Introduction
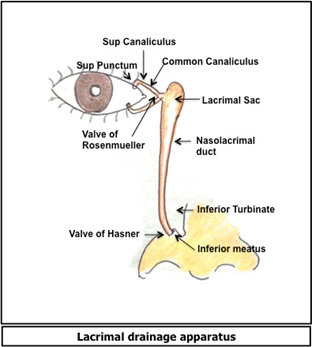
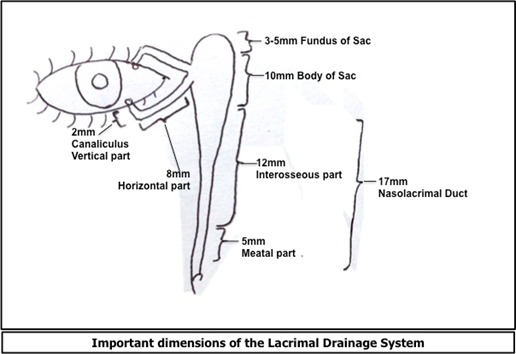
The tears from the conjunctival sac drain into the nose via the lacrimal drainage system. It consists of the inferior and superior puncta, the inferior and superior canaliculi, the common canaliculus, the lacrimal sac, and the nasolacrimal duct (NLD), which drains via the inferior meatus into the nose.
While obstruction at any level in the drainage apparatus, can lead to ‘epiphora’ or excessive watering of the eye, the site of obstruction in infants is most commonly at the Valve of Hasner, which is located at the nasal opening of the nasolacrimal duct1. Symptomatic congenital nasolacrimal duct obstruction (CNLDO) occurs in 5-6% of normal newborns2.
Etiopathogenesis
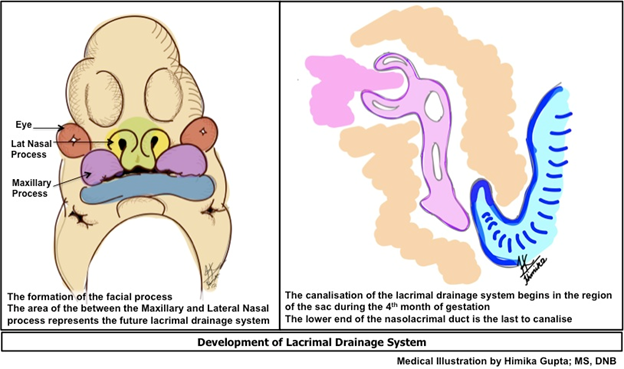
Embryology3
The lacrimal drainage system begins to develop when the embryo is at the 7 mm stage. The region between the lateral nasal and maxillary processes has a groove called the naso-optic fissure. Thickening occurs at the base of this groove and a solid rod of cells separate to move deep to the surface at 43 days of age. This solid cord begins to canalize at 4 months of age, first in the region of the lacrimal sac, then the canaliculi, and lastly the nasolacrimal duct. The membrane of the puncta is canalized by the 7th month of age, whereas the lower end of the nasolacrimal duct often has a persisting membrane even at birth. This imperforate membrane is the most common cause of neonatal epiphora. This also explains the higher incidence of congenital nasolacrimal duct obstruction (CNLDO) in premature infants4. Abnormal embryogenesis can lead to anomalies in other regions of the drainage apparatus too resulting in punctual atresia, absent canaliculus, duplicated canaliculus, lacrimal fistula, and diverticular.
Risk Factors
Children with Down’s syndrome and craniofacial anomalies have a higher incidence of nasolacrimal duct obstruction. Some other syndromes associated with congenital nasolacrimal duct obstruction are Treacher Collins and Crouzons. Some craniofacial anomalies associated with nasolacrimal duct obstruction are cleft lip, cleft palate, bifid uvula, and hemifacial microsomia8.
Congenital Nasolacrimal Duct Obstruction - Pathology
The obstructed nasolacrimal duct (NLD), doesn’t allow the proper drainage of the tears and the mucous secretions, thus the excess tears spill out of the eye resulting in epiphora.
