Algorithm Of Screening And Management11,19, 23, 24
Screening
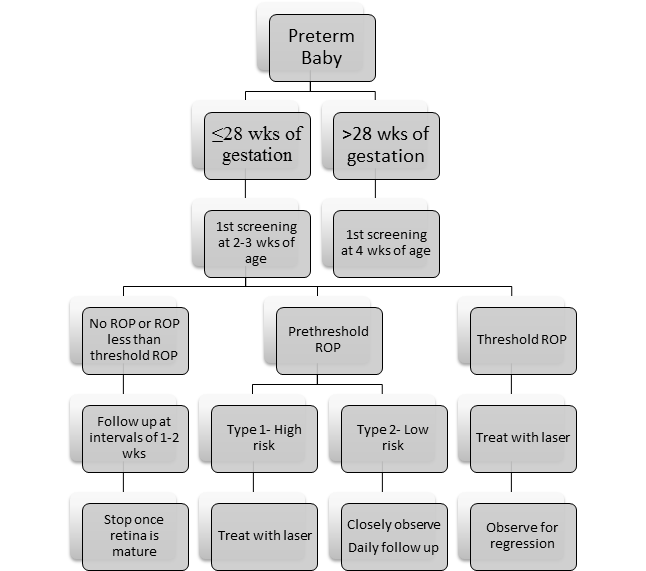
The three important questions are whom to screen when to screen and till when to continue screening.
Whom to screen:
According to the Indian guidelines set forth by the National Neonatology Forum (NNF) Guidelines24
- All Preterms babies born at <34 wks gestational age
- All preterm babies with birth weight <1750 Kgs
- All preterm babies born at 34-36 wks gestational age or 1750-2000 Kgs at birth if:-
- Stormy neonatal course
- Prolonged oxygen delivered
- Oxygen fluctuations
- Sepsis
- Any major surgery
- Severe anemia
That is any preterm baby whom the neonatologist feels is at risk for retinopathy of prematurity.
- All babies whose gestational age is not known.
The last point is especially important in our country as on several occasions one has only a rough estimate from ultrasonographic age.
In the United States, only the preterm babies less than 30 weeks by age or less than 1500 kgs are screened.23 However the Indian guidelines are different from the American Academy of Ophthalmology guidelines as it is found that in developing countries bigger and older babies are getting the disease, and therefore each country has been urged to make their own guidelines.
When to screen?
According to NNF guidelines24:-
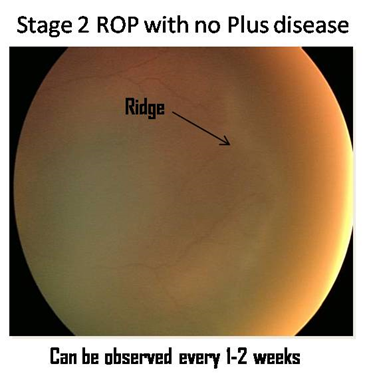
Follow examinations depend upon the fundus findings. An easy algorithm to follow is as given below11:-
| No ROP |
Every two weeks |
| ROP |
Weekly |
| Prethreshold ROP |
Daily |
When to stop screening?
Screening can be stopped when:-
- The retina is completely vascularised. This usually happens at 40-45 weeks of postmenstrual age (Gestational + Chronological Age)24.
- The disease has completely regressed24.
Role of Retcam and Telemedicine
The Netcam or retinal camera is a wide-angle (130°) digital camera that can be operated by technicians to take good quality fundus images. The Netcam can be used for professional record keeping. More importantly, is the role in telemedicine wherein a technician in the peripheral ICUs can image the fundus and transfer the images to the experts at the tertiary care centers. Stanford University Network for Diagnosis of Retinopathy of Prematurity (SUNDROP) telemedicine initiative found that Telemedicine had 100% sensitivity, 99.8% specificity, 93.7% positive predictive value, and 100% negative predictive value for detection of treatment warranted ROP25. Similarly, the telemedicine ROP model developed in Karnataka called the KIDROP model has shown promise in delivering services of the specialists in underserved areas26.
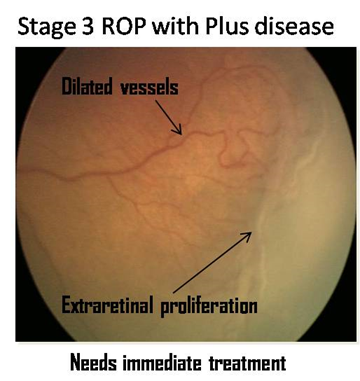
Treatment of threshold & prethreshold ROP
The principle of treatment is to ablate the hypoxic retina either with laser ablation or with cryotherapy. Thus the hypoxic retina is converted to the anoxic retina, which no longer produces VEGF, thus ending the tendency for vascular proliferation.
