Abstract
Intracranial hypertension has gone by numerous different terms over the years. Symptoms of intracranial hypertension include a headache that can be exacerbated by maneuvers such as Valsalva, bending over or coughing. Other symptoms include nausea, vomiting, diplopia, cranial nerve palsy, and tinnitus. The ophthalmic exam is a fundamental part of the diagnosis and management of intracranial hypertension. A direct funduscopic exam of the optic nerves should be performed at each visit. Diagnostic neuroimaging should include MRI and MRV to rule out secondary causes. The largest diagnostic component is the lumbar puncture. Proper positioning includes lateral decubitus with the legs and heads extended at the time of measurement. Weight loss (if obese) and acetazolamide are considered the first-line treatment for intracranial hypertension. Recurrence is estimated to occur in up to 20% of patients. The development of migraine or other headache syndromes is seen in up to 68% following resolution of their intracranial hypertension.
History
The original description of intracranial hypertension has been credited to Heinrich Quincke, who pioneered the lumbar puncture. In 1897, he described a series of patients suffering from increased intracranial pressure with normal cerebral spinal fluid (CSF) and labeled this condition “meningitis serosa”.1 Since Quincke’s initial description, there have been many different names used including serous meningitis, otitis hydrocephalus, toxic hydrocephalus, and hypertensive meningeal hydrops. In 1904, the term “pseudotumor cerebri” was coined by Nonne after similarities in a presentation to patients with an intracranial mass.2 In 1955, Foley suggested “benign intracranial hypertension” to remedy the negative association with a “pseudo-cancer” diagnosis.3 The term idiopathic intracranial hypertension appeared in the 1980s following a series of reports describing permanent visual deficits.
Today, pseudotumor cerebri and idiopathic intracranial hypertension remain the most common terminology amongst both practitioners and the lay public. There remains both ambiguity and confusion with both of these terms. There have been recent attempts to clarify the terminology with some using a broader designation of “pseudotumor cerebri syndrome” which still includes the designation of idiopathic when a cause is not found.4 An easier designation is the use of the terms primary intracranial hypertension (PIH) and secondary intracranial hypertension (SIH).5 The designation of the primary would apply when no precipitant that would result in increased pressure is present. Patients may still have risk factors for increased intracranial pressure, such as female gender, post-pubertal status, obesity, or polycystic ovarian syndrome, though these conditions do not directly result in increased pressure. SIH is then reserved for instances where the intracranial hypertension is the direct result of another condition, such as cerebral sinus venous thrombosis, or minocycline use.
PIH has traditionally been considered a rare entity. The annual incidence in adults residing in the United States is estimated at 0.9 per 100,000, with no good estimates in the pediatric population.6 The annual pediatric incidence is estimated at 0.47 and 1.2 per 100,000 in Germany and Croatia respectively.7,8 In the provinces of Nova Scotia and Prince Edward Island, the annual incidence was 0.9 per 100,000 in children 2 to 15 years old between 1979 and 1994.9 Interestingly from 1997 to 2007, the annual incidence fell to 0.6 per 100,000 in children 2 to 16 years, despite increased childhood obesity during this period.10
Diagnostic Criteria
The diagnostic criteria for adult PIH were developed from a series of 22 patients reported by Dandy in 1937.11 The main limitation of this set of criteria is that imaging of the time was limited to pneumoencephalograms. In 1985, Smith proposed modernization of the criteria into what is now accepted as the modified Dandy criteria to include more current imaging criteria. The criteria stipulate that patients have: 1) signs and symptoms of raised intracranial pressure (headache, nausea, vomiting, transient visual obscurations, or papilledema), 2) absence of localizing neurologic signs with the exception of unilateral or bilateral abducens nerve palsy, 3) CSF opening pressure of >25 cm H20 with normal composition, 4) normal to small ventricles as demonstrated by computed tomography (CT) study (today magnetic resonance imaging (MRI)).12
Secondary Intracranial Hypertension
Numerous medications have been reported to lead to SIH. The most widely known is the tetracycline class of medications, including tetracycline, minocycline and doxycycline.17-19 Chronic corticosteroid use can result in increased pressure following weight gain from chronic use, but also with acute withdrawal or rapid wean.