Rarely in approximately 0.1% of babies with nasolacrimal duct obstruction, a dacryocystocoele is formed5. A dacryocystocoele is a distended lacrimal sac and is usually present perinatally6. It is formed when the amniotic fluid or secretions enter, accumulate, and distend the sac. This distension caused a kinking and functional obstruction of the common canaliculus not allowing the contents to escape. Thus valve-like mechanism leads to tense distension of the sac. It is also called an amniontocoele(amniotic fluid) or mucocele (mucous), indicating at the origin of the distending fluid, however, ‘dacryocystocoele’ is an inclusive term that describes the location of accumulation, as the origin of the fluid is not clinically distinguishable7.
Neonatal dacryocystocoeles are almost always associated with nasolacrimal duct cysts7. These are intranasal cysts formed due to increased hydrostatic pressure in the lacrimal drainage system, which causes outpouching of the imperforate membrane at the lower end of the NLD.
Symptoms
Most commonly, the parents complain of water from the baby’s eye or eyes. Around 5-6% of infants are symptomatic for lacrimal obstruction1. The condition is bilateral in one-third of the babies.1 Parents may also complain of discharge and stickiness or swelling in the region of the sac. Babies with dacryocystocoele may present with difficulty with breathing and poor feeding due to the intranasal nasolacrimal cyst component7.
Signs
The most important thing is to different epiphora from pseudoepiphora. The causes of psuedoepiphora include lid causes like entropion, ectropion, epiblepharon, trichiasis, conjunctival causes like allergy, conjunctivitis, and corneal causes like a foreign body. An important cause of pseudoepiphora is congenital glaucoma, which may simply present as a watering eye, so whenever there is doubt, the intraocular pressure should be checked and the optic disc is evaluated. A delay in the diagnosis of glaucoma could lead to loss of vision.
The child is evaluated under good illumination using a magnifying loupe. The 10 D lens of the direct ophthalmoscope or the 20 D lens of the indirect ophthalmoscope can be used. Some children may require an examination under sedation or general anesthesia.
The affected side shows an increased tear film height. There may be periocular dermatitis due to the constant watering. The sac may be swollen, resulting in a mass just below the medial canthal tendon. Mucoid or mucopurulent discharge can be expressed out of the puncta when the sac is compressed. This finding is commonly reported as ‘ROPLAS positive’ (Regurgitation on Pressure over the Lacrimal Sac). When a dacryocystocoele is formed, however, the sac does not release its contents on compression due to the kinking of the common canaliculus.
The location of the sac swelling always occurring below the medial canthal tendon is important to differentiate it from an encephalocele, another mass lesion that can occur in that region; which is found above the medial canthal tendon.
The stasis of the tears presents a warm and moist environment for the bacteria to proliferate, and this can lead to chronic or acute dacryocystitis. When there is acute dacryocystitis, the surrounding skin is warm and erythematous, and the swelling is tense and tender. The child is maybe febrile. Chronic dacryocystitis present as intermittent or consistent mucopurulent discharge, with crusting of the lids and matting of the eyelashes.
Investigations
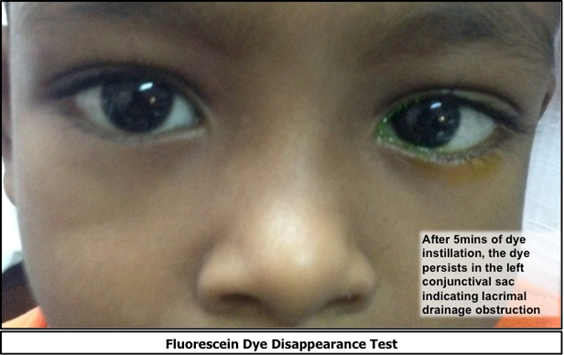
Fluorescein Dye Disappearance test:
It is a fairly sensitive (90%) and specific (100%) test in the evaluation of outflow obstruction in infants16. A drop of 2% Fluorescein solution is put in both the eyes and the excess dye is wiped off. After 5 minutes, the tear meniscus is observed for retained dye. In a normal test, the dye is not present after 5 minutes, while in an obstruction, the dye may get diluted but will remain at the end of 5 minutes.
Treatment
Most obstructions open spontaneously in the first 4-6 weeks of life and around 90% -96% of the babies with nasolacrimal duct obstruction, resolve spontaneously at 1 year of age.1,17
For this reason, conservative management is usually preferred until one year of age, following which surgical intervention can be undertaken if there is no resolution.
