Joana Gomes Vieira, Carolina Oliveira Gonçalves, Marta Silva, Catarina Melancia, Vera Ramos, Clara Abadesso.
Child and Youth Department, Hospital Prof. Doutor Fernando Fonseca, E.P.E., Amadora, Lisbon, Portugal.
ADDRESS FOR CORRESPONDENCE
Joana Gomes Vieira, Alameda Fernão Lopes, nº25 2C. 1495-135 Algés, Lisbon, Portugal.
Email: joana.vieira@hff.min-saude.pt. | | Abstract | Introduction: To this day, pediatric pain in hospitalized children is still underdiagnosed and undertreated. Over the past decade, organizational culture in our pediatric department has been changing to improve pain management practices. We set out to understand how we are managing pain, by describing point prevalence of pain in admitted children, to characterize pain intensity, management and treatment.
Methods: We conducted a cross-sectional survey (adapted from Friedrichsdorf et al.) and included all inpatients in the pediatric department in 15 randomly chosen days over a 9-month period. Patients and families were asked about their experience with pain and its management during admission.
Results: Out of 133 children included in the survey, 120 (90.2%) reported pain at some point during hospitalization and 83.5% (n=111) reported pain in the previous 24 hours. The average worst pain intensity score in the previous 24 hours was 7.6/10 (SD = 1.9). The most frequent cause for pain was acute disease (39.6%, n=44), followed by non-needle related procedures (28.8%, n=32), needle-related procedures (27.9%, n=31) and surgery (3.6%, n=4). Severe pain was reported by 72.8% of patients but registered in only 4.9% of the medical charts. Average score of satisfaction with pain management was 8.6/10 (SD 2.0). The most commonly prescribed analgesics were paracetamol, metamizole and ibuprofen.
Conclusion: Our results revealed procedures to be a major cause of pain, including needle pokes, but also the mandatory nasal swab. Despite the existing hospital policies, there are many aspects to be improved in preventing and managing the patients’ pain.
| | | | Keywords | | Pain, Pediatrics, Surveys and Questionnaires, Quality Improvement. | | | | Introduction | Over the past decades, substantial advances have been made in pediatric pain diagnosis and management, with the development and validation of pain scales adapted to children and improved pharmacologic and non-pharmacologic interventions.1 Nonetheless, to this day, pediatric pain in hospitalized children is still underdiagnosed and undertreated.2,3,4,5,6,7,8
Throughout childhood, painful medical procedures are frequent as is the case with vaccines. Some situations of acute pain, such as acute disease or trauma, might warrant a visit to the emergency room or even hospitalization. In hospitalized children, prevalence of moderate to severe pain varies between 24 to 80%.6,7,8,9,10,11,12 Additionally, diagnostic and therapeutic procedures performed in admitted children are also a source of potentially acute pain that is usually undertreated.2,7
Children living with complex chronic diseases and children undergoing surgical procedures are frequently exposed to painful procedures such as bone marrow aspirations, lumbar punctures, venipunctures and dressing changes.13
In the healthy child, the anxiety associated with vaccines can sometimes lead to immunization non-compliance by both children and caregivers.14 Untreated pain in hospitalized children has both short and long-term consequences.15,16,17,18,19 Exposure to pain in premature infants is associated with increased morbidity (e.g., intraventricular hemorrhage) and mortality15,20 and on the long-term, to higher pain self-ratings during venipuncture by school age,21 poorer cognition and motor function,22 as well as an increased risk for developing problems in adulthood such as chronic pain, anxiety and depressive disorders.23,24,25 Also, inadequate analgesia during procedures in pediatric patients compromises the effect of an adequate analgesia in subsequent procedures.26
In 2004, the World Health Organization (WHO) and the International Association for the Study of Pain issued a joint statement declaring that “The relief of pain should be a human right”.27 Therefore, adequate management of infant and child pain, as advocated by the UN Convention on the Rights of the Child is crucial for the child’s healthy development.23,24,25 It is the duty of any healthcare professional to relief the pain in infants and children.28
Our Pediatric Department is a medical surgical unit formed by an emergency service, a pediatric ward, a pediatric and neonatal intensive care unit (PICU and NICU), an outpatient clinic with several pediatric specialties and a newly formed palliative care team.
Over the past 10 years, our pediatric department has been implementing several strategies to prevent and manage pain, from its evaluation using the appropriate pain scales (the faces pain scale, the numeric pain scale, the Face, Legs, Activity, Cry and Consolability - FLACC behavioral pain assessment scale and Neonatal Infant Pain Scale - NIPS) to its systematic treatment, using pharmacologic and non-pharmacologic interventions, elaborating specific protocols, involving caregivers, raising awareness and training healthcare professionals (HCPs) about adequate pain management.