While the CRYO ROP trial treated all babies with threshold ROP, the ETROP trial found that treating babies with high-risk pre threshold ROP also has benefits.
Laser Ablation
It is the mainstay of treatment at present27. The structural and functional visual outcomes obtained with laser are superior to those with cryotherapy28,29.
Diode red laser (810nm) delivered by laser indirect ophthalmoscopy is used. The procedure is done under topical anesthesia and the neonate is well fed and burped 30-60 minutes prior. The parameters used are 250 mW power, 150 ms duration with a repeat mode interval of 300 ms.27 The avascular retina is lasered to produce ‘greyish’ burns which are confluent. The power may be varied to ensure the right degree of burn, with lower energy for the anterior and superior retina. Excessive energy to the retina can result in exudative retinal detachment. No skip areas are left.
In a single session, around 3000-4000 burns are required to cover the entire avascular region in the zone I disease or APROP. For zone II threshold or pre threshold disease and non APROP eyes, around 1000-2000 spots are usually required.27
There are many advantages of laser over cryotherapy as shown in the table11,28,29.
| |
Cryotherapy |
Laser Ablation |
| Anaesthesia |
General Anaesthesia |
Can be done safely under Topical Anaesthesia |
| Bradycardia |
Higher chance |
Lower chance |
| Post procedure discomfort |
More |
Less |
| Lid edema and chemosis |
More |
Less |
| Post procedure Exudative Retinal Detachment |
Higher chance |
Lower chance |
| Ability to treat posterior disease |
No |
Yes |
| Can perform through small pupil |
Yes |
No |
| Can perform through hazy media |
Yes |
No |
Cryotherapy
The Cryotherapy for Retinopathy of Prematurity (CRYOROP) trial showed the usefulness of cryotherapy in treating infants with threshold disease30. When the laser indirect ophthalmoscopy became available, it soon gained acceptance over cryotherapy. This is because cryotherapy is more painful and requires general anesthesia. Also zone I & II are difficult to reach with the cryoprobe. However, cryotherapy still has a role to play as it is cheaper, can be done in non dilating pupils, hazy media, and where laser delivery systems are not still available11.
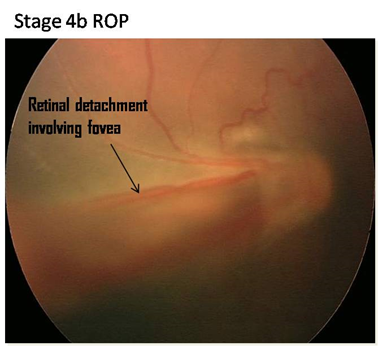
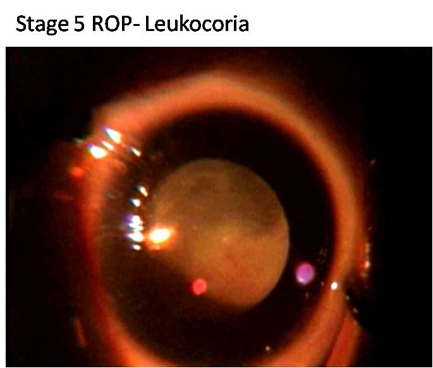
Management of advanced disease
Stage 5 of ROP and most often stage 4 also, require surgical management, in order to preserve some visual function. The options for retinal detachment surgery include scleral buckling and pars plicata vitrectomy which could be lens sparing or with lensectomy.