Given below is a simplified algorithm for the management of congenital nasolacrimal duct obstruction (CNLDO), based on preferred practices, although there is no clear-cut consensus on the timing of undertaking the various interventions18,19.
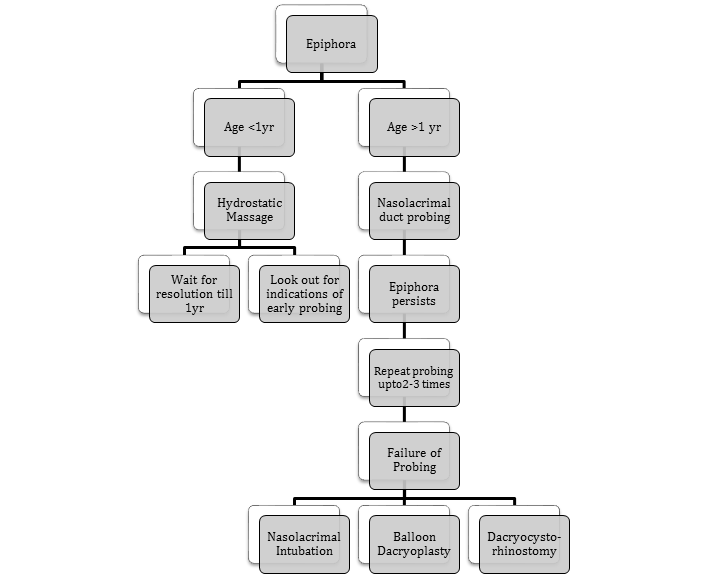
Hydrostatic Massage
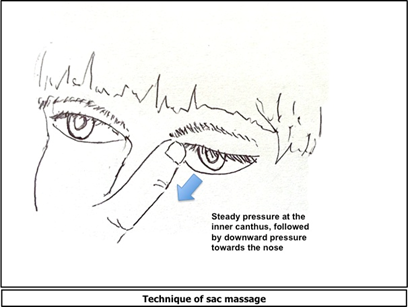
The simple method of hydrostatic sac massage has been found to be effective in the treatment of epiphora20. The massage is begun by pressure at the medial canthus, which is effective in blocking the common canaliculus thus preventing reflux backward through the canaliculi. Then pressure is applied downwards, thus increasing the hydrostatic pressure in the sac, and causing rupture of the membrane.21 The technique of massage is important- and has been studied comparing simple or improper techniques (with a success rate of only 21%) with the proper hydrostatic technique (with a success rate of 91%)22.
Instruction to be given to the parent:
The parent’s nail of the index or little finger that will be used for the massage must be clipped. The hands must be washed well. Vaseline or Baby oil may be applied to the finger as a lubricant23. The finger is then placed firmly as shown in the figure and pressure is then applied in the downward direction. 5-10 such strokes are to be done, 2-4 times a day22,24. The technique should be demonstrated to the parent and should be checked on consequent follow-ups.
Probing
Optimum age:
One school of thought waits for spontaneous resolution till 1 year of age, thus recommends probing at 1 year of age18,25. The logic is that the rate of resolution without surgical intervention is around 90% at 1 year. Thus no surgical intervention is taken up till 1 year of age unless there is a specific indication for early probing.
Indications for early probing include:
- Dacryocystocoele- As there are high chances of acute dacryocystitis. Also, it is often associated with an intranasal cyst which can cause breathing difficulties.
- Severe symptoms- Constant mucopurulent discharge and periocular dermatitis due to constant tearing causing infant discomfort.
- Acute dacryocystitis- Under an intravenous antibiotic cover, early probing is done to allow the tears to drain to reduce the risk of local spread with abscess formation and hematogenous spread and septicemia.
- Congenital cataract and glaucoma- Infants to be posted for ocular surgery having chronic dacryocystitis will require early probing followed by intraocular surgery once the infection clears.
The second school of thought recommends early probing in all infants at 4-6 months of age, to avoid unnecessary months of discomfort to the infant26. Also at 4-6 months, probing can be done as an office procedure under topical anesthesia, thus avoiding the general anesthesia, which will be required at 1 year of age. It has been proposed that the pain felt during the office probing procedure with general anesthesia is akin to the pain of vaccination.