We set out to understand how we are managing pain, by describing point prevalence of pain in admitted children, characterizing pain intensity and its management.
The purpose of this prospective cross-sectional survey is to evaluate patient outcomes (such as pain prevalence, pain intensity and pain satisfaction) and process outcomes (such as pain assessment documentation and pain management interventions), for all inpatients on a typical day.
| | | | Methodology | We conducted a prospective cross-sectional survey (by adapting a previously validated questionnaire,6 with the authors’ permission) and reviewed the medical records of all inpatients in the pediatric department. After approval of the hospital’s ethics committee, the survey was applied in 15 randomly chosen days, over a 9-month period, in the years of 2020 and 2021.
Survey design
On each survey day, all inpatients were included, except patients admitted to the NICU, as well as all profoundly sedated inpatients, including patients on mechanical ventilation.
The interviewer introduced the study directly to patients and their parents. The latter were asked for their verbal consent to participate, as were children aged 5 years or more. The interviewer administered the questionnaire and recorded answers on an electronic spreadsheet. Patients were interviewed directly, if possible, as long as their clinical status was appropriate and they were judged by the interviewer to have the developmental capacity to understand the questions and provide information about their pain and treatment. When this was not the case, the caregiver was interviewed.
The revised survey included general demographic questions (sex, age, nationality), questions about clinical background relevant for pain management (i.e., if the child had a medical condition that implied chronic pain). The remaining questions focused on the reason for admission to the hospital, the length of time in the hospital and the child’s general experience with pain before and during the current admission (in the past 24 hours). If the child did not experience pain, the survey was discontinued at that point. Respondents were then asked to indicate the cause of the single “worst pain” experience while in the hospital during the previous 24 hours (i.e., even if the child had been in the hospital >24 hours) and were asked to provide further details, to rate the pain associated with it on a 0 to 10 scale (0 = no pain; 10 = worst pain imaginable), to describe what was done to manage the pain (e.g., analgesia and/or nonpharmacologic strategies), to rate their satisfaction with how the pain was treated and to share their general feedback about what was done well and what could be improved.
Additionally, the patient’s clinical charts were reviewed to evaluate pain assessment records, pharmacological and nonpharmacological interventions prescribed and administered and painful procedures done in the former 24 hours.
Analysis
Data was analyzed using IBM SPSS Statistics 25®. Descriptive and inferential analysis was performed (Chi-square test was used for evaluating independence of nominal variables). A p-value of <0.05 was considered statistically significant.
| | | | Results | Patient and parent/caregiver characteristics
A total of 133 children participated in the survey. Infants were the most numerous group (28.6%, n=38). The majority (60.9%, n=81) were boys. The child was the main interviewee in 12.0% of cases. The most common reasons for admission were respiratory infections (13.5%, n=18), followed by gastrointestinal infections (6.8%, n=9), urinary tract infections (6.0%, n=8), vaso-occlusive crisis in patients with sickle cell disease (SCD) (6.0%, n=8) and admissions for elective surgery (6.0%, n=8). See Table 1 for more information.
Table 1. Sample Characteristics (n=133).
| |
% (n) |
Sex
Male |
60.9% (81) |
Age (years)
<1
=1 and <3
=3 and <6
6 and <10
10 and =13
>13
|
28.6% (38)
9.8% (13)
3.5% (18)
10.5% (14)
12.8% (17)
24.8% (33)
|
Respondent
Child
Mother
Father
Mother and Father
Other (grandmother, aunt, sister, legal guardian)
|
12.0% (16)
77.4% (103)
4.5% (6)
2.3% (3)
3.8% (5)
|
Child and/or parents origin
Portugal
Africa
South America
Asia
Europe
|
42.1% (56)
46.6% (62)
9.0% (12)
1.5% (2)
0.8% (1)
|
Time in hospital at survey
>24 hours
|
76.7% (102)
|
Most common reasons for hospital visit
Infections
Vaso-occlusive crisis in sickle cell patients
Elective surgery
Seizures
Fever
Diabetes mellitus complications
Abdominal pain
Splenic sequestration
Retropharyngeal/peritonsillar phlegmon
|
30.1% (40)
6.0% (8)
6.0% (8)
3.8% (5)
3.0% (4)
3.0% (4)
2.3% (3)
2.3% (3)
2.3% (3)
|
Location of care (n=132)
Pediatric ward
Emergency room
Short-stay unit
PICU**
|
90.2% (120)
88.0% (117)
69.9% (93)
19.5% (26)
|
* Respiratory infections (13,5%, n=18), Gastrointestinal infections (6.8%, n=9), Urinary tract infections (6.0%, n=8),Skin and soft tissue infections, (3.8%, n=5).