A simplified algorithm for management of advanced disease11,31,
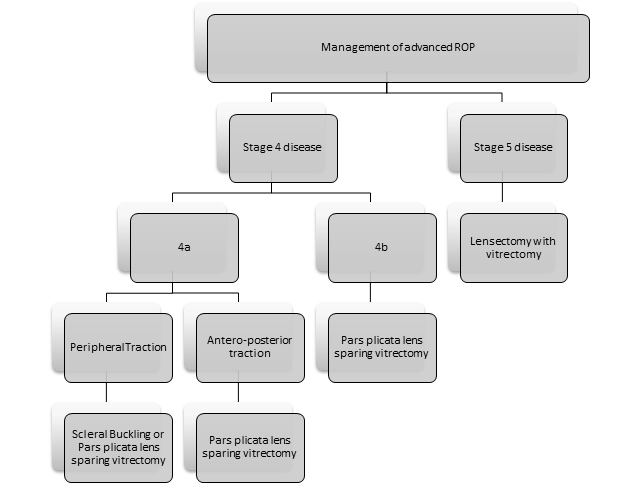
Scleral buckling
It works on the principle that external indentation on the sclera, relieves the traction, thereby allowing the retina to reattach. Although scleral buckling has the advantages of being an external surgery, the problems are induced myopia and thereby amblyopia due to the buckle and the necessity for secondary surgery to remove the buckle. Also, anatomical success rates are better vitrectomy, thus vitrectomy is generally considered superior to buckling32. However scleral buckling still has a role to play in stage 4a disease with traction anterior to the equator31.
Vitrectomy
In subtotal detachment involving the macula and in total retinal detachments, closed globe vitrectomy is performed. 3 ports are made through the pars plicata, a vitrectomy is performed, and careful dissection is performed to release the sites of traction. This is followed by an air-fluid exchange with the placement of an air bubble, or a viscoelastic agent11.
If there is no retroarticular space due to extensive detachment, a lensectomy is done prior to the vitrectomy to allow more anterior dissection31.
In Stage 4 disease, even though retina gets reattached in an acceptable number of neonates (80-90%), the functional outcome has been poor31. In stage 5 disease, however, results have been poor both anatomically as well as surgically33. Recently adjuncts to vitrectomy surgery, like intravitreal triamcinolone34, plasmin35, and bevacizumab36 have shown improved results.
Role of Bevacizumab
Bevacizumab is a humanized anti-VEGF monoclonal antibody, which has been given intravitreal as monotherapy as well as an adjunct36. However there is still a lot of uncertainty surrounding its use, and currently, it is being used only in selected cases of ROP for example infants with small pupils and dense vitreous hemorrhage.
Complications
- Refractive errors- Myopia is a common sequel of regressed ROP in treated and untreated eyes. In one study around70% of high-risk pre threshold ROP eyes were myopic in early childhood, and the proportion with high myopia increased steadily between ages 6 months and 3 years38. Therefore, the child should be closely monitored and the refractive error should be corrected to prevent amblyopia.
- Strabismus39
- Cataract11
- Amblyopia39
- Anterior segment abnormalities- ectropion uveae, iris neovascularization, rigid pupil11
Role Of The Pediatrician
Various risk factors have been found to cause Retinopathy of Prematurity (ROP), of which the most important after gestational age and birth weight is the arterial oxygen saturation.
Unrestricted and unmonitored oxygen delivery to the preterm, which has been proven by meta-analysis to increase the incidence as well as the severity of ROP, should be avoided40. However, lower target ranges have been found to be associated with increased mortality, and an ideal range of arterial oxygen saturation, which prevents ROP without affecting mortality, is still to be established41.
Referral of a preterm baby for ophthalmic examination as per the guidelines of screening is a must and failure to do so can result in blindness.
Screening is to be done for24:-
- All Preterms babies born at <34 wks gestational age
All preterm babies with birth weight <1750 Kgs
All preterm babies born at 34-36 wks gestational age or 1750-2000 Kgs at birth if
:-
- Stormy neonatal course
- Prolonged oxygen delivered
- Oxygen fluctuations
- Sepsis
- Any major surgery
- Severe anemia
4. All babies whose gestational age is not known.
The first screening of the neonate is to be done at 4 weeks after delivery, earlier (2-3 weeks) if the child is born at <28 weeks or is <1200 g24.
Preterm babies who are large for gestational age, such as those born to diabetic mothers, & babies with hydrocephalus, intestinal obstruction, and renal disease, etc. can also develop severe ROP. Similarly, preterm babies born after assisted fertilization techniques, irrespective of birth weight can develop ROP. Babies who have not been given oxygen can also develop ROP.
ROP is a potentially blinding disease and is an important cause of blindness as the neonate at risk has a lifetime ahead of him. Furthermore, the risk of blindness can be substantially reduced by timely diagnosis and treatment. Thus teamwork involving the neonatologist, ophthalmologist, nursing staff, and parents is necessary for bringing the epidemic of ROP under control.
The LV Prasad Eye Institute in Hyderabad has developed an efficient NICU based screening model of ROP, with a Day 30 strategy in which all neonates at risk are screened directly in the NICU by 30th day of age42 and babies screened under this protocol had the better outcomes43.