Studies have found both the 4-6 month office probing as well as the hospital-based 1 year probing under general anesthesia to be equally effective, and thus the decision is up to the discretion of the ophthalmologist27.
Maximum age
While most studies agree that probing is very efficacious to 1 to 2 years of age, some studies have found that the rate of success is lower in older children28,29. This is explained by an increase in the fibrotic changes with increasing age. However others have proposed that decreased success in older children, might be due to a process of natural selection, wherein more and more of the membranous blocks get cured spontaneously, leaving behind a higher percentage of the firm or complex blocks28,29. Complex or firm blocks are those in which a firm resistance is encountered by the probe. Thus, the older children with a membranous block will respond well to probing. Success rates of 72%-80% have been reported in the 2-5 year age group28,29.
Thus, in conclusion, probing can be attempted in children until 4-5 years, with a backup plan of an alternative procedure if a complex block is encountered intraoperatively.
Technique
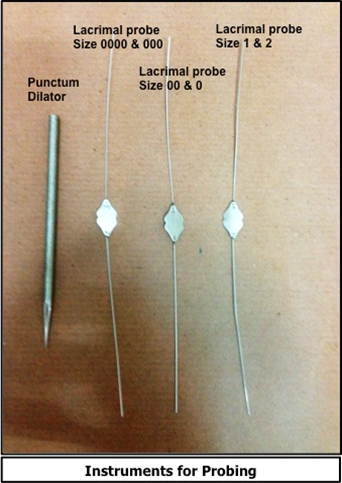
The appropriate anesthesia is given- topical for office-based and general anesthesia for hospital procedures. The punctum dilator may be used to dilate the punctum. The upper lid is preferred by many, as there is less of an acute turn when the probe enters the sac. The lacrimal probe of the appropriate size as per the age of the baby is taken and while laterally stretching the lid the punctum is entered. The stretch straightens out the vertical component of the canaliculus thus allowing for easy passage. Once the hard stop is encountered, the probe is rotated 90 degrees and directed downwards, outwards, and backward to enter the nasolacrimal duct so that the probe now lies vertically against the supraorbital margin. The confirmation that the probe is in the nasolacrimal duct is from the finding that the probe stands straight on its own, and there is a spring back movement of the probe on an attempt to displace it.
The probe is advanced till a give way sensation is encountered as the probe presumably pierces the membrane. The confirmation that the probe has entered the nose is by endoscopically viewing the probe in the inferior meatus, or by the metal-to-metal sensation of another instrument inserted from the nose. The inferior turbinate may additionally be in fractured if closely impacted against the NLD opening on endoscopy.
If one is not able to pass the probe or the probe is encountering too much resistance while passage, one can try with a probe of a smaller diameter and then progressively use probes of greater size to adequately dilate the nasolacrimal duct. Sac syringing with fluorescein dye may be done in the end to ensure patency. The throat pack must be in place before syringing for a child under general anesthesia to decrease the risk of aspiration.
The success of the probing:
The success of the probing is established when there is the resolution of signs and symptoms and the fluorescein dye test becomes normal. If symptoms persist, repeat probing can be done at 1-3 weeks after the first probing. Repeat probing can be attempted 2-3 times, however, repeat probing have lower rates of success.
Silicon Intubation
Indication:
Silicon tubing is flexible, soft, and knots well as this is a good material for lacrimal intubation. Failed probing is an indication of silicon intubation31. Some use intubation as a primary treatment, instead of probing32. Primary intubation has shown better results than probing in patients with bilateral block, Down’s syndrome, and previous history of acute dacryocystitis. Also when probing reveals a tight obstruction, intubation can be done in the same sitting to increase the chances of success.
Technique:
Lacrimal probing is first done with increasing probe diameter. The intubation probe with the silicon tubing attached to it is then inserted through the upper punctum and then retrieved from the nose. The lower punctum is then cannulated with the probe at the other end of the silicon tubing and similarly retrieved from the nostril. The tubing is then tied with multiple knots, the end is trimmed and fixed to the lateral nasal wall with a nonabsorbable suture.
Monocanalicular intubation is also being done and is found to be as efficacious as canalicular intubation by some studies33
The rate of success as found by several different studies is around 66-100%31,32,34,35, with a higher rate of success than probing, in patients with complex nasolacrimal obstructions.
