Kamel Abidi, Merieme Ferjani, Manel Jallouli, Tahar Gargah.
Nephropediatric Department, Charles Nicolle Hospital, Tunisia.
ADDRESS FOR CORRESPONDENCE
Dr. Kamel Abidi, Nephropediatric Department, Charles Nicolle Hospital, 9 April Street, Bab Souika, 1006 Tunis, Tunisia.
Email: abidikamel8@yahoo.fr | | Abstract | | Sclerosing encapsulating peritonitis is the most serious complication of continuous peritoneal dialysis with a mortality that exceeds 30%. It remains rare in children. The diagnosis is difficult and an abdominal CT should be performed promptly to establish the right diagnosis. We present a 12 years old boy who developed sclerosing encapsulating peritonitis while on peritoneal dialysis who was successfully treated with tamoxifen. | | | | Keywords | | peritoneal dialysis, renal transplantation, sclerosing encapsulating peritonitis, tamoxifen | | | | Introduction | | Sclerosing encapsulating peritonitis (EPS) is the most serious complication of continuous peritoneal dialysis (CPD) with a mortality that exceeds 30%. (1) It is characterized by partial or diffuse bowel obstruction accompanied by diffuse appearance of marked sclerotic thickening of the peritoneal membrane or bowel wall. (2) There have been many reports of the incidence of EPS being strongly correlated to the duration of CPD. (1) The diagnosis is difficult and an abdominal CT should be performed promptly to establish the right diagnosis. The existence of peritoneal thickening in the abdominal CT is suggestive of this disease. EPS is rare in children. (1) We present a 12 years old boy who developed EPS while on peritoneal dialysis (PD). | | | | Case Report | A 12-year old boy with end-stage renal failure secondary to posterior urethral valves on continuous ambulatory peritoneal dialysis (CAPD) from 7 years of age presented with vomiting, abdominal pain, diarrhea and fever lasting for five days. During CAPD, he had recurrent peritonitis with frequency of 2.6 episodes/year. He was suspected to have peritonitis based on a cloudy dialysate effluent and peritoneal fluid showing white cell count of 150 cells/cumm with culture growing staphylococcus aureus. He was initially started on ceftazidime (100 mg/kg/day) with vancomycin (40 mg/kg/day) and then only vancomycin was given for 15 days. Over the next 3 days, his symptoms deteriorated. Plain abdominal X-ray abdomen revealed multiple air fluid levels in the left upper quadrant suggestive of intestinal obstruction. Abdominal ultrasound (USG) showed left colon distension with accelerated peristalsis with associated inter-loop fluid. He was treated by nasogastric aspiration and rectal enemas. Peritoneal dialysis was stopped for a while and he was switched on hemodialysis for few weeks. After that his symptoms improved and he returned to peritoneal dialysis. After a year he was admitted for the same symptoms of two days duration. Examination revealed abdominal distension with diffuse tenderness. X-rays revealed multiple and diffuse air fluid levels. Abdominal CT showed clustered small bowel loops within a peritoneal sac like structure, a flattened terminal small bowel and dilatation of the rest of the small bowel before (fig 1, 2) suggestive of sclerosing encapsulating peritonitis. Biopsy was not done as CT abdomen was diagnostic. Laxatives were needed in the beginning for defecation along with nasogastric aspiration and his symptoms improved rapidly. Then treatment with tamoxifen 10 mg daily in addition with prednisone 0.5 mg/kg/day was initiated for three months. His symptoms improved gradually over 2 months with an increase of serum albumin and body weight. Peritoneal dialysis was stopped and he was kept under hemodialysis. He underwent a renal transplantation after 8 months on hemodialysis. Now, 13 months after the transplantation the patient has shown no recurrent symptoms of his EPS.
Fig 1: CT scan of the abdomen showing diffuse thickening of parietal peritoneum, mesenteric thickening with vascular engorgement.
