SEEMA ALAM, RAJEEV KHANNA, UZMA FIRDAUS.
Pediatric Gastroenterology Section,Department of Pediatrics, JNMC, AMU, Aligarh.
ADDRESS FOR CORRESPONDENCE
Dr Seema Alam, Reader, Department of Pediatrics, JN Medical College, AMU, Aligarh, UP. Email seema_alam@hotmail.com | Diarrhea is one of the most common causes of morbidity and mortality in children all over the world especially in developing countries. The median global incidence of diarrhea was 5 and 2.6 episodes per child per year in infants (6-11months) and for all children between 0-4 years respectively. (1) Much higher rates are seen in children from low socioeconomic status. In India there has been an improvement in the situation from 6 diarrheal episodes per child per year in infants in 1984-85 to 2-3 episodes per child per year a decade later. (1) In 1980 it was estimated that 4.6 million children under 5 years age die annually due to diarrhea. Ten years later, the annual number of deaths attributable to diarrhea was estimated to be 3.3 millions which in 2000 was estimated to be 1.6- 2.5 million. (1) Median global mortality is highest among infants (8.5 per 1000 live births) while for children aged 1-4 years it was 4.6 deaths per 1000 live births. (1) Median global percentage of deaths due to diarrhea in 0-5 years is 21 % in the decade 1990-2000. (1) As evident from the developed countries effective interventions, including correct management (ORT, continued feeding and antibiotic in case of dysentery), promotion of exclusive breast-feeding, better weaning practices, improvement in socioeconomic and literacy status have the potential of reducing the diarrheal diseases. As is evident from Table 1 mortality rates have fallen substantially over the last few decades. In contrast incidence of diarrhea has remained stable, which is a worrying factor as multiple episodes have a harmful effect on physical growth and cognitive functions.
Table 1: Diarrheal estimates in children of developing countries
| Author(s) (Publication Year) | Period | Deaths per Year (millions) | Deaths per 1000 per Year | Episodes per Child per Year | Methods | | Snyder & Merson (1982) 4 | 1954-79 | 4.6 | 13.6 | 2.2 | Long. Cohorts and active surveillance | | Bern et al.(1992) 2 | 1980-90 | 3.3 | 7.6 | 2.6 | Long. Cohorts and active surveillance | | Kosek et al (2003) 1 | 1990-2000 | 2.5 | 4.6 | 3.2 | Data from surveys using WHO methods |
The causes of childhood diarrhea vary with location, season and population. With the advent of AIDS, various enteric pathogens are now being associated with diarrheal diseases. In a report presented to World Health Organization in October 2002 a database was prepared on the proportion of diarrheal illness caused by each diarrheal pathogen, at the community and health facility level for each of the WHO regions and 14 sub-regions. This was done for the under-five children in the period 1990-2000. Table 2 represents the global and SEARO (India , Bangladesh , Myanmar and Nepal ) region data. According to the study globally ETEC was the main cause of diarrhea in the community while rotavirus infection was of major concern at the facility level. On the other hand in the Indian subcontinent mixed infections are one of the major problems at the community level and outpatient level while rotavirus is causing almost one fifth of all cases at the inpatient level.
Table 2: Median percentages of diarrheal illness caused by various Enteropathogens in diarrhea of below five years at the community and facility level (1990 to 2000).
| Agents | COMMUNITY | OUTPATIENT | INPATIENT | | | Global | SEARO D | Global | SEARO D | Global | SEARO D | | Salmonella | 0.9 | 0.6 | 3.2 | 1.4 | 4.4 | 4.2 | | Shigella | 4.6 | 4.4 | 5.8 | 10.4 | 5.6 | 9.2 | | Campylobacter | 7.5 | 8.9 | 12.6 | 16.3 | 4.5 | 3.7 | | V.Cholerae | 0.6 | 2.2 | 1.6 | 3.7 | 4 | 11.7 | | ETEC | 4.1 | 13.3 | 8.7 | 15.3 | 9.5 | 9.7 | | EPEC | 8.8 | 9.1 | 9.1 | 9 | 15.6 | 21.1 | | Rotavirus | 8 | 11.6 | 18 | 17.9 | 25.4 | 20.8 | | Giardia | 10.1 | 5.6 | 3 | 1.9 | 1.6 | 1.6 | Cryptosporidium
| 5.7 | 3.9 | 2.5 | 3.2 | 3.4 | 1.3 | | Entamoeba | 3.2 | 5.1 | 0.6 | 0.3 | 1 | 0.6 | | Coinfection | 10.7 | 18.8 | 12 | 19.9 | 12.1 | 11.7 | | Unknown | 25.4 | 16.6 | 23 | 0.7 | 13.2 | 4.4 |
No investigations are routinely required in acute diarrhea. Majority of the cases need no investigations. In a small proportion the following situations may require investigations.
- Stool culture should be done in cases of bloody diarrhea and cholera only.
- When diarrhea is prolonged beyond 5 days than stool examination for giardiasis and amebiasis is justified. Confirmation is with a fresh stool sample showing trophozoites.
- To confirm a case of secondary lactose intolerance stool pH and reducing sugar can be done.
- Diarrhea associated with clinical signs of electrolyte imbalance or metabolic acidosis may need serum sodium, serum potassium or blood gas analysis.
- Complete blood count, peripheral blood smear, chest radiography, urine culture should be done wherever needed in case of sepsis or if extra-intestinal infection is clinically evident.
Standard Management of Acute Childhood Diarrhea
WHO-ORS, feeding and zinc supplementation remain the standard management of all acute childhood diarrhea. The treatment protocols (Fig 1 & 2) have been developed at our centre as per the hydration and nutrition status of the child. Depending upon the hydration status the children are put under Plans A, B or C. If the child has severe malnutrition (Weight for age <60% and Weight for height <70 %) then intravenous systemic antibiotics are started at admission and the fluid replacement is done slowly under close monitoring to avoid fluid overload Protocol based standardized management of severely malnourished patients with diarrhea decreases the mortality by 47%. (5) For those with moderate malnutrition and children below 4 months (6) close watch should be kept for sepsis. It has been seen that in neonatal diarrhea metabolic acidosis may be the commonest cause for lethargy or refusal to feeds. (7)
FIG 1: Treatment Protocol for management of acute childhood diarrhea.
