|
Dermatologic Clue to Bacterial Organism
Krunal Shah, Arun Kumar, L Appaji.
Department of Pediatric Oncology, Kidwai Memorial Institute of Oncology, Bangalore, India.
ADDRESS FOR CORRESPONDENCE
Dr Krunal Shah, 817/B, Padmini Darshan, 7th block Jayanagar, Bangalore 560082, Karnataka, INDIA.
Email: krunalms90@gmail.com
pseudomonas, ecthyma gangrenosum, pediatric oncology
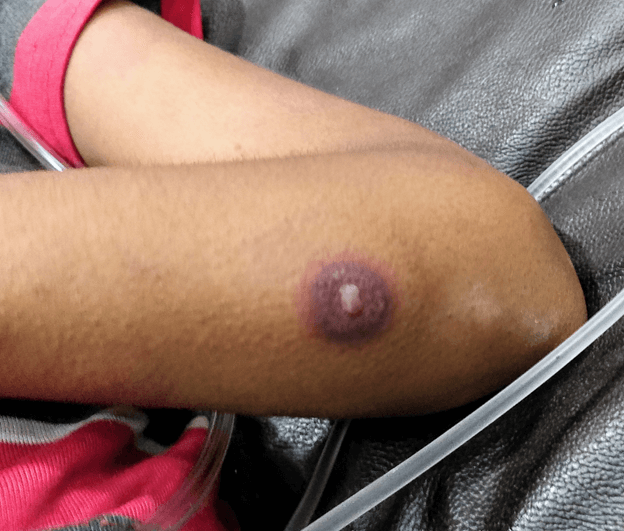
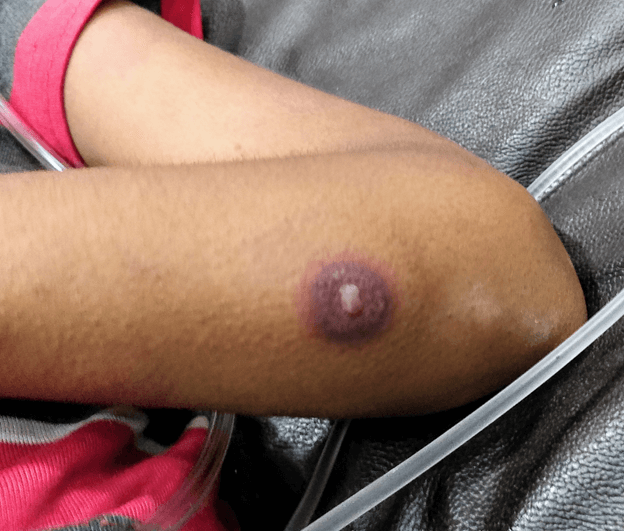
A 12 year old female was receiving chemotherapy for Ewing’s sarcoma. She had received six cycles of vincristine, cyclophosphamide and doxorubicin alternating with ifosfamide and etoposide in the last four months. A week after completion of the seventh cycle, she presented with fever and difficulty in breathing for three days. On initial assessment child had heart rate of 120/min, respiratory rate of 46/min and blood pressure of 80/56 mmHg. Peripheral pulses were feeble and extremities were warm, suggestive of shock. She had multiple maculopapular erythematous rashes on bilateral forearm with a characteristic skin lesion on extensor surface of forearm, just distal to the elbow. It had a central bulla with violaceous indurated halo around it (Figure 1). Respiratory system examination suggested signs of increased work of breathing and bronchial breath sounds in both the lung fields on auscultation. Abdomen was soft without any guarding or tenderness. Child was alert oriented and had no signs of focal neurologic deficit on examination. Complete blood count revealed hemoglobin 8.2 g/dL, total leucocyte count 1200 cells/cumm and platelet count 60,000 cells/cumm. Absolute neutrophil count was 400 cells/cumm. Chest radiograph had bilateral diffuse infiltrates with multifocal air space consolidation. Child was managed as neutropenic sepsis and started on intravenous (IV) piperacillin-tazobactam at 300 mg piperacillin/kg/day in three divided doses and IV vancomycin at 60 mg/kg/day in three divided doses. Blood culture grew pseudomonas aeruginosa 72 hours later. Piperacillin-tazobactam was discontinued and patient was started on IV Colistin (Colistin base activity) at a dose of 3 mg/kg/day in three divided doses since the isolate was only sensitive to a Polymyxin.
Figure 1. Skin lesion showing central bulla with violaceous indurated halo.
|
Figure 1. Skin lesion showing central bulla with violaceous indurated halo
|
What is the skin lesion?