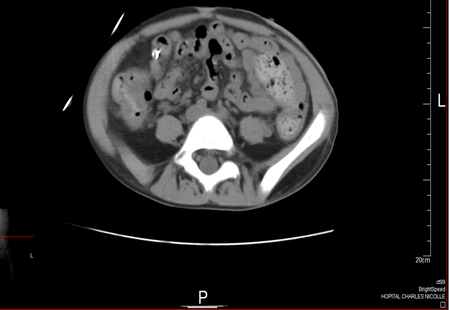
Fig 2: Abdominal CT showing clustered small bowel loops within a peritoneal sac like structure, a flattened terminal small bowel and dilatation of the rest of the small bowel
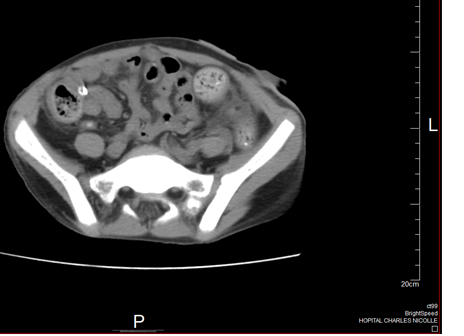
| | | | Discussion | EPS is a rare complication of CAPD. It is reported to affect up to 1.7% of patients undergoing CAPD. It was described and named “Abdominal Cocoon” by Foo et al. (2) Since then, there have been only a few sporadic reports. (3) It is characterized by total or partial encasement of the small bowel by a thick, fibrous membrane. It should be distinguished from simple peritoneal sclerosis. The latter is almost always present, in variable degrees, in PD patients on dialysis for more than a few months and leads to a deterioration of membrane permeability. Bio-incompatibility of solutions and time on PD are its presumed etiological factors. (1) Usually, EPS presents as small bowel obstruction, malnutrition, sometimes with bloodstained dialysate, and is characterized by cocooning of the small bowel by a very thick fibrous sheath, often with sparse calcified plates lining the peritoneum. (3) EPS is due to an inflammatory process resulting in loss of the mesothelial layer of the peritoneum and fibro connective tissue proliferation. (1) Risk factors for EPS include recurrent peritonitis, decreased ultra filtration, chlorhexidine gluconate in alcohol to clean the PD catheter and long administration of hypertonic glucose based dialysis solutions. (3) In particular, studies concluded that prolonged PD duration constitutes the single most significant risk factor for EPS. A recent study (4) showed an exponential increase in EPS incidence with time on PD: from 1.9% for patients on dialysis less than 2 years, to 6.4%, 10.8%, and 19.4% for more than 5, 6, and 8 years, respectively, suggesting the duration of the exposure of the peritoneum to PD to be a major risk factor. However, some cases occur early in the course of PD. Our patient in fact was undergoing PD for five years and used frequently hypertonic PD solutions. A history of severe peritonitis and/or non-resolving peritonitis, especially in the long term PD patient, is also particularly problematic and likely contributory. (4)
Clinically symptoms range from recurrent episodes of abdominal pain, malnutrition, nausea, vomiting, weight loss, low-grade fever, hemorrhagic effluent, ultrafiltration failure, ascites and resistance to recombinant human erythropoietin n. (5) A decline in ultrafiltration is consistent and dialysis effluent may become blood stained. Often early features are nonspecific and diagnosis is missed until the patient develops partial or complete intestinal obstruction. An abdominal mass may also be present due to an encapsulated cluster of dilated small bowel loops. (6) Imaging is useful for preoperative diagnosis but the findings are not pathognomonic. Plain abdominal films are of low utility and may show dilated small bowel loops, air-fluid levels, and calcified plaques. Barium studies may show adherent bowel loops and delayed transit time with a normal mucosal pattern. (1) USG can show peritoneal thickening and a preperitoneal membrane, forming the typical image of trilayer membrane or “sandwich-like” membrane. The abdominal CT is the most sensitive modality for detecting EPS showing a typically thickened bowel wall, thickened peritoneal membranes, loculated fluid collections, increased density of mesenteric fat, peritoneal calcification and dilated loops of bowel adherent to each other or to other abdominal organs and to the abdominal wall. (1) However the final diagnosis is usually based on intraoperative and histopathology findings. In our patient, image of a cocoon bowel on abdominal CT and clinical presentation were diagnostic. We did not do a peritoneal biopsy as it was considered too risky.