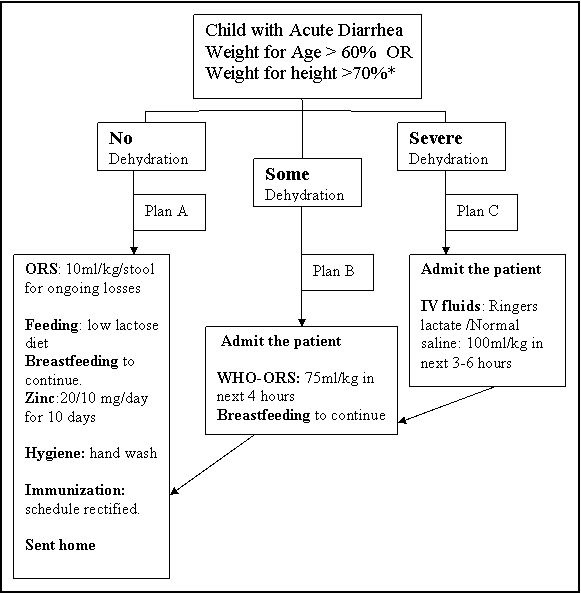
* Antibiotics as per recommendations for those with bloody diarrhea and cholera
FIG 2: Treatment Protocol for management of acute childhood diarrhea in severely malnourished children
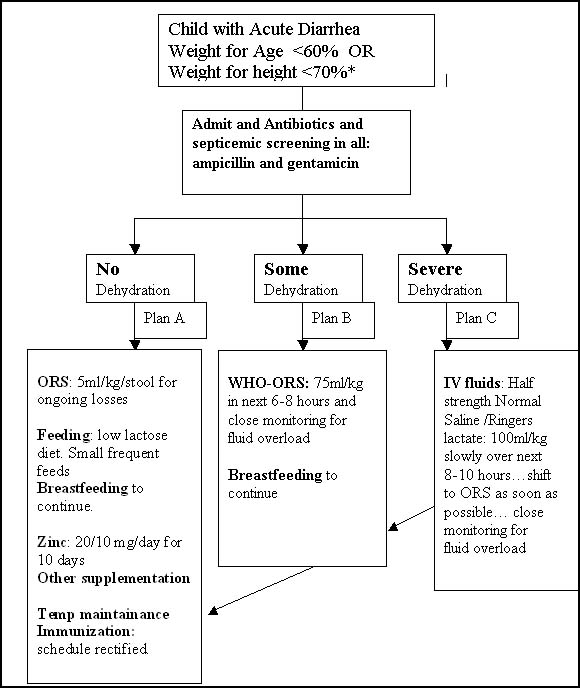
* Antibiotics as per recommendations for those with bloody diarrhea and cholera
Oral Rehydration Solution:
A critical factor in the reduction in diarrheal deaths has been the adoption of oral rehydration solution (ORS) programs for the treatment and prevention of dehydration. (8) Despite very low use rates of ors in large parts of India there has been a decrease in the diarrheal deaths in the past decade. The situation could have been better if the ors use rates were higher. (9) At the same time there is also a very high usage of anti-diarrheal drugs, which could be due to the poor awareness among doctors as well as parental pressure. Improved ors use rates can avert 14 % of the under five mortality. (9) This is would need training of health personnel and education of the masses.
Since the introduction of WHO ors in 1978, there have been attempts to improve the existing formulation by reducing the osmolarity. This was done by reducing glucose and salt concentration in the solution (10-17) or by replacing glucose with a complex carbohydrate or amino acids. (18-20) These solutions generally preserve the 1:1 molar ratio of sodium to glucose that is critical for efficient cotransport of sodium. Studies in animal models and human volunteers have shown that osmolarity of ors may be a critical factor influencing absorption of water and electrolytes from the small intestine. (21) Solutions with osmolarity between 200-250 mmol/L perform better than hypotonic or isotonic solutions. With the exception of rice based ors (20), which significantly reduces stool output in cholera patients, these new ors preparations were not found to be more effective than standard ors and are more expensive. (22) Concerns about hypernatremia and severe glucose malabsorption as well as better water/sodium absorption seen with lower osmolarity in animal models led to the clinical trials of reduced osmolarity ors with reduced sodium & glucose. Studies from all over the world revealed beneficial affects of a reduced osmolarity ors (Total Osmolarity: 245 and 224 mosmls/l) (10,11,13-15) over the standard WHO ors (Total Osmolarity 311 mosmls/l). A large multicenter clinical trial (23) conducted in 675 children with acute non-cholera diarrhea from 5 developing countries revealed that stool output, vomiting and occurrence of hyponatremia were not statistically different between the 2 groups, however the use of unscheduled intravenous fluid following rehydration was reduced in the group receiving reduced osmolarity ors (10% vs. 15%, OR 0.6; 95%CI 0.4-1.0). In a meta-analysis (24) , that evaluated the results of 15 randomized trials, encouraging results about the hypo-osmolar ors was obtained. The use of reduced osmolarity ors was associated with less frequent use of unscheduled intravenous fluid (combined OR, 0.61; 95% CI 0.47-0.81) and less vomiting (combined OR, 0.71; 95% CI, 0.55-0.92). A statistically significant difference in stool output was also noted (standardized mean difference, -0.21; 95% CI -0.31 to -0.12). No significant difference in the incidence of hyponatremia was noted between the 2 groups (Table 3).
Table 3: Meta-analysis of all RCT's comparing reduced osmolarity ors with standard WHO ors in children with acute non-cholera diarrhea
| Outcome | Number of studies | Reduction in odds (95%CI) | | Unscheduled IV | 9 | 39% (19%, 53%) | | Stool output | 12 | 19% (12%, 26%)* | | Vomiting | 6 | 29% (8%, 45%) |
*Reduction in geometric means
Based on the above data, WHO and UNICEF concluded in 2001 (25):
- Reduced osmolarity ors (Table 4) was more effective than standard ors for acute non-cholera diarrhea in children, as measured by clinically important outcomes such as reduced stool output, reduced vomiting, and reduced need for intravenous therapy.
- Among adults with cholera, clinical outcomes were not different although the risk of transient asymptomatic hyponatremia was noted.
- Reduced osmolarity ors was recommended for worldwide use
Table 4: Composition of Reduced Osmolarity ors
| Reduced osmolarity ORS | grams/litre | Reduced osmolarity ORS | mmol/litre | | Sodium chloride | 2.6 | Sodium | 75 | | Glucose, anhydrous | 13.5 | Chloride | 65 | | Potassium chloride | 1.5 | Glucose, anhydrous | 75 | | Trisodium citrate, dihydrate | 2.9 | Potassium
| 20 | | - | - | Citrate | 10 | | - | - | Total Osmolarity | 245 |
Recommendations by IAP National Task Force For The Use Of ors In Diarrhea, August 18-19, 2003 26 and May 5-6, 2006
- All doctors should prescribe ors for all ages in all types of diarrhea.
- The group noted that the new improved universal ors recommended by the WHO is acceptable for all ages and may be made freely available by the Government. However it was proposed that a pediatric ors containing sodium 60 mmol/L, glucose 84 mmol/L, osmolarity 224 mosmol/L is the most suitable solution for children and the industry should be encouraged to produce such a formulation.
- The powder packet to make 1 liter of solution should be continued. Since mothers tend to use ors a glass at a time, a measuring device should be included inside to measure the required amount of powder accurately for 200 ml of fluid.
- The group was deeply concerned that ors was not available free of cost at public institutions. It recommended that measures should be taken by the Government to improve its availability and reduce its cost.
There is a concern for the risk of hyponatremia especially among cholera patients with new reduced osmolarity formulation. No significant difference in the incidence of hyponatremia was found in studies comparing hypoosmolar ors with WHO ors. (15) It is likely that adults with cholera may have asymptomatic hyponatremia with the hypoosmolar solution. In a recent trial among acute diarrhea patients aged 12-60 years no effect of the type of ors was seen on mean serum sodium and relative risk of hyponatremia 24 hours after randomization. (27)
Feeding during Diarrhea:
The relationship between infection and malnutrition is bidirectional. Infection adversely affects nutritional status through reductions in dietary intake and intestinal absorption as well as increased catabolism. On the other hand, malnutrition predisposes to infection because of its negative impact on the barrier protection of the mucous membranes and host immune function.
The Lancet approach indicates that 57% of the annual deaths among under-fives can be prevented through achievement of high coverage of basic public health and nutrition interventions. (9) in the developing world the risk for death during diarrhea in children with mild, moderate and severe malnutrition is 2.32, 5.39 and 12.50 respectively. (28) In India the prevalence of exclusive breastfeeding is about 22 to 81 % across the various states. (9) Data from Delhi reveals that exclusive breastfeeding is 21.1 % at 6 weeks; this falls to about 3-4 % at the 22-weeks. (29) Non-breastfed infants had a higher risk of dying when compared with those who had been predominantly breastfed (HR = 10.5; 95% CI = 5.0-22.0; p < 0.001) as did partially breastfed infants (HR = 2.46; 95% CI = 1.44-4.18; p = 0.001). (29) In a study to see the effect of community based promotion of exclusive breastfeeding, diarrheal illnesses were significantly less in the intervention group. (30) Fully weaned Guatemalan children reduced their energy intake by 30% during acute infections, whereas Bangladeshi children who were still breastfeeding reduced their intakes by only about 7%, suggesting that breastfeeding may protect against diarrhea-induced reductions in intake. It has been observed diarrhea-induced growth deficit (which could be as much as one third of the total growth deficit) was absent in fully breastfed infants in an urban field site in West Africa, and concluded that exclusive breastfeeding prevents the adverse nutritional consequences of diarrhea. (33) Colombian children in these studies who lived in control villages displayed the expected negative relationship between diarrheal prevalence and height at 3 y of age, while there was no effect of diarrhea on the height of those children who lived in villages where food supplements were being distributed. (34) Investigators also described associations between anthropometric indicators of nutritional status and the duration of illness, (35) the severity of fecal purging (36) and most important, the case-fatality rates. (37) In each case, preexisting malnutrition was associated with an increased severity of diarrheal disease.