Removal of the tubing:
The tubing is removed anytime between 6 weeks to 6 months after intubation, and there is no real consensus on the ideal time of removal36,37.
Balloon Dacryoplasty
Indication:
Failed probing is an indication of balloon dacryoplasty38. The results of balloon dacryoplasty and intubation after failed probing are comparable34. It can also be used as a primary modality of treatment, instead of probing38.
Technique:
Initially probing is done which is followed by cannulation of the nasolacrimal duct via the upper canaliculus with the balloon catheter. Once it is confirmed that the catheter is in the right place, so that the balloon is at the level of the valve of Hasner, the balloon is inflated so as to produce a pressure of around 8 atmospheres of pressure. This pressure is maintained for about 90 seconds and then deflated. After a few seconds, the balloon is elevated again to 8 atmospheres and maintained for 60 seconds.
This double cycle is then repeated with the balloon placed at the lower part of the nasolacrimal duct. The balloon catheter is then deflated and then removed.
Reported success rates are 53-95%34,38,39.
Pediatric Dacryocystorhinostomy
Indications:
A dacryocystorhinostomy is usually done when other procedures have failed. It is especially useful when the block is firm or in craniofacial anomalies, where other procedures are likely to fail.
Technique:
The principle involves making an opening in the bones forming the lacrimal fossa. Then sac and nasal mucosal flaps are made and sutured together. Now the tears and the sac secretions empty into the middle meatus of the nose, bypassing the nasolacrimal duct altogether. The approach can be an external one, where there is an external skin incision or endonasal, which is done using an endoscope through the nose.
The success rate for external and endonasal DCR surgery is high, around 83-97%40,41,42 and 87-93% respectively43,44.
Medical Management
When there is accompanying chronic dacryocystitis with mucopurulent discharge, an antibiotic eye drop like Tobramycin 0.3% can be instilled in the eye 3-4 times a day. The parents are advised to clean away the discharge to prevent matting and crusting over the lashes and are taught the hydrostatic massage.
In cases developing the rare complication of acute dacryocystitis, hospital admission is usually required. Intravenous antibiotics are required and the infant is taken up for nasolacrimal duct probing under general anesthesia.
Complications
Acute Dacryocystitis, Preseptal and Orbital Cellulitis:
Acute dacryocystitis occurs due to stasis of tears in the sac and is an uncommon complication of CNLDO occurring in around 2-3% cases9. This infection may spread locally, especially in infants in whom the immune system is not mature. This can cause a preseptal, and can even cause orbital cellulitis, although this is a rare complication.
There is a higher incidence of acute dacryocystitis in congenital dacryocoele reported to be around 33%-65%10,11.
Lacrimal Fistula:
Lacrimal fistula is a rare congenital malformation of the lacrimal system, which may occur with or without nasolacrimal duct obstruction12. Rarely, a lacrimal fistula may arise a complication of acute dacryocystitis, wherein the abscess breaks open onto the surface.
Respiratory Distress:
Congenital dacryocoele with an intranasal cyst, especially if bilateral gives rise to respiratory distress and abnormal feeding, as neonates are obligate nose breathers13.
Anisometropic Amblyopia:
Babies with congenital nasolacrimal duct obstruction have been found to have a greater incidence of anisometropia than the general population. The incidence of anisometropia has been found to be 8.6 to 10% in such babies and found to be higher in unilateral than bilateral CNLDO14,15. Regular refraction to prevent amblyopia is necessary.
1. Congenital Nasolacrimal. In: Basic and clinical science course (BCSC) Section 7: Orbit, Eyelids, and Lacrimal System. San Francisco, CA: American Academy of Ophthalmology; 2012:250.
2. Paul TO, Shepherd R. Congenital nasolacrimal duct obstruction: natural history and the timing of optimal intervention. J PediatrOphthalmol Strabismus. 1994 Nov-Dec;31(6):362-7.
3. Duke-Elder S, Cook C. System of Ophthalmology. In: Duke-Elder S, editor.Normal and abnormal development Part 1: Embryology. Vol. 3. St Louis: CV Mosby; 1963. pp. 241–245.
4. Lorena SH, Silva JA, Scarpi MJ. Congenital nasolacrimal duct obstruction in premature children. J PediatrOphthalmol Strabismus. 2013 Jul-Aug;50(4):239-44.