Every neonatal intensive care unit and neonatologist should have organized the logistics of a good screening protocol so that no baby goes unscreened.
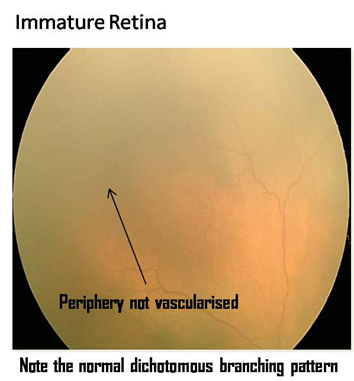
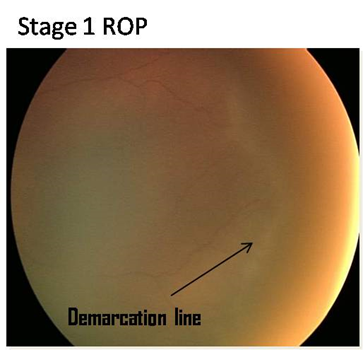
Photographs
The photographs have been provided by Dr. Subhadra Jalali, Associate Director, Consultant - Srimati Kannuri Santhamma center for vitreoretinal diseases, and Jasti V Ramanamma Children’s Eye Care Centre, L.V.Prasad Eye Institute, Kallam Anji Reddy Campus, Hyderabad. She has received the Sight Savers grant (2003-2005) for setting up treatment and training centers for the control of childhood blindness in the field of ROP and is on the Ministry of Health National task force on ROP. Her work in ROP has made a tremendous difference in preventing blindness in the twin cities of Hyderabad and Secunderabad as well as establishing a screening protocol that is worthy of emulation all over India and other developing countries.
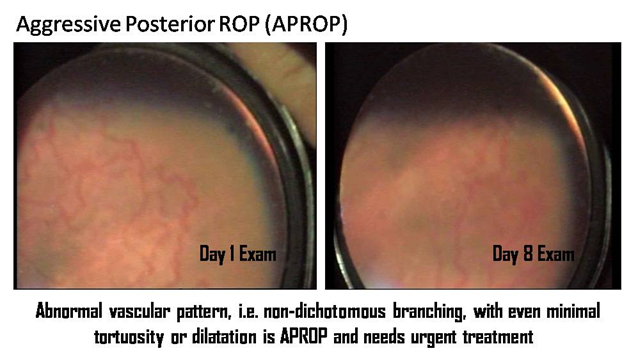
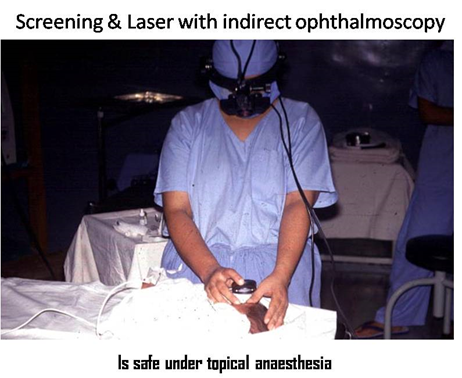
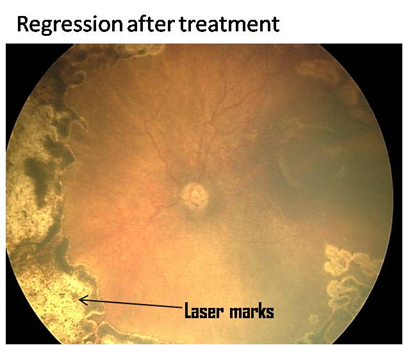
1. Patel DK, Tajunisah I, Gilbert C, Subrayan V. Childhood blindness and severe visual impairment in Malaysia: a nationwide study. Eye (Lond). 2011 Apr;25(4):436-42.
2. Gilbert C. Retinopathy of prematurity: a global perspective of the epidemics, population of babies at risk and implications for control. Early Hum Dev. 2008 Feb;84(2):77-82.
3. Hack M, Fanaroff AA. Outcomes of children of extremely low birthweight and gestational age in the 1990s. SeminNeonatol. 2000 May;5(2):89-106.
4. Valentine PH, Jackson JC, Kalina RE, Woodrum DE. Increased survival of low birth weight infants: impact on the incidence of retinopathy of prematurity. Pediatrics. 1989 Sep;84(3):442-5.