A meta-analysis (38) that was conducted to re-examine the issue of lactose free diet in diarrhea indicated that the rates of treatment failure were nearly twofold greater (22 vs. 12%) in the groups that received lactose-containing milk feeding. However, the excess rate of treatment failure was confined to those studies that enrolled children with initial severe dehydration. Among the studies that enrolled children with mild or no dehydration, there was no difference in the treatment failure rates. Hence it is safe to manage majority of children by using lactose-containing milk, especially if they have no dehydration. Dehydrated children may benefit from reduced lactose intake (5 gm of lactose per 150 kcal) and close supervision during the early phase of therapy. In a study done at our centre among children less than 4 months with persistent diarrhea, 71% of the infants were receiving lactose free formulae at admission. (39) Almost all the children improved on replacement of the lactose free formulae with low lactose milk cereal diet. Low lactose milk cereal diet provides stimulus for earlier regeneration of mucosal lactase, which is not possible with a lactose free diet. Moreover, the commercially prepared lactose free preparations have high antigenic value which in turn may result in hypersensitivity reaction in the intestinal mucosa and cause prolongation of diarrhea. A proportional hazards regression model controlling for age, diarrheal etiology, and severity of dehydration on admission revealed that the frequently fed group (on cow's milk) had a significantly shorter duration of diarrhea, greater weight gain and lower fecal frequency as well as fecal weight. (40) This could be due to the presence of decreased lactose load in the frequent smaller feeds, which can be achieved in mixed diets based on staple foods. As reviewed previously (41), a number of studies examined the use of mixed diets based on staple foods, and others assessed the effects of individual food components, such as dietary fiber and micronutrients, on the outcome of diarrhea. In general, children fared at least as well with mixed diets as they did with more highly processed formulas, and dietary fiber was found to reduce the duration of the period of liquid stool excretion. (42)
Exclusive breastfeeding in those less than 6 months should be continued. In those top fed, relactation should be encouraged especially if the child is less than 4 months of age. (39) Bottle-feeding should be discouraged. Milk cereal diets fed with katori help in rectifying the faulty feeding habits. Low lactose milk cereal diet frequently fed to the child has better palatability and helps in early recovery from diarrhea. Low lactose milk cereal diets and lactose free diets are easy to prepare, economical and better acceptable (Table 5). Only if indicated then the child should be shifted from to lactose free diet (Table 6). Encourage the child to take 100 kcal / kg and tube feeds can be considered if severe anorexia is present. Once the child recovers from diarrhea on lactose free diet the milk should be introduced gradually over 2-3 weeks on follow up visits. Intensive nutrition education significantly improves the status of malnourished children with or without supplementary feeding. So if effective feeding has been instituted and instructed to mother during diarrhea, it not only has positive effect on the severity of diarrhea but also improves long- term nutrition of the child.
Table 5: Low Lactose and Lactose Free Diets: Ingredients, Caloric content and Feeding Schedule
|
|
Ingredients and
amounts
|
Calories/gm
|
Feeding schedule
|
|
Low
lactose milk cereal diet*
|
Milk
= 50 ml
**Puffed
rice powder= 2 tsf
Sugar
= 1 tsf
Oil
= 1 tsf
|
0.8
|
<
1 year : one feed every 2-3 hrly
1-5
year : Two such feed every 3-4 hrly
|
|
Lactose
free diet*
|
Egg
white = 2 tsf
**Puffed
rice powder = 2 tsf
Sugar
= 1 tsf
Oil
= 1 tsf
Water
= 50 ml
|
0.8
|
<
1 year : one feed every 2-3 hrly
1-5
year : Two such feed every 3-4 hrly
|
* for children < 6 months puffed rice powder (murmura) can be halved and oil can be increased to 1.5 tsf.
** precooked semolina (suji) or precooked porridge (dalia) or banana can also replace the puffed rice powder. In case of vegetarians, double the amount of semolina or porridge can replace puffed rice powder and egg white.
Table 6: Indications for shift from Low Lactose to Lactose Free Diet
- To confirm Purge rate of more than 15 loose stools per day after 48 hours.
- Persistence of dehydration after 48 hours of admission.
- Loss of weight or no gain of weight despite adequate calories for two consecutive days.
Zinc Supplementation in Diarrhea:
Zinc deficiency is present in 30-50 % children living in developing countries. (43) The high prevalence of zinc deficiency in these countries can be explained by increased prevalence of malnutrition, low dietary intake of complementary animal source foods (rich in zinc & with higher bioavailability), insufficiency of the breast milk to fulfill the requirements after 6 months of age, higher consumption of cereals and legumes containing phytates which inhibit zinc absorption and low content of zinc in soil and crops. (44-46) Also, such children suffer from frequent diarrheal illnesses resulting in excessive fecal losses of zinc. (47) Zinc deficiency, on the other hand, predisposes to frequent diarrheal episodes, increasing their duration and severity, creating a vicious cycle. Zinc deficiency reduces brush border enzymes, disrupts intestinal mucosa and increases mucosal permeability and intestinal secretion, thus making the diarrheal episode more severe and prolonged. Zinc supplementation has been known to cause early regeneration of intestinal mucosa thus improving intestinal permeability, restoration of intestinal brush border enzymatic function, overall causing reduction in intestinal secretion and regulation of water and electrolyte transport. Zinc by maintaining the integrity of the gut mucosa reduces and prevents the fluid losses. These responses begin to occur within 48 hours of starting zinc supplementation. (48,49) As is evident from the Table 7, zinc supplementation reduces the incidence (11-15%) (50-52) and prevalence (18-30%) (50,52) of diarrhea in children less than 5 years. Pooled analysis (53) (Zinc Investigators' Collaborative Group taken 3 randomized controlled trials (54-56) on zinc in acute diarrhea from Indonesia, India and Bangladesh) revealed 15 % faster recovery, 24 % reduction in episodes lasting more than 7 days and 16% reduction in the mean duration