Patients being treated with recombinant growth hormones can develop symptoms of intracranial hypertension including headache and optic nerve edema.20, 21 The timing from initiation of growth hormone to presentation is often in the first 12 weeks of treatment, though it can occur years after treatment was started.21 Treatment includes cessation of the growth hormone and often acetazolamide until symptom resolution. Once symptoms resolve, patients can be restarted on a lower dose of growth hormone with gradual titration without recurrence of symptoms.
Other medications reported in the literature include lithium, nalidixic acid, hypervitaminosis A, hypovitaminosis A, and All-Trans Retinoic Acid, a vitamin A derivative. Oral contraceptives are reported to be associated with SIH, though the association is likely due to CVST from the use of contraceptives.
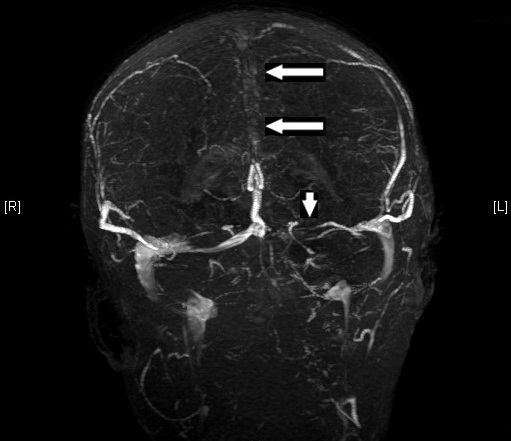
Cerebral Venous Sinus Thrombosis (CVST) is a common non-medication cause of SIH. Superior sagittal sinus and the transverse sinuses are most often seen (figure 1), though thrombosis of any venous outflow can result in increased pressure. Prior to the widespread use of antibiotics to treat chronic otitis or mastoiditis, patients would develop thrombosis of the sigmoid sinus or jugular vein, formerly referred to as otitic hydrocephalus. Hypercoagulable states such as acute post-partum period, oral contraceptive use, cancer (lymphoma/leukemia), and various inherited coagulopathies can also result in CVST.
The initiation of feeding in cases of poor nutrition from starvation or malabsorption syndromes has resulted in a transient increase in intracranial pressure.22,23 Often a bulging of the anterior fontanel is the only symptom in infants. Older children may develop irritability and cranial suture separation. Fortunately, in most cases, it is a transient process that self resolves within a few days to weeks.
Figure 1. Superior sagittal sinus thrombosis (large arrows) and transverse sinus thrombosis (small arrow).
Common Causes of Secondary Intracranial Hypertension
- Medications
- Minocycline / Tetracycline / Doxycycline
- Corticosteroids (especially withdrawal)
- Growth hormone
- Lithium carbonate
- Cyclosporine A
- Cytarabine
- Nalidixic acid
- Retinoic acid
- Vitamin A (excess or deficiency)
- Vitamin D
-
Medical conditions
- Cerebral venous thrombosis
- Refeeding syndrome
- Adrenal insufficiency (often on steroids)
- Congestive heart failure
- Hypoparathyroidism (early in correction)
- Pregnancy/eclampsia
- Crohn’s disease
- Meningitis/encephalitis
- Intracranial hemorrhage
- Lyme disease
- Demyelinating disease / multiple sclerosis
- Leukemia
- Lymphoma
- Hydrocephalus
- Craniofacial syndrome
- Chiari Malformation
Presentation
In pediatrics, PIH is typically divided into pre-pubertal and pubertal groups. Pubertal patients have the same risk factors as adults, whereas obesity and gender are not prominent risk factors in pre-pubertal patients. Balcer et al. found that obesity did correlate with an increased risk of PIH in children over 11 years old.13 Bursztyn et al. found similar results in those >12 years old.10
Symptoms of intracranial hypertension tend to be less evident in younger individuals. In the pre-pubertal population, optic disc edema incidentally discovered on routine ophthalmologic examination is seen. Asymptomatic cases are often younger, have a male predominance, and a lower percentage of obesity.14 Up to 48% of cases lack papilledema, yet have other symptoms consistent with elevated intracranial pressure and documented elevated opening pressure.7,15,16
The most consistent symptom is a headache that is typically constant with variable severity throughout the day. Headaches tend to be most severe in the morning following prolonged horizontal positioning and may be exacerbated by maneuvers such as Valsalva, bending over, or coughing. Other symptoms include nausea, vomiting, and diplopia.