5. Gopal L, Sharma T, Ramchandran S, Shanmugasundaram R, Asha V. Retinopathy of prematurity. A study. Indian J Ophthalmol 1995;43:50-61
6. Charan R, Dogra MR, Gupta A, Narang A. The incidence of retinopathy of prematurity in a neonatal care unit. Indian J Ophthalmol 1995;43:123-26.
7. Varughese S, Jain S, Gupta N, Singh S, Tyagi V, Puliyel JM. Magnitude of the problem of retinopathy of permaturity. Experience in a large maternity unit with a medium size level-3 nursery. Indian J Ophthalmol 2001:49:187-88.
8. Gilbert C. Changing challenges in the control of blindness in children. Eye (Lond). 2007 Oct;21(10):1338-43.
9. Rashtriya Bal Swasthya Karyakram, Operation guidelines. Ministry of Health and Family Welfare, India Government.http://nrhm.gov.in/images/pdf/programmes/RBSK/Operational_Guidelines/Operational%20Guidelines_RBSK.pdf
10. The India Newborn Action Plan. Ministry of Health and Family Welfare, India Government, 2014. http://www.newbornwhocc.org/INAP_Final.pdf
11. Chakrabarti M, Stephen V,Rani S, Chakrabarti A. Retinopathy of Prematurity: Screening and Management. Kerala Journal of Ophthalmology. 2007 Dec: 429-38.
12. RETROLENTAL fibroplasia. Br Med J. 1953 Sep 12;2(4836):610-1.
13. Kanski J, Bowling B. Clinical Ophthalmology: A systematic approach. 6th Ed. China: Elsevier; 2011.
14. A. Patz, L.E. Hoeck, E. DeLaCruz, Studies on the effect of high oxygen administration in retrolental fibroplasia: I. Nursery observations, Am. J. Ophthalmol. 35 (1952) 1248–1252.
15. Kretzer FL, Hittner HM. Human retinal development: relationship to the pathogenesis of retinopathy of prematurity. In: McPherson AR, Hittner HM, Kretzer FL, eds. Retinopathy of prematurity: current concepts and controversies. Toronto: Decker, 1986: 27-52.
16. Smith LE. Pathogenesis of retinopathy of prematurity. Growth Horm IGF Res. 2004 Jun;14Suppl A:S140-4.
17. Kim, T.-i., Sohn, J., Pi, S.-y. and Yoon, Y. H. (2004), Postnatal risk factors of retinopathy of prematurity. Paediatric and Perinatal Epidemiology, 18: 130–134.
18. Holmstrom G, Broberger U, Thomassen P. Neonatal risk factors for retinopathy of prematurity –a population-based study. ActaOphthalmologicaScandinavica. 2003:204-07.
19. Jalali S, Anand R, Kumar H, Dogra MR, Azad R, Gopal L. Programme planning and screening strategy in retinopathy of prematurity. Indian J Ophthalmol 2003;51:89-97.
20. International Committee for the Classification of Retinopathy of Prematurity. The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol. 2005 Jul;123(7):991-9.
21. Multicenter trial of cryotherapy for retinopathy of prematurity. Preliminary results. Cryotherapy for Retinopathy of Prematurity Cooperative Group. Arch Ophthalmol. 1988 Apr;106(4):471-9.
22. Good WV; Early Treatment for Retinopathy of Prematurity Cooperative Group. Final results of the Early Treatment for Retinopathy of Prematurity (ETROP) randomized trial. Trans Am Ophthalmol Soc. 2004;102:233-48; discussion 248-50.
23. Retinopathy of Prematurity. In: Basic and clinical science course (BCSC) Section 6: Pediatric Ophthalmology and Strabismus. San Francisco, CA: American Academy of Ophthalmology; 2012:280-7.
24. Pejaver RK, Vinekar A, Bilagi A. National Neonatology Foundation's Evidence Based Clinical Practice Guidelines for Retinopathy of Prematurity, NNF India, Guidelines, 2010. p. 253-62.
25. Fijalkowski N, Zheng LL, Henderson MT, Wallenstein MB, Leng T, Moshfeghi DM. Stanford University Network for Diagnosis of Retinopathy of Prematurity (SUNDROP): four-years of screening with telemedicine. Curr Eye Res. 2013 Feb;38(2):283-91.
26. Vinekar A, Gilbert C, Dogra M, Kurian M, Shainesh G, Shetty B, Bauer N. The KIDROP model of combining strategies for providing retinopathy of prematurity screening in underserved areas in India using wide-field imaging, tele-medicine, non-physician graders and smart phone reporting. Indian J Ophthalmol. 2014 Jan;62(1):41-9.