of diarrhea. There was more variability (9-30%) in reduction of frequency of stools (50,54,57) or stool output (55,58,59) in the various studies. The pooled analysis (53) and the study by Bhatnagar et al (59) have found no differences in effects in the subgroups based on age, sex, zinc levels and nutrition, however more effect was seen in the pooled analysis in those with lower zinc levels. Bhandari et al (50) have found more significant effect in subjects more than 12 months of age, with serum zinc levels more than 60 µg/dL and without stunting or wasting. Sazawal et al (54) have found an increasingly significant effect of zinc in stunted and wasted children and those with serum zinc levels < 60 µg /dL, but without any association with gender. Overall, there doesn't seem to be ample evidence to suggest a specific target group whom zinc should be given during episodes of diarrhea. There were also slightly more chances of vomiting in the zinc supplemented group. (50,57,59) There was an attempt to add zinc in the ors but the beneficial effect was not seen in the supplemented group. (60) This was possibly due to the variable amount of zinc received depending upon the ors intake. The IAP National Task Force in 2003 (26) and 2006 has recommended zinc supplementation for the duration of diarrhea and for 7 days after cessation of diarrhea in all children older than 3 months of age with diarrhea. The dosage is 10 mg/day for infants and 20/day mg for older children. Preferably only zinc preparation should be used. It is recently been reported that zinc supplementation may not be helpful in young infants with diarrhea. (61) We need more studies in children below 3 months to decide the issue. It was seen in recently published study that in the management of acute watery diarrhea, zinc plus ors along with culturally appropriate messages in local language does not affect overall ors use generally and decreases antibiotic/antidiarrheal use with children having good adherence without side effects. (62)
Table 7: Results of Randomized Controlled Trials in children with Acute Diarrhea comparing effect of Zinc with that of Placebo
| ffect studied | Trial | No. of subjects
(Zinc/Placebo treated) | Inference | Effect size
(95% CI) | | Effect on incidence | Bhandari et al 50 | 1093 / 1133 | 12% reduction | 0.88 (0.82-0.95) | | Baqui et al 51 | 3974 / 4096 | 15% reduction | 0.85 (0.76-0.96) | | Rahman et al 52 | 345 / 161 | 11% reduction | 0.89 (0.79-0.99) | | Effect on prevalence | Bhandari et al 50 | 1093 / 1133 | 30% reduction | -1.30 (-0.61 to -2.0) | | Rahman et al 52 | 345 / 161 | 18% reduction | 0.82 (0.78-0.87) | | Effect on recovery | Pooled analysis53 | 1252 / 1194 | 15% faster recovery | 0.85 (0.76 to 0.95) | | Strand et al 57 | 442 / 449 | 26% faster recovery | 1.26 (1.08-1.46) | | Bahl et al60 | 404 / 401 | 11% faster recovery | 0.89 (0.80-0.99) | | Bhatnagar et al 59 | 132 / 134 | 24% faster recovery | 0.76 (0.59 to 0.97) | | Effect on episodes lasting > 7 days | Pooled analysis53 | 1252 / 1194 | 24% reduction | 0.76 (0.63-0.91) | | Bhandari et al 50 | 1093 / 1133 | 21% reduction | 0.79 (0.65-0.95) | | Sazawal et al 54 | 462 / 485 | 39% reduction | 0.61 (0.30-0.94)* | | Strand et al 57 | 442 / 449 | 43% reduction | 0.57 (0.38-0.86) | | Bahl et al60 | 404 / 401 | 39% reduction | 0.61 (0.33-1.12) | | Bhatnagar et al 59 | 132 / 134 | 9% reduction | 0.09 (0.01-0.73) | | Effect on mean duration | Pooled analysis53 | 1252 / 1194 | 16% reduction | 0.16 (0.07-0.26) | | Baqui et al 51 | 3974 / 4096 | 24% reduction | 0.76 (0.65-0.90) | | Bhatnagar et a 50 l | 132/134 | 9 hrs earlier (24% less) | 0.76 (0.59- 0.97) | | Effect on frequency | Strand et al 57 | 442 / 449 | 9% reduction | 0.91 (0.85-0.97) | | Sazawal et al 54 | 456 / 481 | 21% reduction | 0.79 (0.30-0.94) | | Bhandari et al 50 | 1093 / 1133 | 23% reduction | 0.77 (0.63-0.94) | | Effect on Stool output | Roy et al55 | 57/54 | 91 g less | -91 g | | Dutta et al 58 | 44/36 | 900 g less | -900 g (-1200 to -590) | | Bhatnagar et al 59 | 132/134 | 31% less | 0.69 (0.48-0.99) | | Adverse Effect vomiting / regurgitation | Bhandari et al 50 | 1093 / 1133 | 1.7 days more | 1.7 (1.3-2.1) days | | Strand et al 57 | 442 / 449 | 14% more | 1.7 (1.4-2.2) | | Bhatnagar et al 59 | 132/134 | 3% more | 1.1 (0.69-1.9) |
* subgroup analysis in children who were enrolled in the study within 4 days of onset of diarrhea.
Antimicrobials and Antisecretory drugs
Bacillary dysentery or Shigellosis: Various studies on shigellosis done recently are available in the tabulated form (Table 8). High rates of resistance to various antibiotics are present, hence one of the fluoroquinolones (ciprofloxacin /norfloxacin/ ofloxacin) should be the first line of treatment for Shigellosis. If there is no improvement (disappearance of blood), on the fluoroquinolone, within 48 hours then depending upon the general condition the patient should be shifted to a oral (Cefixime) or intravenous (ceftriaxone) cephalosporin. Drug dosage and schedule should be as mentioned in table 9. Based on its efficacy, safety and reduced cost, ciprofloxacin has been recommended by WHO as the first line Antibiotic for shigellosis. (76)
Table 8: Percentage resistance to antimicrobials in the shigella isolates (2001-2005) from the Indian subcontinent.
| Ref | Place | Co
(%) | NA
(%) | Cx&/Nx
(%) | Cef
(%) | serogroup | Isolates
(n) | MDR
(%) | | 63 | Siliguri | 100 | 100 | NT | NT | S. dysenteriae | 30 | | | 64 | Diamond Harbour | 100 | 100 | 100 | NT | S. dysenteriae | 4 | 100 | | 65 | Kolkata | 97 | 97 | 97 | NT | S. dysenteriae | 36 | 97 | | 66 | Kolkata | 100 | 100 | 100 | NT | S. dysenteriae | 152 | 100 | | 67 | Kolkata | 100 | 100 | 75 | NT | S. dysenteriae | 17 | 100 | | 68 | Kolkata | 95 | 59 | 6 | NT | S. flexneri | 68 | 90 | | 69 | Aizawl | 100 | 100 | 100 | 0 | S. dysenteriae | 169 | 100 | | 70 | Dhaka | 98 | 98 | 0 | NT | S. dysenteriae | 122 | 98 | | 71 | Dhaka | 55 | 9 | 0 | NT | S. flexneri | 193 | 40 | | 72 | Karachi | 88 | 39 | 0 | 0 | S. flexneri | 193 | 40 | | 73 | Lahore | NT | 6 | 0 | 0 | S. dysenteriae | 77 | 70 | | 74 | Chandigarh | 58 | 63 | 12 | 14 | S. flexneri | 55 | 43 | | 75 | Vellore | NT | 94 | NT | NT | S. sonnei | | | | | 99* | 97* | 38.5* | 0* | | | 97** |
Co=Cotrimoxazole, NA=Nalidixic acid, Cx&/Nx=Ciprofloxacin&/Norfloxacin, Cef=Cefixime/Ceftriaxone
NT=Not tested
MDR= Multidrug resistance
* Median percentage resistance to the antimicrobials
** Median percentage Multidrug resistance.
Table 9: Drugs dosage and schedules for the antibiotics against Shigellosis.