Ecthyma gangrenosum. It is a characteristic skin lesion associated with Pseudomonas aeruginosa bacteremia. The organism invades tunica media and adventitia of the cutaneous blood vessels causing secondary ischemic necrosis.1 The cutaneous lesions rapidly progress from erythematous macule to areas of induration that eventually develop a central necrotic pustule or bulla. These are commonly seen in axillary and anogenital region, but may occur at any anatomic region.2 Skin lesion resembling ecthyma gangrenosum may be seen in septicemia due to various organism like Aeromonas hydrophila, Klebsiella pneumonia, Serratia marcescens, Xanthomonas maltophilia, disseminated nontuberculous mycobacteria and Fusarium solani.3 Although not pathognomonic, ecthyma gangrenosum provides a clue to pseudomonas aeruginosa as the causative organism especially in immunocompromised patients where blood culture may not always be conclusive.4 Early identification of these lesions may aid in deciding appropriate antibiotic regimen with antipseudomonal activity. Cefepime or piperacillin-tazobactam are recommended as first-line agents for critically ill oncology patients.5 Multidrug resistance is defined as non-susceptibility to at least one antibiotic in at least three classes for which P. aeruginosa susceptibility is generally expected: penicillins, cephalosporins, fluoroquinolones, aminoglycosides, and carbapenems.6 “Difficult-to-treat” resistance (DTR) is defined as P. aeruginosa exhibiting non-susceptibility to all of the following: piperacillin-tazobactam, ceftazidime, cefepime, Aztreonam, meropenem, imipenem-cilastin, ciprofloxacin, and levofloxacin.7 The preferred options for the treatment of DTR-P. aeruginosa are ceftolozane-tazobactam, ceftazidime-avibactam, and imipenem-cilastatin-relebactam.6 Colistin or polymyxin B can be considered in combination with the ß-lactam-ß-lactamase inhibitor as a last option if no preferred agent demonstrates activity against DTR-P. aeruginosa.8 The usual duration of antibiotic therapy is 10 to 14 days.9 Surgical debridement may be necessary for ecthyma gangrenosum with extensive skin necrosis, especially those involving peri-anal region.10 |
| |
| Compliance with ethical standards |
|
Funding: None
|
|
|
Conflict of Interest: None
|
|
- Bettens S, Delaere B, Glupczynski Y, Schoevaerdts D, Swine C. Ecthyma gangrenosum in a non-neutropaenic, elderly patient: case report and review of the literature. Acta Clin Belg. 2008;63:394-397. [CrossRef] [PubMed]
- Sarkar S, Patra AK, Mondal M. Ecthyma gangrenosum in the periorbital region in a previously healthy immunocompetent woman without bacteremia. Indian Dermatol Online J. 2016;7:36-39. [CrossRef] [PubMed] [PMC free article]
- Vaiman M, Lazarovitch T, Heller L, Lotan G. Ecthyma gangrenosum and ecthyma-like lesions: review article. Eur J Clin Microbiol Infect Dis. 2015;34:633-639. [CrossRef] [PubMed]
- UpToDate. Pseudomonas aeruginosa skin and soft tissue infections. Available on URL: https://www.uptodate.com/contents/pseudomonas-aeruginosa-skin-and-soft-tissue-infections?source=history_widget. Accessed May 3, 2021.
- Arshad M, Nowalk A, Tamma PD. Multidrug-Resistant Gram-Negative Infections in Transplant and Oncology Patients. Pediatric Transplant and Oncology Infectious Diseases E-Book, 2020: 97. [CrossRef] [PubMed] [PMC free article]
- Tamma PD, Aitken SL, Bonomo RA, Mathers AJ, van Duin D, Clancy CJ. Infectious Diseases Society of America Guidance on the Treatment of Extended-Spectrum β-lactamase Producing Enterobacterales (ESBL-E), Carbapenem-Resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with Difficult-to-Treat Resistance (DTR-P. aeruginosa). Clin Infect Dis. 2021;72:e169-e183. [CrossRef] [PubMed]
- Kadri SS, Adjemian J, Lai YL, Spaulding AB, Ricotta E, Prevots DR, et al. Difficult-to-treat resistance in gram-negative bacteremia at 173 US hospitals: Retrospective cohort analysis of prevalence, predictors, and outcome of resistance to all first-line agents. Clin Infect Dis. 2018;67:1803-1814. [CrossRef] [PubMed] [PMC free article]
- Sabuda DM, Laupland K, Pitout J, Dalton B, Rabin H, Louie T, Conly J. Utilization of colistin for treatment of multidrug-resistant Pseudomonas aeruginosa. Can J Infect Dis Med Microbiol. 2008;19:413-418. [CrossRef] [PubMed] [PMC free article]
- Bassetti M, Vena A, Croxatto A, Righi E, Guery B. How to manage Pseudomonas aeruginosa infections. Drugs Context. 2018;7:212527. [CrossRef] [PubMed] [PMC free article]
- Khalil BA, Baillie CT, Kenny SE, Lamont GL, Turnock RR, Pizer BL, et al. Surgical strategies in the management of ecthyma gangrenosum in paediatric oncology patients. Pediatr Surg Int. 2008;24:793-797. [CrossRef] [PubMed]
|
|
DOI: https://doi.org/10.7199/ped.oncall.2022.16 |
| |
Cite this article as:
Shah K, Kumar A, Appaji L. Dermatologic Clue to Bacterial Organism. Pediatr Oncall J. 2022;19. doi: 10.7199/ped.oncall.2022.16
|