**PICU: Pediatric Intensive Care Unit
Patient outcomes
Most patients (74.4%, n= 99) did not experience pain routinely before admission. However, 25.6% of patients (n=34) had a previous disease that implied chronic or recurrent pain. Of those, 61.8% (n=21) had SCD, 8.8% (n=3) had chronic urinary tract infections, 5.9% (n=2) had cerebral palsy and 5.9% (n=2) had type 1 diabetes and were on a subcutaneous insulin regimen with multiple daily administrations.
Of the 133 children interviewed, 120 (90.2%) reported pain at some point during hospitalization and 83.5% (n=111) reported pain in the previous 24 hours. On a scale of 0 to 10 (no pain to worst pain possible), average worst pain intensity score in the previous 24 hours was 7.6 (SD = 1.9).
The most frequent cause for pain was acute disease (39.6%, n=44), followed by non-needle related procedures (28.8%, n=32), needle-related procedures (27.9%, n=31) and surgery (3.6%, n=4). More than half (57.1%) of adolescents chose acute disease as their most significant source of pain, versus 33.7% in the non-adolescent group (p=0.029). Further details are provided in Table 2.
Table 2. Pain in the previous 24 hours: causes and intensity (n=111).
| Pain in the last 24 hours, n=111 |
Average intensity (1 to 10 scale) |
SD |
| Acute disease, 39.6% (n=44) |
7.8 |
1.7 |
| Abdominal pain, n=21 |
7.9 |
2.1 |
| Musculoskeletal pain, n=20* |
8.1 |
1.1 |
| Headache, n=2 |
5.5 |
0.5 |
| Other, n=1 |
6.0 |
0.0 |
| Needle-related procedures, 27.9% (n=31) |
7.4 |
1.8 |
| IV line insertion/blood sampling, n=23 |
7.3 |
1.6 |
| Intramuscular injection, n=3 |
6.0 |
0.8 |
| Subcutaneous injection, n=1 |
10.0 |
0.0 |
| Central catheter insertion, n=1 |
9.0 |
0.0 |
| Lumbar puncture, n=1 |
5.0 |
0.0 |
| Other, n=2** |
10.0 |
0.0 |
| Non-needle procedures, 28.8% (n=32) |
7.3 |
2.1 |
| Nasal swab, n=18 |
8.0 |
1.9 |
| Urinary catheter insertion, n=3 |
7.3 |
0.9 |
| Catheter removal, n=2 |
3.5 |
1.5 |
| Physiotherapy, n=2 |
7.0 |
1.0 |
| Other, n=7 *** |
6.9 |
1.9 |
| Surgery, 3.6% (n=4) |
9.3 |
0.8 |
* Including vaso-occlusive crisis in sickle cell disease patients (n=9)
** Peripheral intravenous line infiltration
***Nasogastric tube insertion (n=3), nasal irrigation (n=2), intratimpanic injection (n=1), chest tube insertion (n=1).
When asked about what had been done to minimize the child’s pain, respondents (n=111) pointed out: education and information about a painful procedure/acute illness (71.2%, n=79), caregiver participation (56.8%, n=63), adequately positioning the child (43.2%, n=48), appropriate medication (41.4%, n=46), distraction (39.6%, n=44), encouraging the child to participate (32.4%, n=36), sucrose/pacifier (18.0%, n=20) and comforting measures (swaddling, rocking) (17.1%, n=19). The application of heat/cold to relieve pain was significantly higher in the group of 6 to 10 years (33.3% vs 3.0%, p=0.000). Medication was significantly less used in infants, when compared to older children and adolescents (26.5% vs. 48.1%, p=0.033). Of note, in children aged <1 year, needle and non-needle procedures were reported as the major sources of pain.
When considering the strategies used, by the cause of pain: distraction was less used in acute disease, versus other causes of pain (p=0.002). Application of heat/cold was significantly more used in acute disease (p=0.014). Use of pacifier/sucrose was significantly higher in all procedures, when compared to acute disease and surgery (27.0% vs 6.3, p=0.005). Medication was significantly more used in acute disease and surgery, when compared to all procedures. (69.0% vs 20.6%, p=0.000). Music was exclusively used in procedures (p=0.017).
When the children who reported pain in the previous 24 hours (n=111) were asked to rate satisfaction with pain management on a scale of 0 (not satisfied at all) to 10 (very satisfied), average score of satisfaction was 8.6 (SD 2.0). Of note, 48 respondents (43.2%) reported 10/10 satisfaction, whereas 8 (7.1%) reported 5/10 or less. Low satisfaction about pain management did not correlate with higher pain scores (p=0.183).