Cranial nerve VI palsy is the most common nerve palsy with patients complaining of diplopia with the horizontal gaze. Tinnitus is experienced by 10% of pediatric PIH patients and is often unreported until asked.8 Patients will report a “whooshing” sound coinciding with their heartbeat.
Papilledema refers to the presence of optic disc edema in the setting of verified increased intracranial pressure. Optic disc edema in absence of increased intracranial pressure can be seen in optic neuritis, neuroretinitis, anterior ischemic optic neuropathy, and infiltration of the optic nerve head by tumor cells. An afferent pupillary defect is rare in intracranial hypertension, and if found should raise concern for the possibility of optic neuritis. Pseudo-papilledema gives the appearance of optic nerve edema to those unfamiliar with the fundus exam (and even the experienced at times). Common culprits are anomalous optic nerves and optic nerve drusen. Unilateral or marked asymmetric edema should raise concern for one of these conditions.
1. Quincke H. Uber Meningitis serosa and verwandte Zustande. Dtsch Z Nervenheilkd. 1897;9:149-68.
2. Nonne M. Ueber Falle vom Symptomkomplex "tumor cerebri" mit Ausgang in Heilung. Dtsch Z Nervenheilkd. 1904;27:169-216.
3. Foley J. Benign forms of intracranial hypertension; toxic and otitic hydrocephalus. Brain. 1955;78(1):1-41. Epub 1955/01/01.
4. Friedman DI. The pseudotumor cerebri syndrome. Neurologic clinics. 2014;32(2):363-96. Epub 2014/04/08.
5. Aylward SC. Pediatric idiopathic intracranial hypertension: a need for clarification. Pediatr Neurol. 2013;49(5):303-4. Epub 2013/08/21.
6. Schexnayder LK, Chapman K. Presentation, investigation and management of idiopathic intracranial hypertension in children. Current Paediatrics. 2006;16(5):336-41.
7. Tibussek D, Distelmaier F, von Kries R, Mayatepek E. Pseudotumor cerebri in childhood and adolescence -- results of a Germany-wide ESPED-survey. Klinische Padiatrie. 2013;225(2):81-5. Epub 2013/03/26.
8. Dessardo NS, Dessardo S, Sasso A, Sarunic AV, Dezulovic MS. Pediatric idiopathic intracranial hypertension: clinical and demographic features. Collegium antropologicum. 2010;34 Suppl 2:217-21. Epub 2011/02/10.
9. Gordon K. Pediatric pseudotumor cerebri: descriptive epidemiology. The Canadian journal of neurological sciences Le journal canadien des sciences neurologiques. 1997;24(3):219-21.
10. Bursztyn LL, Sharan S, Walsh L, LaRoche GR, Robitaille J, De Becker I. Has rising pediatric obesity increased the incidence of idiopathic intracranial hypertension in children? Canadian journal of ophthalmology Journal canadien d'ophtalmologie. 2014;49(1):87-91.
11. Dandy WE. Intracranial Pressure without Brain Tumor: Diagnosis and Treatment. Ann Surg. 1937;106(4):492-513. Epub 1937/10/01.
12. Smith JL. Whence pseudotumor cerebri? J Clin Neuroophthalmol. 1985;5(1):55-6. Epub 1985/03/01.
13. Balcer LJ, Liu GT, Forman S, Pun K, Volpe NJ, Galetta SL, et al. Idiopathic intracranial hypertension: relation of age and obesity in children. Neurology. 1999;52(4):870-2. Epub 1999/03/17.