27. Jalali S, Azad R, Trehan HS, Dogra MR, Gopal L, Narendran V. Technical aspects of laser treatment for acute retinopathy of prematurity under topical anesthesia. Indian J Ophthalmol. 2010 Nov-Dec;58(6):509-15.
28. Ng EY, Connolly BP, McNamara JA et al. A comparison of laser photocoagulation with cryotherapy for threshold retinopathy of prematurity at 10 years: part 1. Visual function and structural outcome. Ophthalmology 2002;109(5):928-934.
29. Connolly BP, Ng EY, McNamara JA et al. A comparison of laser photocoagulation with cryotherapy for threshold retinopathy of prematurity at 10 years: part 2. Refractive outcome. Ophthalmology 2002;109(5):936-941.
30. Palmer EA, Hardy RJ, Dobson V, Phelps DL, Quinn GE, Summers CG, Krom CP, Tung B; Cryotherapy for Retinopathy of Prematurity Cooperative Group. 15-year outcomes following threshold retinopathy of prematurity: final results from the multicenter trial of cryotherapy for retinopathy of prematurity. Arch Ophthalmol. 2005 Mar;123(3):311-8.
31. Roohipoor R, Karkhaneh R, Riazi-Esfahani M, Ghasemi F, Nili-Ahmadabadi M. Surgical management in advanced stages of retinopathy of prematurity; our experience. J Ophthalmic Vis Res. 2009 Jul;4(3):185-90. PubMed PMID: 23198072; PubMed Central PMCID: PMC3498568.
32. Hartnett ME, Maguluri S, Thompson HW, McColm JR. Comparison of retinal outcomes after scleral buckle or lens-sparing vitrectomy for stage 4 retinopathy of prematurity. Retina.2004;24(5):753–7.
33. Hubbard GB 3rd. Surgical management of retinopathy of prematurity. CurrOpinOphthalmol. 2008 Sep;19(5):384-90.
34. Lakhanpal RR, Fortun JA, Chan-Kai B, Holz ER. Lensectomy and vitrectomy with and without intravitreal triamcinolone acetonide for vascularly active stage 5 retinal detachments in retinopathy of prematurity. Retina. 2006;26(7):736–40.
35. Tsukahara Y, Honda S, Imai H, et al. Autologous plasmin-assisted vitrectomy for stage 5 retinopathy of prematurity: a preliminary trial. Am J Ophthalmol. 2007;144(1):139–41.
36. Law JC, Recchia FM, Morrison DG, Donahue SP, Estes RL. Intravitrealbevacizumab as adjunctive treatment for retinopathy of prematurity. J AAPOS. 2010 Feb;14(1):6-10.
37. Geloneck MM, Chuang AZ, Clark WL, Hunt MG, Norman AA, Packwood EA, Tawansy KA, Mintz-Hittner HA; for the BEAT-ROP Cooperative Group. Refractive Outcomes Following BevacizumabMonotherapy Compared With Conventional Laser Treatment: A Randomized Clinical Trial.JAMAOphthalmol. 2014 Aug 7.
38. Quinn GE, Dobson V, Davitt BV, Hardy RJ, Tung B, Pedroza C, Good WV; Early Treatment for Retinopathy of Prematurity Cooperative Group. Progression of myopia and high myopia in the early treatment for retinopathy of prematurity study: findings to 3 years of age.
39. Kushner BJ. Strabismus and amblyopia associated with regressed retinopathy of prematurity.ArchOphthalmol. 1982 Feb;100(2):256-61.
40. Askie LM, Henderson-Smart DJ, Ko H. Restricted versus liberal oxygen exposure for preventing morbidity and mortality in preterm or low birth weight infants. Cochrane Database Syst Rev. 2009 Jan 21;(1):CD001077.
41. Askie LM. Optimal oxygen saturations in preterm infants: a moving target. Curr Opin Pediatr. 2013 Apr;25(2):188-92.
42. Jalali S. Lecture ROP Dos & Dont's. LV Prasad Eye Institute.http://www.lvpei.org/rop/videos/video3.html
43. Jalali S, Anand R, Rani PK, Balakrishnan D. Impact of the day-30 screening strategy on the disease presentation and outcome of retinopathy of prematurity. The Indian twin cities retinopathy of prematurity report number 3. Indian J Ophthalmol. 2014 May;62(5):610-4.