The question “what could be done to improve the child’s pain management” prompted eight respondents to share concerns about medication (either the medication and its potential side effects had not been thoroughly explained, or they believed the medication to be insufficient). Four said they expected more empathy and/or patience towards the child.
When asked to provide further details about what was, in their opinion, really well done in managing the child’s pain, respondents (n=21) pointed out several characteristics of the nursing staff, namely the attentiveness given to children and their needs (57.1%, n=12), the carefulness in speaking and performing painful procedures in the child (9.5%, n=2), the calm (9.5%, n=2) and clear communication that was used (9.5%, n=2).
More than half of children and parents (n = 68, 63.0%) who had questions or worries about pain felt that healthcare professionals listened to them, 9.3% (n=10) did not feel heard, 10.2% (n=11) did not recall and 17.6% (n=19) had questions but did not pose them to the hospital staff.
Process outcomes
Almost all patients (132/133) had an evaluation of pain registered in their medical chart (scales used in the department: faces pain scale, numeric pain scale, FLACC, NIPS). Children received an average of 6.0 (SD = 3.4) pain assessments during the previous 24-hour period.
In the chart review, the average worst pain intensity was 1.3/10 (SD = 2.4) for these 111 children included, which is lower than worst pain intensity recall reported by children and parents who completed the survey (see Fig. 1), for which average intensity was 7.5/10 (SD=1.8). The difference between assessed and self-reported pain was higher in the subgroup of surgery (9.3 vs. 2.3) than in the group of acute disease (7.8 vs. 2.0). Since only basal pain is registered in the patient’s chart (and not procedural pain) it was not possible to determine this difference in the two remainder subgroups.
The intensity of pain assessed in the survey (either self- and/or proxy-reported pain) can be further categorized in mild pain (1-3/10, 3.9%, n=4), moderate pain (4-6/10, 23.3%, n=24) and severe pain (7-10/10, 72.8%, n=75). The former category is in flagrant contrast with 4.9% of severe pain documented in medical charts (Figure 1).
Figure 1. Self/proxy-assessment versus nurses’ evaluation of the “worst pain” in the former 24 hours by pain intensity category.
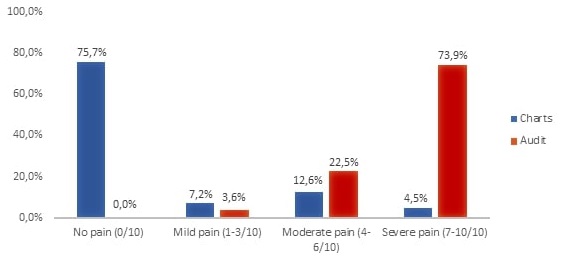
Severe pain (7-10/10) was registered in the chart of five patients. These patients were hospitalized due to: vaso-occlusive crisis in a SCD patient, dehydration and intractable vomiting, myalgia and lower limb weakness, postoperative pilonidal sinus and a case of abdominal pain in a patient with malaria (see Table 3).
Table 3. Characteristics of children with severe pain in the previous, as documented in their medical records.
| Patient |
Age |
Time in hospital (days) |
Reason for admission |
No. of assessments in 24 hours |
Analgesics: fixed-interval (FI) / on-demand (OD) |
Registered nonpharmacologic interventions |
| Prescribed |
Administered |
| 1 |
17 |
7 |
Vaso-occlusive crisis |
5 |
FI and OD |
FI |
None |
| 2 |
8 |
3 |
Dehydration and intractable vomiting |
7 |
OD |
OD, 3 times |
None |
| 3 |
11 |
9 |
Investigation of myalgia and muscle weakness |
3 |
OD |
None |
None |
| 4 |
15 |
1 |
Outpatient sinus pilonidalis surgery |
3 |
FI |
FI |
None |
| 5 |
12 |
4 |
Malaria |
7 |
OD |
OD, 2 times |
None |
From the 111 patients who reported pain in the previous 24 hours, 31 (27.9%) had no analgesia prescribed in their medical chart. In this group of 111 patients, almost half (49.5%, n=55) had painful procedures registered in their charts in that period. There were no records of nonpharmacologic interventions in any of the 133 patient’s charts.
The most commonly prescribed analgesics were paracetamol (n=85, 63.9%), metamizole (n=22, 16.5%), ibuprofen (n=19, 14.3%) and ketorolac (n=9, 6.8%), followed by opioids: morphine (n=9, 6.8%), tramadol (n=5, 3.8%), fentanyl (n=3, 2.3%) (see Table 4).