14. Bassan H, Berkner L, Stolovitch C, Kesler A. Asymptomatic idiopathic intracranial hypertension in children. Acta neurologica Scandinavica. 2008;118(4):251-5. Epub 2008/03/18.
15. Faz G, Butler IJ, Koenig MK. Incidence of papilledema and obesity in children diagnosed with idiopathic ''benign'' intracranial hypertension: case series and review. J Child Neurol. 2010;25(11):1389-92. Epub 2010/04/02.
16. Aylward SC, Aronowitz C, Roach ES. Intracranial Hypertension Without Papilledema in Children. J Child Neurol. 2015. Epub 2015/05/28.
17. Walters BN, Gubbay SS. Tetracycline and benign intracranial hypertension: report of five cases. Br Med J (Clin Res Ed). 1981;282(6257):19-20. Epub 1981/01/03.
18. Friedman DI, Gordon LK, Egan RA, Jacobson DM, Pomeranz H, Harrison AR, et al. Doxycycline and intracranial hypertension. Neurology. 2004;62(12):2297-9. Epub 2004/06/24.
19. Weese-Mayer DE, Yang RJ, Mayer JR, Zaparackas Z. Minocycline and Pseudotumor cerebri: The well-known but well-kept secret. Pediatrics. 2001;108(2):519-20. Epub 2001/08/09.
20. Rogers AH, Rogers GL, Bremer DL, McGregor ML. Pseudotumor cerebri in children receiving recombinant human growth hormone. Ophthalmology. 1999;106(6):1186-9; discussion 9-90. Epub 1999/06/12.
21. Noto R, Maneatis T, Frane J, Alexander K, Lippe B, Davis DA. Intracranial hypertension in pediatric patients treated with recombinant human growth hormone: data from 25 years of the Genentech National Cooperative Growth Study. J Pediatr Endocrinol Metab. 2011;24(9-10):627-31. Epub 2011/12/08.
22. Roach ES, Sinal SH. Increased intracranial pressure following treatment of cystic fibrosis. Pediatrics. 1980;66(4):622-3. Epub 1980/10/01.
23. Roach ES, Sinal SH. Initial treatment of cystic fibrosis. Frequency of transient bulging fontanel. Clinical pediatrics. 1989;28(8):371-3. Epub 1989/08/01.
24. Frisen L. Swelling of the optic nerve head: a staging scheme. Journal of neurology, neurosurgery, and psychiatry. 1982;45(1):13-8. Epub 1982/01/01.
25. Johnston I, Paterson A. Benign intracranial hypertension. II. CSF pressure and circulation. Brain. 1974;97(2):301-12. Epub 1974/06/01.
26. Corbett JJ, Mehta MP. Cerebrospinal fluid pressure in normal obese subjects and patients with pseudotumor cerebri. Neurology. 1983;33(10):1386-8. Epub 1983/10/01.
27. Bono F, Lupo MR, Serra P, Cantafio C, Lucisano A, Lavano A, et al. Obesity does not induce abnormal CSF pressure in subjects with normal cerebral MR venography. Neurology. 2002;59(10):1641-3. Epub 2002/11/27.
28. Whiteley W, Al-Shahi R, Warlow CP, Zeidler M, Lueck CJ. CSF opening pressure: reference interval and the effect of body mass index. Neurology. 2006;67(9):1690-1. Epub 2006/11/15.
29. Rangwala LM, Liu GT. Pediatric idiopathic intracranial hypertension. Surv Ophthalmol. 2007;52(6):597-617. Epub 2007/11/22.
30. Avery RA, Shah SS, Licht DJ, Seiden JA, Huh JW, Boswinkel J, et al. Reference range for cerebrospinal fluid opening pressure in children. N Engl J Med. 2010;363(9):891-3. Epub 2010/09/08.
31. Lee MW, Vedanarayanan VV. Cerebrospinal fluid opening pressure in children: experience in a controlled setting. Pediatr Neurol. 2011;45(4):238-40. Epub 2011/09/13.