| Drug | Dosage (mg/kg/day) | Divided dose | Duration (days) | | Ciprofloxacin ( PO ) | 20 | 2 | 5 | | Norfloxacin ( PO ) | 20 | 2 | 5 | | Ofloxacin ( PO ) | 15 | 2 | 5 | | Ceftriaxone (IV) | 100 | 2 | 5 | | Cefixime ( PO ) | 8 | 2 | 5 |
PO = Per oral, IV= intravenous
Cholera:
Resistance to cotrimoxazole (77,78), ampicillin (79) and furazolidone (77,78) , in Vibrio cholerae isolates, has uniformly been described. tetracycline [500mg qid for 3 days] and doxycycline [300mg single dose] are the drugs of choice in treatment of cholera in adults (http://www.who.int/cholera). In children the effective single dose therapy has not been identified. It is well known that the adverse effects like enamel and skeletal defects associated with tetracyclines occur on prolonged use. High degree of sensitivity of Vibrio cholerae to fluoroquinolones and cephalosporins has been reported. (80,81) The high cost and multiple dose schedules of these drugs are the limitations to their usage. Successful treatment in 60% cases, but with high relapse rate, with single dose ciprofloxacin [20mg/kg] was seen in a recent study. (82) Single dose of azithromycin [20mg/kg max: 1gm] as an alternative treatment has also been reported. The high cost of the drug is the major disadvantage. (83)
Probiotics: The mechanisms by which the probiotics helps in diarrhea are still not very well understood. The competitive blockage of receptors thereby preventing the adhesion & invasion of the virus, enhanced immune response, down regulation of the host's secretory & motility defenses and /or inactivation of the virus particles by substances produced by the lactobacilli could be the possible mechanisms. A meta-analysis (84) published in 2006 concluded that there is insufficient evidence about the prophylactic or therapeutic role of probiotics in acute childhood diarrhea in the developing world where the main burden of the disease lies. More recently the LGG was not found to be effective in severe dehydrating diarrhea in at least 2 studies done in the developing world. (85,86) In a recently published study from our centre we did not see any significant beneficial effect of the tyndallized lactobacillus acidophilus on acute childhood diarrhea. (87) At least three meta-analyses have earlier concluded that probiotics could shorten duration of acute diarrhea, especially due to rotavirus in children. (88-90) Among the lactobacilli, the live strain lactobacillus rhamnosus GG was found most effective. Almost all of the studies included in the meta-analysis were from the developed world except for a study from Pakistan where earlier recovery from acute non-bloody diarrhea was seen with lactobacillus rhamnosus GGbut the study included a very small sample size. (91) Most of the RCT's to evaluate the role of lactobacillus rhamnosus GG in acute childhood diarrhea has been done in rotaviral predominant diarrhea. The efficacy of the lactobacilli needs to be studied in the developing world, where the etiologic profile is different.
Racecadotril: Another antisecretory drug, and opiate receptor agonist, called Racecadotril is much in the news nowadays. The enkephalins, endogenous opiate substances, act as neurotransmitters of digestive tract where they cause antisecretory activity without affecting intestinal transit time or motility. Enkephalins after release are rapidly inactivated by enzyme enkephalinase. Racecadotril, an enkephalinase inhibitor, reinforces the physiological activity of endogenous enkephalins. The antisecretory mechanism involves activation of d opiate receptor leading to reduction in secretion of intracellular CAMP. Two RCTs (both funded by the manufacturer's of the drug) have studied the efficacy of Racecadotril in acute childhood diarrhea. In 135 Peruvian boys (3-35 months), 46% reduction in 48 hours stool output was seen in the Racecadotril group as compared to the placebo group. (92) Similar results have been reported by Cezard et al (93) in 172 boys and girls less than 4 years of age. Addition of the girls in the one of the study group makes the primary outcome questionable because urine separation from stool output would be difficult in girls. Moreover 9 cases from the racecadotril and 4 from placebo group were withdrawn during the study. Five cases from the racecadotril and 1 from the placebo group were withdrawn because of adverse events like vomiting or dehydration. In adults with severe cholera comparable total stool output, total ors intake or duration of diarrhea between the racecadotril and loperamide groups has been recently reported. (94) The results of this trial further strengthen the role of standard management of cholera with ors, feeding and doxycycline. A recent RCT (95) comparing racecadotril and loperamide for stopping acute diarrhea in adults found comparable clinical success rates and mean duration of diarrhea. More patients on loperamide had reactive constipation and itching was notably higher in the racecadotril group. Well designed RCTs with adequate sample size and independent of any competitive interest, studying the efficacy and safety of racecadotril in acute childhood diarrhea, are needed before we reach any conclusion regarding the role of the drug in the management of acute diarrhea. nitazoxanide(2-acetyloxy-N-benzamide), is a synthetic oral antiprotozoal agent which has been approved by Drug Controller General of India (100mg in 1-3 years and 200mg in 4-11 years: bd. for 3 days respectively) for treatment of diarrhea caused by Cryptosporidium parvum and Giardia lamblia ( http://cdsco.nic.in/DRUGSAPRVD.htm ). The efficacy of single treatment with nitazoxanide is 100% against cryptosporidial diarrhea (98-100) but even third treatment with nitazoxanide could achieve only 80% eradication rate of Giardia lamblia. (99) In a double blind randomized controlled trial nitazoxanide had equivalent efficacy to metronidazole in children with giardiasis. (101) Moreover nitazoxanide did not show improvement in the resolution of diarrhea, eradication rate and mortality among HIV- seropositive children with cryptosporidial diarrhea. (100) Therefore relatively costly nitazoxanide has an established role against cryptosporidial diarrhea in immuno-competent children but no added benefits are seen over metronidazole in Giardia lamblia infection in children.
Conclusion: It is important to follow the recommendations in the management of acute diarrhea strictly, since indiscriminate usage of drugs results in development of resistance. Addition of so called harmless but unnecessary drugs shifts the emphasis from the standard management (oral rehydration solution, feeding and zinc supplementation) of acute childhood diarrhea. Not to forget that such additions also bring unnecessary financial burden on the community. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Kosek M, Bern C, Guerrant RL. The global burden of diarrhoeal disease, as estimated from studies published between 1992 and 2000. Bulletin of the World Health Organization 2003, 81(3): 197-204. [PubMed] [PMC free article]
- Bern C, Martines J, de Zoysa, I, Glass RI. The magnitude of the global problem of diarrhoeal disease: a ten-year update. Bulletin of the World Health Organization 1992; 70:705-14. [PubMed] [PMC free article]
- Snyder JD, Merson MH. The magnitude of the global problem of acute diarrhoeal disease: a review of active surveillance data. Bulletin of the World Health Organization, 1982, 60: 605-613. [PubMed] [PMC free article]
- Lanata CF , Black R E. Improving Diarrheal Estimates. Presented to: Monitoring and Evaluation Team Child and Adolescent Health and Development World Health Organization. Geneva , Switzerland. October 2002.