Table 4. Scheduled and unscheduled pharmacologic pain management interventions.
| |
All children, n=133 |
Acute disease, n=44 |
Needle-related procedures, n=31 |
Non-needle procedures, n=32 |
Surgery, n=4 |
| n |
% |
n |
% |
n |
% |
n |
% |
n |
% |
| Paracetamol / Acetaminophen |
85 |
63.9% |
33 |
75.0% |
18 |
58.1% |
20 |
62.5% |
14 |
100.0% |
| Metamizole |
22 |
16.5% |
11 |
25.0% |
4 |
12.9% |
1 |
3.1% |
4 |
100.0% |
| Ibuprofen |
19 |
14.3% |
6 |
13.6% |
6 |
19.4% |
5 |
15.6% |
1 |
25.0% |
| Ketorolac |
9 |
6.8% |
6 |
13.6% |
1 |
3.2% |
0 |
0.0% |
2 |
50.0% |
| Morphine |
9 |
6.8% |
8 |
18.2% |
0 |
0.0% |
0 |
0.0% |
1 |
25% |
| Tramadol |
5 |
3.8% |
2 |
4.5% |
0 |
0.0% |
1 |
3.1% |
0 |
0.0% |
| Chloral Hydrate |
4 |
3.0% |
1 |
2.3% |
2 |
6.5% |
1 |
3.1% |
0 |
0.0% |
| Fentanyl |
3 |
2.3% |
2 |
4.5% |
1 |
3.2% |
0 |
0.0% |
0 |
0.0% |
| Propofol |
3 |
2.3% |
0 |
0.0% |
2 |
6.5% |
1 |
3.1% |
0 |
0.0% |
| Ketamine |
1 |
0.8% |
0 |
0.0% |
1 |
3.2% |
0 |
0.0% |
0 |
0.0% |
| Naproxen |
1 |
0.8% |
0 |
0.0% |
0 |
0.0% |
0 |
0.0% |
0 |
0.0% |
| Baclofen |
1 |
0.8% |
1 |
2.3% |
0 |
0.0% |
0 |
0.0% |
0 |
0.0% |
| Ropivacaine |
0 |
0.0% |
0 |
0.0% |
0 |
0.0% |
0 |
0.0% |
1 |
100.0% |
Of the 111 children who reported pain in the previous 24 hours, 19 (17.1%) had moderate to severe pain registered in their charts. When analyzing prescribed analgesia, from those 19 patients, 11 (9.9%) did not have an opioid prescription and they were not in compliance with the 2020 WHO guidelines.29 When analyzing the analgesia administration, from this group of 19 patients with moderate to severe pain, 14 (73.7%) were given less than half the on-demand prescribed analgesics.
When considering the four patients who reported surgery as their major source of pain in the previous 24 hours, only two of them had on-demand opioid analgesics prescribed.
| | | | Discussion | Our study confirms the findings of Friedrichsdorf et al.6 in the sense that pain is still common, underrecognized and undertreated in pediatric inpatients. Consistent with these authors’ results, we also found that most pediatric inpatients experienced pain at some point in the previous 24 hours, with the main causes being acute disease and procedures. However, we found a much higher prevalence of severe pain (72.8%) than what has been previously reported3,6 which is around 33 to 48%.
In our audit, pain assessment and documentation were present in almost all inpatients, while Friedrichsdorf et al.6 report this documentation missing for more than one third of inpatients.
In striking contrast with Friedrichsdorf et al.,6 we found the main source of pain in hospitalized children to be acute disease (39.6% vs. 5%), followed by procedures (28.8% vs. 8%). Pain management in acute illness is undoubtedly faulty. This could be due to inadequate use of pain scales, insufficient analgesia and/or inadequate application of our local pain management protocols.
The second main source of pain in hospitalized children was pain associated with non-needle procedures. Since March 2020 a nasal swab to detect SARS-CoV-2 is required in every admitted patient. This has been a major source of pain and discomfort for children and adolescents, with most of children in the procedural pain category reporting this exam as their main source of pain, with an average 8.0/10 intensity. Besides the new mandatory nasal swab, the pandemic also changed the way HCPs interact with patients, with facial masks hindering nonverbal communication and the protective personal equipment being a source of anxiety for children.30 The use of some distraction techniques, like blowing bubbles were discontinued in healthcare settings, as protective measures were deemed the utmost priority. It is yet to be determined how this will impact our ability to perform or reinvent nonpharmacologic interventions to prevent and treat pediatric pain.
In our study, needle-related procedures were reported as being the main cause of pain in 27.9%, which is lower than reported by Friedrichsdorf et. al6 (39%). We hypothesize that the strategies and protocols implemented in our department are responsible for this encouraging result and that this may be a good step towards reducing needle-associated pain and fear.