32. Newman NJ, Selzer KA, Bell RA. Association of multiple sclerosis and intracranial hypertension. J Neuroophthalmol. 1994;14(4):189-92. Epub 1994/12/01.
33. Williams BJ, Skinner HJ, Maria BL. Increased intracranial pressure in a case of pediatric multiple sclerosis. J Child Neurol. 2008;23(6):699-702. Epub 2008/06/10.
34. Narula S, Liu GT, Avery RA, Banwell B, Waldman AT. Elevated cerebrospinal fluid opening pressure in a pediatric demyelinating disease cohort. Pediatr Neurol. 2015;52(4):446-9. Epub 2015/02/15.
35. Newborg B. Pseudotumor cerebri treated by rice reduction diet. Arch Intern Med. 1974;133(5):802-7. Epub 1974/05/01.
36. Ko MW, Chang SC, Ridha MA, Ney JJ, Ali TF, Friedman DI, et al. Weight gain and recurrence in idiopathic intracranial hypertension: a case-control study. Neurology. 2011;76(18):1564-7. Epub 2011/05/04.
37. Distelmaier F, Sengler U, Messing-Juenger M, Assmann B, Mayatepek E, Rosenbaum T. Pseudotumor cerebri as an important differential diagnosis of papilledema in children. Brain & development. 2006;28(3):190-5. Epub 2005/12/22.
38. Schoeman JF. Childhood pseudotumor cerebri: clinical and intracranial pressure response to acetazolamide and furosemide treatment in a case series. J Child Neurol. 1994;9(2):130-4. Epub 1994/04/01.
39. Celebisoy N, Gokcay F, Sirin H, Akyurekli O. Treatment of idiopathic intracranial hypertension: topiramate vs acetazolamide, an open-label study. Acta neurologica Scandinavica. 2007;116(5):322-7. Epub 2007/10/10.
40. Abubaker K, Ali Z, Raza K, Bolger C, Rawluk D, O'Brien D. Idiopathic intracranial hypertension: lumboperitoneal shunts versus ventriculoperitoneal shunts--case series and literature review. Br J Neurosurg. 2011;25(1):94-9. Epub 2011/02/18.
41. Digre KB, Bruce BB, McDermott MP, Galetta KM, Balcer LJ, Wall M. Quality of life in idiopathic intracranial hypertension at diagnosis: IIH Treatment Trial results. Neurology. 2015. Epub 2015/05/23.
42. Mulla Y, Markey KA, Woolley RL, Patel S, Mollan SP, Sinclair AJ. Headache determines quality of life in idiopathic intracranial hypertension. The journal of headache and pain. 2015;16(1):521. Epub 2015/05/20.
43. Stiebel-Kalish H, Kalish Y, Lusky M, Gaton DD, Ehrlich R, Shuper A. Puberty as a risk factor for less favorable visual outcome in idiopathic intracranial hypertension. Am J Ophthalmol. 2006;142(2):279-83. Epub 2006/08/01.
44. Gospe SM, 3rd, Bhatti MT, El-Dairi MA. Anatomic and visual function outcomes in paediatric idiopathic intracranial hypertension. The British journal of ophthalmology. 2015. Epub 2015/08/14.
45. Tibussek D, Schneider DT, Vandemeulebroecke N, Turowski B, Messing-Juenger M, Willems PH, et al. Clinical spectrum of the pseudotumor cerebri complex in children. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 2010;26(3):313-21. Epub 2009/11/11.
46. Ravid S, Shahar E, Schif A, Yehudian S. Visual Outcome and Recurrence Rate in Children With Idiopathic Intracranial Hypertension. J Child Neurol. 2015. Epub 2015/02/26.
47. Friedman DI, Rausch EA. Headache diagnoses in patients with treated idiopathic intracranial hypertension. Neurology. 2002;58(10):1551-3. Epub 2002/05/30.
48. Yri HM, Ronnback C, Wegener M, Hamann S, Jensen RH. The course of headache in idiopathic intracranial hypertension: a 12-month prospective follow-up study. European journal of neurology : the official journal of the European Federation of Neurological Societies. 2014;21(12):1458-64. Epub 2014/07/30.