- Ahmed T, Begum B, Badiuzzaman, Ali M, Fuchs G. Management of severe malnutrition and diarrhea. Indian J Pediatr 2001; 68: 45-51. [CrossRef] [PubMed]
- Haider R, Kabir I, Fuchs GJ, Habte D. Neonatal diarrhea in diarrhea treatment centre in Bangladesh: Clinical presentation, breastfeeding management and outcome. Indian Pediatrics 2000;37: 37-43. [PubMed]
- Victora CG, Bryce J, Fontaine O, Monasch R. Reducing deaths from diarrhea through oral rehydration therapy. Bull World Health Organ. 2000;78: 1246-1255. [PubMed] [PMC free article]
- Jones G, Schultink W , Babille M Child Survival in India. Indian J Pediatr, June 2006; Volume 73: 479-87. [CrossRef] [PubMed]
- Rautanen T, El-Radhi S, Vesikari T. Clinical experience with a hypotonic oral rehydration solution in acute diarrhoea. Acta Paediatrica , 1993; 82 :52-4. [CrossRef] [PubMed]
- El-Mougi M, El-Akkad N, Hendawi A, Hassan M, Amer A, Fontaine O, Pierce NF Is a low osmolarity ORS solution more efficacious than standard WHO ORS solution? J Pediatr Gastroenterol Nutr , 1994; 19 :83-86 ]. [CrossRef] [PubMed]
- Duggan C, Fontaine O, Pierce NF, Glass RI, MahalanabisD, Alam NH et al. Scientific rationale for a change in the composition of oral rehydration solution. JAMA, 2004; 291: 2628-31. [CrossRef] [PubMed]
- International Study Group on Reduced-Osmolarity ORS Solutions. Multicentre evaluation of reduced osmolarity oral rehydration salts solution. Lancet 1995; 345:282-5. [CrossRef]
- Santosham M, Fayad I, Zikri MA, Hussein A, AmponsahA, Duggan C et al. A double-blind clinical trial comparing World Health Organization oral rehydration solution with a reduced osmolarity solution containing equal amounts of sodium and glucose. J Pediatr 1996;128:45-52 [CrossRef]
- Alam S, Afzal K, Maheshwari M, Shukla I. Controlled trial of hypo-osmolar versus World Health Organization oral rehydration solution. Indian Pediatr 2000; 37: 952-959. [PubMed]
- Faruque ASG, Mahalanabis D, Hamadani JD, Zetterstrom R. Reduced osmolarity oral rehydration salt in cholera. Scand J Infect Dis 1996;28:87-90. [CrossRef] [PubMed]
- Alam NH , Majumder RN, Fuchs GJ, and CHOICEStudy Group. Efficacy and safety of oral rehydration solution with reduced osmolarity in adults with cholera: a randomized double-blind clinical trial.Lancet 1999;354:296-9. [CrossRef]
- Ribiero H Jr, Ribiero T, Mattos A, et al. Treatment of acute diarrhea with oral rehydration solutions containing glutamine. J Am Coll Nutr. 1994;13: 251-255. [CrossRef]
- Molla AM, Molla A, Nath SK , Khatun M. Food based oral rehydration salt solution for acute childhood diarrhea. Lancet. 1989; 2 : 429-431. [CrossRef]
- Fontaine O, Gore SM, Pierce NF. Rice based oral rehydration solution for treating diarrhea. Cochrane Database Syst Rev. 2000(2); CD001264. [PubMed]
- Bhan MK, Mahalanabis D, Fontaine O, Pierce NF. Clinical trials of improved oral rehydration salt formulations: a review. Bull World Health Organ. 1994;72:945-955. [PubMed] [PMC free article]
- Farthing MJ. Disease related animal models for optimizing oral rehydration solution composition. Acta Paediatr Scand Suppl. 1989;364:23-30. [CrossRef] [PubMed]
- CHOICE Study Group. Multicentre, randomized, double blind clinical trial to evaluate the efficacy and safety of a reduced osmolarity oral rehydration salts solution in children with acute watery diarrhoea. Pediatrics, 2001; 107: 613-18. [CrossRef]
- Hahn S, Kim Y, Garner P. Reduced osmolarity oral rehydration solution for treating dehydration due to diarrhoea in children: systematic review. BMJ 2001;323: 81-5. [CrossRef] [PubMed] [PMC free article]
- Reduced osmolarity oral rehydration salts (ORS) formulation. A report from a meeting of experts jointly organized by UNICEF and WHO. UNICEF HOUSE, New York , USA , 18 July, 2001. WHO/FCH/CAH/0.1.22.
- Bhatnagar S, Bhandari N, Mouli UC, Bhan MK. Consensus Statement of IAP National Task Force: Status Report on Management of Acute Diarrhea. Indian Pediatrics 2004; 41:335-348. [PubMed]
- Pulungsih SP , Punjabi NH , Rafli K , Rifajati A , Kumala S , Simanjuntak CH , Yuwono , Lesmana M , Subekti D , Sutoto , Fontaine O. Standard WHO-ORS versus reduced-osmolarity ORS in the management of cholera patients. J Health Popul Nutr. 2006 Mar; 24(1):107-12. [PubMed]
- Edejer TT, Aikins M, Black RE, Wolfson L, Hutubessy R, Evans DB. Cost effectiveness analysis of strategies for child health in developing countries. BMJ, doi:10.1136 /bmj.38652.550278.7C (published 10 November 2005 ).
- Bahl R , Frost C , Kirkwood BR , Edmond K , Martines J , Bhandari N , Arthur P. Infant feeding patterns and risks of death and hospitalization in the first half of infancy: multicentre cohort study. Bull World Health Organ. 2005 Jun; 83 (6):418-26. [PubMed] [PMC free article]
- Effect of community-based promotion of exclusive breastfeeding on diarrhoeal illness and growth: a cluster randomised controlled trial Bhandari N, Bahl R, Mazumdar S, Martines J, Black RE, Bhan MK, and the other members of the Infant Feeding Study Group. Lancet 2003; 361: 1418-23 [CrossRef]
- Martorell, R., Yarbrough, C. & Klein, R. E. (1980) The impact of ordinary illnesses on the dietray intakes of malnourished children. Am. J. Clin. Nutr. 33: 345-350. [CrossRef] [PubMed]
- Brown, K. H., Black, R. E., Robertson, A. D. & Becker, S. (1985) Effects of season and illness on the dietary intake of weanlings during longitudinal studies in rural Bangladesh. Am. J. Clin. Nutr. 41: 343-355. [CrossRef] [PubMed]
- Rowland, M.G.M., Rowland, S.G.J.G. & Cole, T. J. (1988) Impact of infection on the growth of children from 0 to 2 years in an urban West African community. Am. J. Clin. Nutr. 47: 134-138. [CrossRef] [PubMed]
- Lutter, C. K., Mora, J. O., Habicht, J.-P., Rasmussen, K. M., Robson, D. S. & Sellers, S. G. (1989) Nutritional supplementation: effects on child stunting associated with diarrhea. Am. J. Clin. Nutr. 50: 1-8. [CrossRef] [PubMed]
- Black, R. E., Brown, K. H. & Becker, S. (1984) Malnutrition is a determining factor in diarrheal duration, but not incidence, among young children in a longitudinal study in rural Bangladesh. Am. J. Clin. Nutr. 39: 87-94. [CrossRef]
- Palmer, D. L., Koster, F. T., Alam, A.K.M.J. & Islam, M. R. (1976) Nutritional status: a determinant of severity of diarrhea in patients with cholera. J. Infect. Dis. 134: 8-14. [CrossRef]
- Samadi, A., Chowdhury, A. I., Huq, M. I. & Shahid, N. S. (1985) Risk factors for death in complicated diarrhoea of children. Br. Med. J. 290: 1615- 1617. [CrossRef]
- Sadhu BK, Isolauri E, Walker-Smith J A, Banchini G, Van Caillie-Bertrand M, Dias JA et al. Early feeding in childhood gastroenteritis. J.Pediatr.Gastroenterol. Nutr. 1997; 24: 522-527. [CrossRef]
- Alam S, Ahmad SA, Kumar S. Dietary Regimen For Persistent Diarrhea in infants under four months. Indian Pediatrics 2001; 38: 396-400. [PubMed]
- Wan C, Phillips MR, Dibley MJ, Liu Z. Randomised trial of different rates of feeding in acute diarrhea. Arch Dis Child 1999; 81 :487-491. [CrossRef] [PubMed] [PMC free article]
- Brown, K. H. (1994) Dietary management of acute diarrheal disease: contemporary scientific issues. J. Nutr. 124: 1455S-1460S. [CrossRef] [PubMed]
- Brown, K. H., Perez, F., Peerson, J. M., Fadel, J., Brunsgaard, G., Ostrom, K. M. & MacLean, W. C., Jr. (1993) Effect of dietary fiber (soy polysaccharide) on the severity, duration, and nutritional outcome of acute, watery diarrhea in children. Pediatrics 92: 241-247. [PubMed]
- Sandstead HH. Is zinc deficiency a public health problem? Nutrition 1995;11:87-92. [PubMed]
- Hambridge KM. Zinc deficiency in young children. Am J Clin Nutr l997;65:160-161.
- Gibson RS. Zinc nutrition in develop in g countries. Nutr Res Rev 1994;7:151-73. [CrossRef] [PubMed]
- Krebs FM, Hambidge KM. Zinc requirements and zinc intakes of breast-fed infants. Am J Clin Nutr l986;43:288-292. [CrossRef]
- Ruz M, Solomons N. Fecal zinc excretion of endogenous zinc during oral rehydration therapy for acute diarrhea: nutritional implications. J Trace Elem Exp Med 1995; 7:89-100.