5. MacEwen CJ, Young JD. Epiphora during the first year of life. Eye (Lond). 1991;5 ( Pt 5):596-600.
6. Kanski J, Bowling B. Clinical Ophthalmology: A systematic approach. 6th Ed. China: Elsevier; 2011.
7. Lueder GT. The association of neonatal dacryocystoceles and infantile dacryocystitis with nasolacrimal duct cysts (an American Ophthalmological Society thesis). Trans Am Ophthalmol Soc. 2012 Dec;110:74-93.
9. Lueder GT. The Association of Neonatal Dacryocystoceles and Infantile Dacryocystitis with Nasolacrimal Duct Cysts (An American Ophthalmological Society Thesis). Transactions of the American Ophthalmological Society2012;110:74-93.
10. Shekunov J, Griepentrog GJ, Diehl NN, Mohney BG. Prevalence and clinical characteristics of congenital dacryocystocele. J AAPOS. 2010 Oct;14(5):417-20.
11. Wong RK, VanderVeen DK. Presentation and management of congenital dacryocystocele. Pediatrics. 2008 Nov;122(5):e1108-12.
12. Welham RA, Bergin DJ. Congenital lacrimal fistulas. Arch Ophthalmol. 1985 Apr;103(4):545-8.
13. Mazzara CA, Respler DS, Jahn AF. Neonatal respiratory distress: sequela of bilateral nasolacrimal duct obstruction. Int J PediatrOtorhinolaryngol. 1993 Jan;25(1-3):209-16.
14. Pyi Son MK, Hodge DO, Mohney BG. Timing of congenital dacryostenosis resolution and the development of anisometropia. Br J Ophthalmol. 2014 Aug;98(8):1112-5.
15. Kipp MA, Kipp MA Jr, Struthers W. Anisometropia and amblyopia in nasolacrimal duct obstruction. J AAPOS. 2013 Jun;17(3):235-8.
16. MacEwen CJ, Young JD. The fluorescein disappearance test (FDT): an evaluation of its use in infants. J PediatrOphthalmol Strabismus. 1991 Nov-Dec;28(6):302-5.
17. MacEwen CJ, Young JD. Epiphora during the first year of life. Eye (Lond). 1991;5 ( Pt 5):596-600.
18. Dotan G, Nelson LB. Congenital Nasolacrimal Duct Obstruction: Common Management Policies Among Pediatric Ophthalmologists. J PediatrOphthalmol Strabismus. 2014 Nov 4:1-6.
19. Schellini SA, Ariki CT, Sousa RL, Weil D, Padovani CR. Management of congenital nasolacrimal duct obstruction--latinamerican study. OphthalPlastReconstr Surg. 2013 Sep-Oct;29(5):389-92.
20. CRIGLER LW. THE TREATMENT OF CONGENITAL DACRYOCYSTITIS. JAMA.1923;81(1):23-24.
21. Kushner BJ. Congenital nasolacrimal system obstruction. Arch Ophthalmol. 1982 Apr;100(4):597-600.
22. Shivpuri D, Puri A. Congenital nasolacrimal duct obstruction: the proper technique of massage. Indian Pediatr. 1994 Mar;31(3):337-40.
23. Pawar P, Patil A, Patel M, Shah S. Use of Oil as a Lubricant makes Lacrimal Sac Massage Efficient in Congenital NasoLacrimal Duct Obstruction (CNLDO). MVP Journal of Medical Sciences. 2014 Jan .Volume 1 Issue 1.
24. Murthy R. Congenital Nasolacrimal Duct Obstruction.Kerela Journal of Ophthalmology. 2007 Jun;19(2):191-196.
25. Takahashi Y, Kakizaki H, Chan WO, Selva D. Management of congenital nasolacrimal duct obstruction. ActaOphthalmol. 2010 Aug;88(5):506-13.
26. Miller AM, Chandler DL, Repka MX, Hoover DL, Lee KA, Melia M, Rychwalski PJ, Silbert DI; Pediatric Eye Disease Investigator Group, Beck RW, Crouch ER 3rd, Donahue S, Holmes JM, Quinn GE, Sala NA, Schloff S, Wallace DK, Foster NC, Frick KD, Golden RP, Lambert SR, Tien DR, Weakley DR Jr. Office probing for treatment of nasolacrimal duct obstruction in infants. J AAPOS. 2014 Feb;18(1):26-30.