One quarter of our sample had a previous disease associated with chronic or recurrent pain which is more than twice what Friedrichsdorf et. al6 reported. The majority of our group with a previous painful disease are patients with SCD. Immigrants, mainly from Portuguese-speaking African Countries, compose a major part of the population served by our hospital.
Of the 133 children in our audit, 63.9% were prescribed paracetamol, 37.6% NSAIDs and 12.9% an opioid (see Table 4). The first two were prescribed more often in our audit than in Friedrichsdorf et. al,6 but the latter is half of what these authors reported and lower than expected specially with such a high prevalence of severe pain in the acute disease and surgery groups. In fact, opioid analgesia was not prescribed in almost one-tenth of cases of moderate to severe pain, which is not in compliance with the three-step analgesic ladder implemented by the WHO to manage pain in children.29
When comparing our results to those of Friedrichsdorf et al.,6 we found higher scores of self-reported pain in every subgroup: acute disease (7.8/10 vs. 5.8/10), needle-related procedures (7.4/10 vs. 4.8/10), non-needle related procedures (7.3/10 vs. 6.7/10) and surgery (9.3/10 vs. 7.1/10). Also, as in Friedrichsdorf et al.,6 the worst pain intensity scores reported by children and parents in our audit were much higher, on average, than the average worst pain scores documented in the patients’ chart (see Figure 1). We speculate that pain is being inaccurately assessed, perhaps due to inappropriate use of pain scales, but we acknowledge this could be due to nursing bias or patient/parent recall bias. In any case, institutional protocols should be put in practice to improve training of HCPs in assessing children and adolescent’s pain.
Pain management comprises the rigorous assessment and logging of pain in the patient’s chart. The fact that procedural pain is not registered in the patient’s chart prevents the healthcare team from correctly addressing the issue. Another aspect of note is the absence of records of nonpharmacological interventions in the patient’s charts. These types of interventions are important parts of our medical and nursing practice and, as we can confirm with our results, have a great impact on the patient and on how pain is perceived. As such, as with any intervention, they need to be appropriately registered in the chart.
Several other aspects in this audit are worthy of reflection and improvement. In our study, 9.3% of children and parents who had questions or worries about pain did not feel heard and 17.6% had questions but did not pose them to the HCPs. This should make us reassess how we are communicating with children and parents, since it is the healthcare provider’s responsibility to ensure that the patient understands not only the clinical picture and its treatment, but also to keep the channel of communication open for future questions.
Limitations
Several aspects in our audit limit its generalizability. First, it comprises only one center. Second, other pediatric centers with a smaller investment in pediatric pain protocols, resources and training may not be comparable, especially in the patient and caregiver satisfaction scores. Finally, the fact that the survey took place during the SARS-CoV-2 pandemic may have introduced new biases in our results, specifically when looking at the nonpharmacological interventions for pain which were limited by factors such as the use of Personal Protective Equipment (PPE) and social distancing. In fact, it has been shown that COVID-19 containment measures impact communication and family-centered care in pediatric hospital settings.30
This is to the best of our knowledge the first audit for the prevention and treatment of pain done in a Portuguese pediatric department. In addition to the caregiver’s opinion, we were fortunate enough to survey children aged five or more, which endows our study with a clearer picture of how we are managing our patients.