- Fuchs GJ. Possibilities for zinc in the treatment of acute diarrhea. Am J Clin Nutr 1998;68(suppl):480S-3S. [CrossRef] [PubMed]
- Roy SK, Behrens RH, Haider R, Akrumuzzaman SM, Mahalanabis D, Wahed MA, et al. Impact of zinc supplementation on intestinal permeability in Bangladeshi children with acute diarrhoea and persistent diarrhoea syndrome. J Pediatr Gastroenterol Nutr 1992;15:289--96. [CrossRef] [PubMed]
- Bhandari N, Bahl R, Taneja S, Strand T, Molbak K, Johan R, et al. Substantial reduction in severe diarrheal morbidity by daily zinc supplementation in young north Indian children. Pediatrics 2002;109:86. [CrossRef]
- Baqui AH, Zaman K, Persson LA, Arifeen SE, Yunus M, Begum N, et al. Simultaneous weekly supplementation of iron and zinc is associated with lower morbidity due to diarrhea and acute lower respiratory infection in Bangladeshi infants. J Nutr 2003;133:4150-4157. [CrossRef] [PubMed]
- Rahman MM, Vermund SH, Wahed MA, Fuchs GJ, Baqui AH, Alvarez JO. Simultaneous zinc and vitamin A supplementation in Bangladeshi children: randomised double blind controlled trial. BMJ 2001;323;314-318. [CrossRef] [PubMed] [PMC free article]
- Zinc Investigators' Collaborative Group. Bhutta ZA, Bird SM, Black RE, Brown KH, Gardner JM, Hidayat A, et al. Therapeutic effects of oral zinc in acute and persistent diarrhea in children in developing countries: pooled analysis of randomized controlled trials. Am J Clin Nutr 2000;72:1516-1522. [CrossRef] [PubMed]
- Sazawal S, Black RE, Bhan MK, Bhandari N, Sinha A, Jalla S. Zinc supplementation in young children with acute diarrhea in India. N Engl J Med 1995;333:839--44. [CrossRef] [PubMed]
- Roy SK, Tomkins AM, Akramuzzaman SM, Behrens RH, Haider R, Mahalanabis D. Randomised controlled trial of zinc supplementation in malnourished Bangladeshi children with acute diarrhoea. Arch Dis Child 1997;77;196-200. [CrossRef] [PubMed] [PMC free article]
- Hidayat A, Achadin A, Sunoto, Soedarmo SP. The effect of zinc supplementation in children under three years of age with acute diarrhea in Indonesia. Med J Indonesia 1998;88:1564-160. [CrossRef]
- Strand TA, Chandyo RK, Bahl R, et al. Effectiveness and efficacy of zinc for the treatment of acute diarrhea in young children. Pediatrics 2002;109:898--903. [CrossRef] [PubMed]
- Dutta P, Mitra U, Datta A, Niyogi SK, Dutta S, Manna B, et al. Impact of zinc supplementation in malnourished children with acute diarrhea. J Trop Pediatr 2000;46:259-263. [CrossRef] [PubMed]
- Bhatnagar S, Bahl R, Sharma PK, Kumar GT, Saxena SK, Bhan MK. Zinc with oral rehydration therapy reduces stool output and duration of diarrhea in hospitalized children: a randomized controlled trial. J Pediatr Gastroenterol Nutr 2004; 38:34-40. [CrossRef] [PubMed]
- Bahl R, Bhandari N, Saksena M, et al. Efficacy of zinc-fortified oral rehydration solution in 6- to 35-month-old children with acute diarrhea. J Pediatr 2002;141: 677--82. [CrossRef] [PubMed]
- Fischer Walker CL, Bhutta, Z A., Bhandari N, Teka T, Shahid F, Taneja S et al Zinc Supplementation for the Treatment of Diarrhea in Infants in Pakistan, India and Ethiopia. J of Pediatr Gastroenterol & Nutr. September 2006; 43(3):357-363. [CrossRef] [PubMed]
- Awasthi S , INCLEN Childnet Zinc Effectiveness for Diarrhea (IC-ZED) Group. Zinc supplementation in acute diarrhea is acceptable, does not interfere with oral rehydration, and reduces the use of other medications: a randomized trial in five countries J Pediatr Gastroenterol Nutr. 2006 Mar;42(3):300-5 [CrossRef] [PubMed]
- Sarkar K, Ghosh S, Niyogi SK, Bhattacharya SK. Shigella dysenteriae type 1 with reduced susceptibility to fluoroquinolones. Lancet 2003; 361 : 785. [CrossRef]
- Sur D, Niyogi SK, Sur S, Datta KK, Takeda Y, Nair GB et al. Multidrug-resistant Shigella dysenteriae type 1: forerunners of a new epidemic strain in eastern India. J Emerg Infect Dis 2003; 9 : 404-405. [CrossRef] [PubMed] [PMC free article]
- Dutta S, Ghosh A, Ghosh K, Dutta D, Bhattacharya SK, Nair GB, Yoshida S. Newly Emerged Multiple-Antibiotic-Resistant Shigella dysenteriae type 1 Strains in and around Kolkata, India, Are Clonal, Journal of Clinical Microbiology, 2003; 41: 12: 5833-5834. [CrossRef] [PubMed] [PMC free article]
- Dutta S, Dutta D, Dutta P, Matsushita S, Bhattacharya SK, Yoshida S. Shigella dysenteriae Serotype 1, Kolkata, India. Emerging Infectious Diseases 2003; 9: 11: 1471-1474. [CrossRef] [PubMed] [PMC free article]
- Pazhani GP, Sarkar B, Ramamurthy T, Bhattacharya SK, Takeda Y, Niyogi SK. Clonal Multidrug-Resistant Shigella dysenteriae type 1 Strains Associated with Epidemic and Sporadic Dysenteries in Eastern India Antmicrobial Agents Chemother 2004; 48: 2: 681-684.
- Niyogi SK , Pazhani GP. Multiresistant Shigella species isolated from childhood cases from Kolkata , India. Jpn J Inf Dis 2003; 56: 33-34. [PubMed]
- Niyogi SK, Sarkar K, Lalmalsawma P, Pallai N, Bhattacharya SK. An Outbreak of Bacillary Dysentery Caused by Quinolone-resistant Shigella dysenteriae type 1 in a Northeastern State of India. J Health Popul Nutr 2004 Mar; 22(1): 97. [PubMed]
- Talukder KA, Islam AM, Khajanchi BK, Dutta DK, Islam Z, Safa A, Alam K, Hossain A, Nair GB, Sack DA. Temporal Shifts in the Dominance of Serotypes of Shigella dysenteriae from 1999 to 2002 in Dhaka, Bangladesh. J. Clin. Microbiol, 2003; 41: 11: 5053-5058. [CrossRef] [PubMed] [PMC free article]
- Talukder KA, Islam Z, Islam MA, Dutta DK, Safa A, Ansaruzzaman M, Faruque ASG, Shahed SN, Nair GB, Sack DA. Phenotypic and genotypic characterization of provisional serotype Shigella flexneri 1c and clonal relationships with 1a and 1b strains isolated in Bangladesh. J. Clin. Microbiol. 2003; 41: 1: 110-117. [CrossRef] [PubMed] [PMC free article]
- Zafar A , Sabir N , Bhutta ZA. Frequency of isolation of shigella serogroups/serotypes and their antimicrobial susceptibility pattern in children from slum areas in Karachi. J Pak Med Assoc. 2005; 55(5): 184-188. [PubMed]
- Ahmed K, Shakoori FR, Shakoori AR. Aetiology of Shigellosis in Northern Pakistan. J Health Popul Nutr 2003; 21: 1: 32-39. [PubMed]
- Taneja N, Mohan B, Khurana S, Sharma M. Antimicrobial resistance in selected bacterial enteropathogens in north India. Indian J Med Res 2004; 120: 39-43. [PubMed]
- Jesudason MV. Shigella isolation in Vellore , south India (1997-2001). Indian J Med Res 2002; 115: 11-13. [PubMed]
- International Note - Antibiotics in the management of shigellosis. WHO Weekly Epidemiological Record 2004; 79: 24.
