|
Lamellar Ichthyosis
Ashok D Rathod, Rajesh K Kulkarni.
Department of Pediatrics, Grant Medical College and Sir JJ Hospital, Mumbai.
ADDRESS FOR CORRESPONDENCE
Dr Rajesh K Kulkarni, Lecturer in Pediatrics, CWC Building, Grant Medical College and Sir JJ Hospital, JJ Hospital campus, Byculla, Mumbai 400008.
Email: docrajesh75@yahoo.com
|
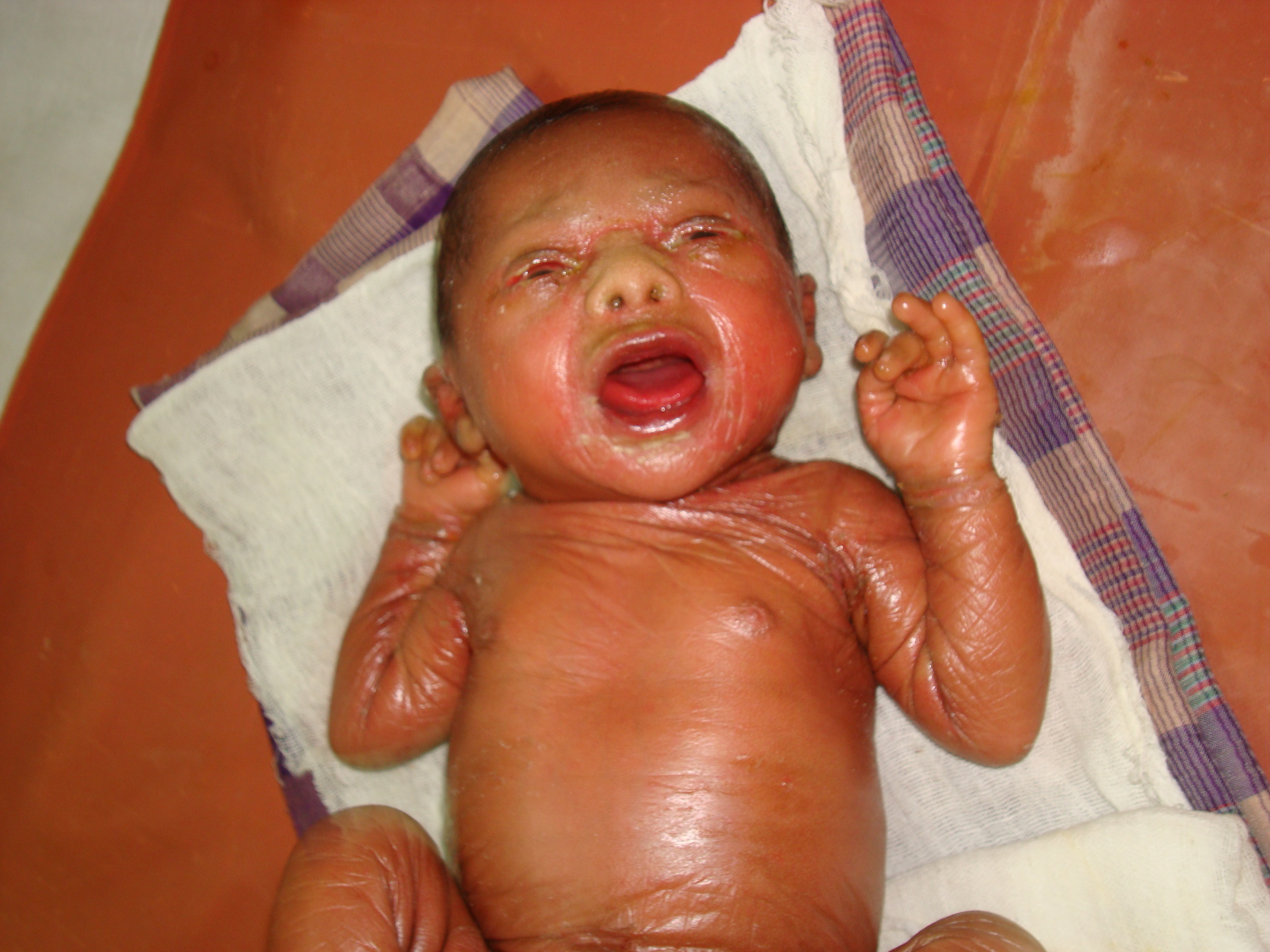
What is the diagnosis?
Lamellar ichthyosis {LI} is an autosomal recessive disorder that is apparent at birth and is present throughout life. Genetic studies have been performed on families with classic lamellar ichthyosis and show markers on band 14q11 in the region of the TGM1 gene locus {1}. The newborn presents encased in a tough, film like membrane resembling oiled parchment that fissures when stretched. This collodion membrane is shed by 10-14 days, revealing generalized erythema and scaling. The lips and mucous membranes tend to be spared. Other associated features are ectropion, eclabium, bilateral conjunctivitis, small and deformed ears, and inflexible digits due to taut skin. Babies with this condition are at high risk for sepsis and hypernatremic dehydration due to the abnormal skin barrier. The aim of treatment in this condition is to avoid the complications like infection and dehydration. Emollients should be applied after showering or bathing. The stratum corneum can absorb 6 times its weight in water, and a heavy emollient, such as petrolatum jelly {Vaseline} or water-in-oil preparations should be applied while the skin is still wet. Alpha-hydroxy acids, such as Lactic acid help reduce corneocyte adhesion and decrease the thickness of the epidermis. Urea creams can help soften scales. Salicylic acids in combination with propylene glycol help to remove dark scaling. Topical retinoic acids decrease thickened scaling. Systemic retinoids are reserved for severe disease that is refractory to conventional therapy. Newer therapies that have resulted in clinical improvement are topical N -acetylcysteine, which has an antiproliferative effect {2} Tazarotene topical 0.05 percent, a receptor-selective retinoid {3} and calcipotriol, a synthetic derivative of vitamin D-3 {4}. Gene therapy serves to be a novel therapeutic approach to lamellar ichthyosis {5}.
This baby did not respond well to Emollients and was started on Isotretinoin orally. With this, the scaling reduced to a great extent and the baby was discharged at 6 weeks of life. The baby is doing well on follow up and has been advised plastic surgery for ectropion.
References
1. Richard G. Molecular genetics of the ichthyoses. Am J Med Genet C Semin Med Genet. 2004, 131C: 32-44
2. Redondo P, Bauzá A. Topical N-acetylcysteine for lamellar ichthyosis. Lancet. 1999, 354: 1880.
3. Stege H, Hofmann B, Ruzicka T, Lehmann P. Topical application of Tazarotene in the treatment of nonerythrodermic lamellar ichthyosis. Arch Dermatol. 1998, 134: 640
4. Kragballe K, Steijlen PM, Ibsen HH, van de Kerkhof PC, Esmann J, Sorensen LH, Axelsen MB. Efficacy, tolerability, and safety of Calcipotriol ointment in disorders of keratinization. Results of a randomized, double-blind, vehicle-controlled, right, left comparative study. Arch Dermatol. 1995, 131: 556-560
5. Akiyama M, Shimizu H. An update on molecular aspects of the non-syndromic ichthyoses. Exp Dermatol. 2008, 17: 373-382.
July 2009 Vol 6 Issue 7 Art # 41 |