discussion | The overall AAD incidence in our study was 5.3% which is at the lower end of the range reported in literature.1,2 Our incidence is lower than 10.4% reported in Turkish and Thai outpatient study population5,6, but aligns with the general understanding of AAD as uncommon adverse event. The significantly higher incidence in infants (1-12 months, 10.9%) strongly reinforces age as a primary risk factor, attributed to their developing and less resilient gut microbiota.4,9,10
In our study, aminopenicillins (5.3%), cephalosporins (4.9%), and macrolides (4.8%) were prescribed. Antibiotics commonly associated with diarrhoea include cephalosporins, lincosamides (especially clindamycin), and aminopenicillins.2,9,11Despite this, the incidence of AAD with above antibiotics in our study was low, and there was no marked difference in AAD among these antibiotic classes. Interestingly, the mean duration of antibiotic therapy did not significantly differ between children with and without AAD (5.71 vs. 5.69 days). This suggests that in short-course outpatient regimens, the type of antibiotic and host factors like age might be more important than treatment duration, potentially indicating a rapid onset of dysbiosis.13 However, other studies done in hospitalized patients have shown that those treated with antibiotics for 3 days had a significantly lower frequency of AAD compared to those treated for longer durations (P = 0.009).11
In our study, AAD was generally mild and self-limiting, with 80% of cases occurring within one week of antibiotic initiation, highlighting the early impact of antibiotics. The mean duration of 1.87 days (SD 1.23) is shorter than some other reports.4 There was no significant difference in the onset or the duration of AAD by antibiotic type (p = 0.811 and p = 0.134, respectively). Antibiotic-associated diarrhoea is commonly classified as mild to moderate and self-limited, but rare cases of severe AAD have been reported.12,13 In our study, while six children required antibiotic discontinuation, only one necessitated hospitalization, indicating that severe outcomes remain uncommon in the outpatient context.
These findings have several clinical implications. They underscore the need for judicious antibiotic prescribing,adequate parental counselling about the rarity of AAD, its typical benign course in the majority, and when to seek medical advice is essential to reduce anxiety. This study also questions the need for combining antibiotics with probiotics when used in children either separately or in combination.
Our study has limitations. As a single-centre study, generalisation to other regions may be limited. Microbiological investigations were performed only in a subset of cases, which might limit our ability to fully exclude all infectious causes. Furthermore, reliance on parental reporting for stool characteristics, while common, introduces some subjectivity.
In conclusion, oral AAD is mild and self-limiting adverse event in outpatient children, with infants being most susceptible. The findings emphasize the importance of judicious antibiotic prescribing, robust parental education to prevent premature withdrawal of antibiotics and also there is no rationale in combining antibiotics with probiotics to prevent AAD.
| | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Turck D, Bernet JP, Marx J, Kempf H, Giard P, Walbaum O, Lacombe A, Rembert F, Toursel F, Bernasconi P, Gottrand F, McFarland LV, Bloch K. Incidence and risk factors of oral antibiotic-associated diarrhea in an outpatient pediatric population. J Pediatr Gastroenterol Nutr. 2003 Jul;37(1):22-6. doi: 10.1097/00005176-200307000-00004. [CrossRef] [PubMed]
- McFarland, L. V. (2008). Antibiotic-associated diarrhoea: epidemiology, trends and treatment. Future Microbiology, 3(5), 563-578. [CrossRef] [PubMed]
- World Health Organization. . Diarrhoeal disease. https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease.
- Högenauer C, Hammer HF, Krejs GJ, Reisinger EC. Mechanisms and management of antibiotic-associated diarrhea. Clin Infect Dis. 1998;27:702-710. doi: 10.1086/514958. [CrossRef] [PubMed]
- Damrongmanee A, Ukarapol N. Incidence of antibiotic-associated diarrhea in a pediatric ambulatory care setting. J Med Assoc Thai. 2007 Mar;90(3):513-7.
- Tanır Basaranoğlu, S., Karaaslan, A., Salı, E. et al. Antibiotic associated diarrhea in outpatient pediatric antibiotic therapy. BMC Pediatr 23, 121 (2023). https://doi.org/10.1186/s12887-023-03939-w [CrossRef] [PubMed] [PMC free article]
- Goldenberg JZ, Yap C, Lytvyn L, et al. Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children. Cochrane Database Syst Rev. 2017;(12):CD006095. [CrossRef] [PubMed]
- Alam S, Mushtaq M. Antibiotic associated diarrhea in children. Indian Pediatr. 2009 Jun;46(6):491-6. PMID: 19556659.
- Owens RC, Donskey CJ, Gaynes RP, et al. Antimicrobial-associated risk factors for Clostridium difficile infection. Clin Infect Dis. 2008;46(suppl 1):S19-S31. [CrossRef] [PubMed]
- Tamburini, S., et al. (2016). The microbiome in early life: implications for health outcomes. Nature Medicine, 22(8), 719-729. [CrossRef] [PubMed]
- Wiström J, Norrby SR, Myhre EB, et al. Frequency of antibiotic-associated diarrhoea in 2462 antibiotic-treated hospitalized patients: a prospective study. J Antimicrob Chemother. 2001;47(1):43-50. [CrossRef] [PubMed]
- Arvola T, Laiho K, Torkkeli S, et al. Prophylactic Lactobacillus GG reduces antibiotic-associated diarrhea in children with respiratory infections: a randomized study. Pediatrics. 1999;104(5):e64. [CrossRef] [PubMed]
- National Clostridium difficile Standards Group: Report to the Department of Health. J Hosp Infect. 2004;56(suppl 1):1-38. [CrossRef]
- Horosheva TV, Vodyanoy V. Sorokulova I. Efficacy of Bacillus probiotics in prevention of antibiotic-associated diarrhoea: a randomized, double-blind, placebo-controlled clinical trial. JMM Case Rep. 2014;1(3):1-6. [CrossRef]
DOI: https://doi.org/10.7199/ped.oncall.2027.6
|
| Cite this article as: | | Madhavan R, Srinivasan M, Matthai J. Incidence, risk factors and severity of antibiotic associated diarrhoea in children treated as outpatient in a tertiary care children’s hospital. Pediatr Oncall J. 2026 Feb 02. doi: 10.7199/ped.oncall.2027.6 |
|