Various treatment options have been suggested based on the progressive stage of the disease including immunosuppression, corticosteroids with parenteral nutrition, tamoxifen and surgical debridement. (7) The initial step in therapy should be the cessation of CAPD. Food intake is also necessary either parenteral or enteral. In the presence of intestinal obstruction and potential bowel necrosis, surgery is warranted and consists in adhesiolysis of the membranes. The outcome is influenced by the grade of the thickness of the adhesions. The prognosis is particularly poor in cases of long-term EPS and in cases with strong adhesions of the thickened membranes because of the danger of perforating the intestine. tamoxifen is a non-steroidal anti-estrogen that stimulates the production of transforming growth factor which stimulates the metalloproteinase MMP1 and MMP9. MMP9 degrades and denaturated type IV collagen and so favors mesothelial healing. (4) The addition of tamoxifen to steroid was very effective in our patient. | | | | Conclusion | | EPS is rare in children undergoing PD. There have been many reports of EPS being strongly related to the longevity of PD. EPS should be considered in children with abdominal complaints who have been treated with PD for a prolonged time. The mortality rate of EPS is high due to recurrence and sepsis. EPS may no longer be a fatal complication if PD is interrupted promptly and adequate food intake is provided as soon as possible. Surgery may resolve problems in case of bowel obstruction but exposes to other risks: enterotomies, anastomotic leaks and enterocutaneous fistulas. Even after kidney transplantation the risk of recurrence exists. | | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Masataka H, Bradley A. Warady. Long-term peritoneal dialysis and encapsulating peritoneal sclerosis in children. Pediatr Nephrol. 2010; 25 :75-81. [CrossRef] [PMC free article]
- Foo KT, Ng KC, Rauff A, Foong WC, Sinniah R. Unusual small intestinal obstruction in adolescent girls: the abdominal cocoon. Br J Surg. 1978;65:427-430. [CrossRef]
- Tan R, Betjes M, Cransberg K. Post-transplantation encapsulating peritoneal sclerosis in a young child. Nephrol Dial Transplant. 2011; 26 :3822-3824. [CrossRef]
- Moustafellos P, Hadjianastassiou V, Roy D, Velzeboer NE, Maniakyn N, Vaidya A, et al. Tamoxifen therapy in encapsulating sclerosing peritonitis in patients after kidney transplantation. Transplant Proc. 2006;38:2913-2914. [CrossRef]
- Tan FL, Loh D, Prabhakaran K. Sclerosing encapsulating peritonitis in a child secondary to peritoneal dialysis. J Pediatr Surg. 2005; 40 : e 21-23.
- Takeichi T, Narita Y, Lee KJ, Yamamoto H, Asonuma K, Inomata Y. Sclerosing encapsulating peritonitis after living donor liver transplantation: a case successfully treated with tamoxifen: report of a case. Surg Today. 2013; 43 :1326-1329. [CrossRef]
- Sharma D, Nair RP, Dani T, Shetty P. Abdominal cocoon-A rare cause of intestinal obstruction. Int J Surg Case Rep. 2013; 4:955-957. [CrossRef] [PMC free article]
DOI: https://doi.org/10.7199/ped.oncall.2015.11
|
| Cite this article as: | | Abidi K, Ferjani M, Jallouli M, Gargah T. Sclerosing Encapsulating Peritonitis due to Peritoneal Dialysis Successfully Treated with Tamoxifen. Pediatr Oncall J. 2015;12: 14-16. doi: 10.7199/ped.oncall.2015.11 |
|