| | | | Conclusion | | Our results revealed procedures to be a major cause of pain, including needle pokes, but also the mandatory nasal swab for every child admitted to the hospital in this new pandemic era. New studies are needed to ascertain how we can prevent and treat pain in performing such an anxiety-ridden procedure. A very large number of our patients (25%) is suffering from chronic pain and are indubitably in need of improved multidisciplinary ambulatory care. Despite the existing hospital policies, the systematic pain assessment, the use of integrative strategies and parents/patients’ satisfaction, there is still room for improvement in pain management. Continuous training of HCPs in the area of pediatric pain is of the utmost importance as to provide high quality care for children. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Walker SM. Pain in children: recent advances and ongoing challenges. Br J Anaesth. 2008 Jul;101(1):101-10. doi: 10.1093/bja/aen097. [CrossRef] [PubMed]
- Taylor EM, Boyer K, Campbell FA. Pain in hospitalized children: a prospective cross-sectional survey of pain prevalence, intensity, assessment and management in a Canadian pediatric teaching hospital. Pain Res Manag. 2008 Jan-Feb;13(1):25-32. doi: 10.1155/2008/478102. [CrossRef] [PubMed] [PMC free article]
- Stevens BJ, Harrison D, Rashotte J, Yamada J, Abbott LK, Coburn G, et al. Pain assessment and intensity in hospitalized children in Canada. J Pain. 2012 Sep;13(9):857-65. doi: 10.1016/j.jpain.2012.05.010. [CrossRef] [PubMed]
- Twycross A, Collis S. How well is acute pain in children managed? A snapshot in one English hospital. Pain Manag Nurs. 2013 Dec;14(4):e204-e215. doi:10.1016/j.pmn.2012.01.003. [CrossRef] [PubMed]
- Birnie KA, Chambers CT, Fernandez CV, Forgeron PA, Latimer MA, McGrath PJ, et al. Hospitalized children continue to report undertreated and preventable pain. Pain Res Manag. 2014 Jul-Aug;19(4):198-204. doi: 10.1155/2014/614784. [CrossRef] [PubMed] [PMC free article]
- Friedrichsdorf SJ, Postier A, Eull D, Weidner C, Foster L, Gilbert M, et al. Pain Outcomes in a US Children's Hospital: A Prospective Cross-Sectional Survey. Hosp Pediatr. 2015 Jan;5(1):18-26. doi: 10.1542/hpeds.2014-0084. [CrossRef] [PubMed]
- Shomaker K, Dutton S, Mark M. Pain Prevalence and Treatment Patterns in a US Children's Hospital. Hosp Pediatr. 2015 Jul;5(7):363-70. doi: 10.1542/hpeds.2014-0195. [CrossRef] [PubMed]
- Walther-Larsen S, Pedersen MT, Friis SM, Aagaard GB, Rømsing J, Jeppesen EM, et al. Pain prevalence in hospitalized children: a prospective cross-sectional survey in four Danish university hospitals. Acta Anaesthesiol Scand. 2017 Mar;61(3):328-337. doi: 10.1111/aas.12846. [CrossRef] [PubMed]
- Harrison D, Joly C, Chretien C, Cochrane S, Ellis J, Lamontagne C, et al. Pain prevalence in a pediatric hospital: raising awareness during Pain Awareness Week. Pain Res Manag. 2014 Jan-Feb;19(1):e24-30. doi: 10.1155/2014/737692. [CrossRef] [PubMed] [PMC free article]
- Strohbuecker B, Mayer H, Evers GC, Sabatowski R. Pain prevalence in hospitalized patients in a German university teaching hospital. J Pain Symptom Manage. 2005 May;29(5):498-506. doi: 10.1016/j.jpainsymman.2004.08.012. PMID: 15904752. [CrossRef] [PubMed]
- Kozlowski LJ, Kost-Byerly S, Colantuoni E, Thompson CB, Vasquenza KJ, Rothman SK, et al. Pain prevalence, intensity, assessment and management in a hospitalized pediatric population. Pain Manag Nurs. 2014 Mar;15(1):22-35. doi: 10.1016/j.pmn.2012.04.003. [CrossRef] [PubMed]
- Groenewald CB, Rabbitts JA, Schroeder DR, Harrison TE. Prevalence of moderate-severe pain in hospitalized children. Paediatr Anaesth. 2012 Jul;22(7):661-8. doi: 10.1111/j.1460-9592.2012.03807.x. [CrossRef] [PubMed]
- Beal JA. Pediatric Pain Remains Undertreated. MCN Am J Matern Child Nurs. 2021 Sep-Oct 01;46(5):300. doi: 10.1097/NMC.0000000000000745. [CrossRef] [PubMed]
- Taddio A, Ipp M, Thivakaran S, Jamal A, Parikh C, Smart S, Sovran J, Stephens D, Katz J. Survey of the prevalence of immunization non-compliance due to needle fears in children and adults. Vaccine. 2012 Jul 6;30(32):4807-12. doi: 10.1016/j.vaccine.2012.05.011. Epub 2012 May 19. PMID: 22617633. [CrossRef] [PubMed]