27. Schnall BM. Pediatric nasolacrimal duct obstruction. CurrOpinOphthalmol. 2013 Sep;24(5):421-4.
28. Kashkouli MB, Beigi B, Parvaresh MM, Kassaee A, Tabatabaee Z. Late and very late initial probing for congenital nasolacrimal duct obstruction: what is the cause of failure? The British Journal of Ophthalmology 2003;87(9):1151-1153.
29. Honavar SG, Prakash VE, Rao GN. Outcome of probing for congenital nasolacrimal duct obstruction in older children. Am J Ophthalmol. 2000 Jul;130(1):42-8.
30. Paul TO, Shepherd R. Congenital nasolacrimal duct obstruction: natural history and the timing of optimal intervention. J PediatrOphthalmol Strabismus. 1994 Nov-Dec;31(6):362-7.
31. Lim CS, Martin F, Beckenham T, Cumming RG. Nasolacrimal duct obstruction in children: outcome of intubation. J AAPOS. 2004 Oct;8(5):466-72.
32. Memon MN, Siddiqui SN, Arshad M, Altaf S. Nasolacrimal duct obstruction in children: outcome of primary intubation. J Pak Med Assoc. 2012 Dec;62(12):1329-32.
33. Komínek P, Cervenka S, Pniak T, Zeleník K, Tomášková H, Matoušek P. Monocanalicular versus bicanalicular intubation in the treatment of congenital nasolacrimal duct obstruction. Graefes Arch ClinExpOphthalmol. 2011 Nov;249(11):1729-33.
34. Repka MX, Chandler DL, Holmes JM, Hoover DL, Morse CL, Schloff S, Silbert DI, Tien DR; Pediatric Eye Disease Investigator Group. Balloon catheter dilation and nasolacrimal duct intubation for treatment of nasolacrimal duct obstruction after failed probing.ArchOphthalmol. 2009 May;127(5):633-9.
35. Leone CR Jr, Van Gemert JV. The success rate of silicone intubation in congenital lacrimal obstruction. Ophthalmic Surg. 1990 Feb;21(2):90-2.
36. Welsh MG, Katowitz JA. Timing of Silastic tubing removal after intubation for congenital nasolacrimal duct obstruction. OphthalPlastReconstr Surg. 1989;5(1):43-8.
37. Migliori ME, PuttermanAM. Silicone intubation for the treatment of congenital lacrimal duct obstruction: successful results removing the tubes after six weeks. Ophthalmology. 1988 Jun;95(6):792-5.
38. Yüksel D, Ceylan K, Erden O, Kiliç R, Duman S. Balloon dilatation for treatment of congenital nasolacrimal duct obstruction. Eur J Ophthalmol. 2005 Mar-Apr;15(2):179-85.
39. Tao S, Meyer DR, Simon JW, Zobal-Ratner J. Success of balloon catheter dilatation as a primary or secondary procedure for congenital nasolacrimal duct obstruction. Ophthalmology. 2002 Nov;109(11):2108-11.
40. Nowinski TS, Flanagan JC, Mauriello J. Pediatric dacryocystorhinostomy. Arch Ophthalmol. 1985 Aug;103(8):1226-8.
41. Barnes EA, Abou-Rayyah Y, Rose GE. Pediatric dacryocystorhinostomy for nasolacrimal duct obstruction. Ophthalmology. 2001 Sep;108(9):1562-4.
42. Shrestha R, Sobti D, Chi SL, Saiju R, Richard M. Surgical outcome of pediatric dacryocystorhinostomy in Nepal. J AAPOS. 2014 Aug;18(4):368-9.
43. Komínek P, Cervenka S, Matousek P, Pniak T, Zeleník K. Primary pediatric endonasaldacryocystorhinostomy--a review of 58 procedures. Int J PediatrOtorhinolaryngol. 2010 Jun;74(6):661-4.
44. Celenk F, Mumbuc S, Durucu C, Karatas ZA, Aytaç I, Baysal E, Kanlikama M. Pediatric endonasal endoscopic dacryocystorhinostomy. Int J PediatrOtorhinolaryngol. 2013 Aug;77(8):1259-62.