- Sengupta PG , Niyogi SK , Bhattacharya SK. An outbreak of El tor cholera in Aizwal town of Mizoram , India. J Commun Dis. 2000 Sep; 32(3): 207-211. [PubMed]
- Samal B , Ghosh SK , Mohanty SK , Patnaik K. Epidemic of Vibrio cholerae serogroup O139 in Berhampur, Orissa. Indian J Med Res. 2001 Jul; 114:10-11. [PubMed]
- Garg P , Chakraborty S , Basu I , Datta S , Rajendran K , Bhattacharya T , Yamasaki S , Bhattacharya SK , Takeda Y , Nair GB , Ramamurthy T. Expanding multiple antibiotic resistance among clinical strains of Vibrio cholerae isolated from 1992-7 in Calcutta , India. Epidemiol Infect. 2000 Jun; 124(3): 393-399. [CrossRef] [PubMed] [PMC free article]
- Taneja N , Biswal M , Tarai B , Sharma M. Emergence of Vibrio cholerae O1 Biotype El Tor serotype Inaba in north India. Jpn J Infect Dis. 2005 Aug; 58(4): 238-240. [PubMed]
- Das S , Gupta S. Diversity of Vibrio cholerae strains isolated in Delhi , India , during 1992-2000. J Health Popul Nutr. 2005 Mar; 23(1): 44-51. [PubMed]
- Saha D , Khan WA , Karim MM , Chowdhury HR , Salam MA , Bennish ML. Single-dose ciprofloxacin versus 12-dose erythromycin for childhood cholera: a randomised controlled trial. Lancet. 2005 Sep 24-30; 366(9491): 1054-1055. [CrossRef]
- Bhattacharya MK, Dutta D, Ramamurthy T,Sarkar D, Singharoy A, Bhattacharya SK. Azithromycin in the treatment of cholera in children Acta Pediatr 2003;92 (6) :676-678.
- Sazawal S, Hiremath G, Dhingra U, Malik P, Deb S, Black R E. Efficacy of probiotics in prevention of acute diarrhoea: a meta-analysis of masked, randomised, placebo-controlled trials. Lancet Infect Dis 2006; 6: 374-82. [CrossRef]
- Costa-Ribeiro H, Ribeiro TC, Mattos AP, Valois SS, Neri DA, Almeida P, Cerqueira CM, Ramos E, Young RJ, Vanderhoof JA. Limitations of probiotic therapy in acute, severe dehydrating diarrhea. J Pediatr Gastroenterol Nutr. 2003; 36(1): 112-115. [CrossRef] [PubMed]
- Salazar-Lindo E, Miranda-Langschwager P, Campos-Sanchez M, Chea-Woo1 E and Bradley Sack R. Lactobacillus casei strain GG in the treatment of infants with acute watery diarrhea: A randomized, double-blind, placebo controlled clinical trial [ISRCTN67363048] BMC Pediatrics 2004, 4:18. [CrossRef] [PubMed] [PMC free article]
- Khanna V, Alam S, Malik A, Malik A. Efficacy of tyndalized Lactobacillus acidophilus in Acute Diarrhea. Indian J Pediatr 2005; 72: 935-8. [CrossRef] [PubMed]
- Huang JS, Bousvaros A, Lee JW, Diaz A, Davidson EJ. Efficacy of probiotic use in acute diarrhea in children: a meta-analysis. Dig Dis Sci 2002; 47(11): 2625-2634. [CrossRef] [PubMed]
- Van Niel CW, Feudtner C, Garrison MM, Christakis DA. Van Niel CW, Feudtner C, Garrison MM, Christakis DA. Lactobacillus therapy for acute infectious diarrhea in children: a meta-analysis. Pediatrics. 2002; 109(4): 678-684. [CrossRef] [PubMed]
- Szajewska H, Mrukowicz JZ. Probiotics in the treatment and prevention of acute infectious diarrhea in infants and children: a systematic review of published randomized, double blind, placebo-controlled trials. J Pediatr Gastroenterol Nutr. 2001; 33 Suppl 2:S17-25. [CrossRef] [PubMed]
- Raza S, Graham SM, Allen SJ, Sultana S, Cuevas L, Hart CA. Lactobacillus GG promotes recovery from acute nonbloody diarrhea in Pakistan. Pediatr Infect Dis J. 1995; 14:107-111. [CrossRef] [PubMed]
- Salazar-Lindo E, Santisteban-Ponce J, Chea-Woo E, Gutierrez M. Racecadotril in the treatment of acute watery diarrhea in children. N Engl J Med 2000; 343: 463-467. [CrossRef] [PubMed]
- Cezard JP, Duhamel JF, Meyer M, Pharaon I, Bellaiche M, Maurage C. et al. Efficacy and tolerability of Racecadotril in acute diarrhea in children. Gastroenterology 2001; 120: 799-805 [CrossRef] [PubMed]
- Alam NH, Ashraf H, Khan WA, Karim MM, Fuchs GJ. Efficacy and tolerability of Racecadotril in the treatment of cholera in adults: A double blind, randomized, controlled clinical trial. Gut 2003; 52: 1419-1423. [CrossRef] [PubMed] [PMC free article]
- Wang H-H, Shieh M-J, Liao K-F. A blind, randomized comparison of Racecadotril and loperamide for stopping acute diarrhea in adults. World J Gastroenterol 2005; 11(10): 1540-1543. [CrossRef] [PMC free article]
- Cezard JP, Salazar-Lindo E, Racecadotril in Acute Diarrhea. Indian Pediatr 2005; 42: 502-503. [PubMed]
- Bhan MK. Racecadotril. Is There Enough Evidence to Recommend it for Treatment of Acute Diarrhea? Editorial. Indian Pediatr 2004; 41: 1203-1204. [PubMed]
- Rossignol J-F A, Ayoub A, Ayers M S. Treatment of Diarrhea caused by Cryptosporidium parvum: A Prospective Randomized Double-Blind Placebo-Controlled Study of Nitazoxanide. J Inf Dis 2001; 184:103-106. [CrossRef] [PubMed]
- Elvia D, Jaime M, Enrique R, Rosamaria B. Epidemiology and control of intestinal parasites with Nitazoxanide in children in Mexico: Am J Trop Med Hyg 2003; 68(4): 384-385. [CrossRef]
- Amadi B, Mwiya M, Musuku J, Watuka RN A, Sianongo S, Ayoub A, Kelly P. Effect of Nitazoxanide on morbidity and mortality in Zambian children with Cryptosporidiosis: a randomised controlled trial. Lancet 2002; 360 (9343 ): 1375-1380. [CrossRef]
- Davila-Gutierrez CE, Vasquez C, Trujillo-Hernaandez B, Miguel H. Nitazoxanide Compared With Quinfamide And Mebendazole In The Treatment Of Helminthic Infections And Intestinal Protozoa In Children. Am J Trop Med Hyg 2002; 66(3): 251-254. [CrossRef] [PubMed]
|
| Cite this article as: | | ALAM S, KHANNA R, FIRDAUS U. ACUTE CHILDHOOD DIARRHEA: A REVIEW OF RECENT ADVANCES IN THE STANDARD MANAGEMENT. Pediatr Oncall J. 2006;3: 72. |
|