- Anand KJ, Barton BA, McIntosh N, Lagercrantz H, Pelausa E, Young TE, et al. Analgesia and sedation in preterm neonates who require ventilatory support: results from the NOPAIN trial. Neonatal Outcome and Prolonged Analgesia in Neonates. Arch Pediatr Adolesc Med. 1999 Apr;153(4):331-8. doi: 10.1001/archpedi.153.4.331. [CrossRef]
- Holsti L, Grunau RE, Oberlander TF, Whitfield MF. Specific Newborn Individualized Developmental Care and Assessment Program movements are associated with acute pain in preterm infants in the neonatal intensive care unit. Pediatrics. 2004 Jul;114(1):65-72. doi: 10.1542/peds.114.1.65. [CrossRef] [PubMed] [PMC free article]
- Doesburg SM, Chau CM, Cheung TPL, Moiseev A, Ribary U, Herdman AT, et al. Neonatal pain-related stress, functional cortical activity and visual-perceptual abilities in school-age children born at extremely low gestational age. Pain. 2013 Oct;154(10):1946-1952. doi: 10.1016/j.pain.2013.04.009. [CrossRef] [PubMed] [PMC free article]
- Ranger M, Chau CM, Garg A, Woodward TS, Beg MF, Bjornson B, et al. Neonatal pain-related stress predicts cortical thickness at age 7 years in children born very preterm. PLoS One. 2013 Oct 18;8(10):e76702. doi: 10.1371/journal.pone.0076702. [CrossRef] [PubMed] [PMC free article]
- Ranger M, Zwicker JG, Chau CM, Park MT, Chakravarthy MM, Poskitt K, et al. Neonatal Pain and Infection Relate to Smaller Cerebellum in Very Preterm Children at School Age. J Pediatr. 2015 Aug;167(2):292-8.e1. doi: 10.1016/j.jpeds.2015.04.055. [CrossRef] [PubMed]
- Taddio A, Katz J, Ilersich AL, Koren G. Effect of neonatal circumcision on pain response during subsequent routine vaccination. Lancet. 1997 Mar 1;349(9052):599-603. doi: 10.1016/S0140-6736(96)10316-0. [CrossRef] [PubMed]
- Valeri BO, Ranger M, Chau CM, Cepeda IL, Synnes A, Linhares MB, et al. Neonatal Invasive Procedures Predict Pain Intensity at School Age in Children Born Very Preterm. Clin J Pain. 2016 Dec;32(12):1086-1093. doi: 10.1097/AJP.0000000000000353. [CrossRef] [PubMed] [PMC free article]
- Grunau RE, Whitfield MF, Petrie-Thomas J, Synnes AR, Cepeda IL, Keidar A, et al. Neonatal pain, parenting stress and interaction, in relation to cognitive and motor development at 8 and 18 months in preterm infants. Pain. 2009 May;143(1-2):138-46. doi: 10.1016/j.pain.2009.02.014. [CrossRef] [PubMed] [PMC free article]
- Brattberg G. Do pain problems in young school children persist into early adulthood? A 13-year follow-up. Eur J Pain. 2004 Jun;8(3):187-99. doi: 10.1016/j.ejpain.2003.08.001. [CrossRef] [PubMed]
- Hestbaek L, Leboeuf-Yde C, Kyvik KO, Manniche C. The course of low back pain from adolescence to adulthood: eight-year follow-up of 9600 twins. Spine (Phila Pa 1976). 2006 Feb 15;31(4):468-72. doi: 10.1097/01.brs.0000199958.04073.d9. [CrossRef] [PubMed]
- Victoria NC, Murphy AZ. Exposure to Early Life Pain: Long Term Consequences and Contributing Mechanisms. Curr Opin Behav Sci. 2016 Feb;7:61-68. doi: 10.1016/j.cobeha.2015.11.015. [CrossRef] [PubMed] [PMC free article]
- Weisman SJ, Bernstein B, Schechter NL. Consequences of inadequate analgesia during painful procedures in children. Arch Pediatr Adolesc Med. 1998 Feb;152(2):147-9. doi: 10.1001/archpedi.152.2.147. [CrossRef] [PubMed]
- World Health Organization supports global effort to relieve chronic pain. Indian J Med Sci. 2004 Oct;58(10):451-2.
- Twycross, A., Dowden, S. & Stinson, J. (2013) Managing Pain in Children: A Clinical Guide for Nurses and Healthcare Professionals. John Wiley & Sons, Ltd. Hoboken, USA.
- World Health Oirganization. WHO Guidelines on the Pharmacological Treatment of Persisting Pain in Children with Medical Illnesses. Geneva: World Health Organization; 2012. Available at: https://www.paho.org/en/node/69972, accessed October 2022.
- Shack AR, Arkush L, Reingold S, Weiser G. Masked paediatricians during the COVID-19 pandemic and communication with children. J Paediatr Child Health. 2020 Sep;56(9):1475-1476. doi: 10.1111/jpc.15087. Epub 2020 Aug 8. [CrossRef] [PubMed] [PMC free article]
DOI: https://doi.org/10.7199/ped.oncall.2024.6
|
| Cite this article as: | | Vieira J G, Gonçalves C O, Silva M, Melancia C, Ramos V, Abadesso C. Point prevalence of pain in a Pediatric Department of a Portuguese level-II hospital: a prospective cross-sectional survey. Pediatr Oncall J. 2024;21: 13-20. doi: 10.7199/ped.oncall.2024.6 |
|