Himali Meshram.
Pediatric Unit, Apollo Clinics, Nagpur, Maharashtra, India.
ADDRESS FOR CORRESPONDENCE
Dr Himali Meshram , 401, Shivam Apartment, Manish Nagar, Nagpur, 440015, Maharashtra, India.
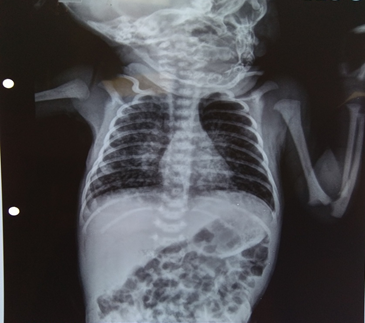
Email: gshimali@yahoo.com | | Keywords | | Congenital, tuberculosis, drug-resistant. | | | | Abstract | | Tuberculosis (TB) is a major health problem in our country. Though TB is highly prevalent in women of childbearing age, only few cases of congenital tuberculosis (TB) have been reported. We present a 21-day old child with congenital drug-resistant (DR) TB who was detected to have rifampicin resistance (RR) mycobacterium tuberculosis (MTB) on GeneXpert. Subsequently on screening the mother, she was detected to have pulmonary TB with sputum showing RR-MTB on GeneXpert. | | | | Introduction | | Tuberculosis (TB) is relatively common in pregnant women; the prevalence of active TB in pregnant and postpartum women from high burden countries is upper to 60 cases per 100,000 population per year and from low burden TB countries, the prevalence is lower to 20 cases per 100,000 population per year.1 Congenital TB is defined as TB occurring in infants as a result of maternal TB when the illness involves the genital track or the placenta.2 Less than 300 cases of congenital TB have been reported till now. Very few neonates with drug-resistant (DR) congenital TB have been reported. Espiritu et al reported a case of multidrug-resistant (MDR) tuberculosis (TB) in a Peruvian infant.3 Kulhari et al reported a 46-day old male infant with congenitally acquired MDR-TB.4 We present a 21-day old child with congenital DR-TB who was detected to have rifampicin resistance (RR) on GeneXpert. Unfortunately the child succumbed to his disease. Mother was subsequently detected to have pulmonary TB with sputum showing RR mycobacterium tuberculosis (MTB) on GeneXpert suggesting perinatal acquisition. | | | | Case Report | A 21 day old male neonate first by birth order presented with fever, lethargy and poor feeding for 12 days. He was born to a 24 year old mother at term by normal vaginal delivery and had a birth weight of 2.2 kg. Mother had no illness in pregnancy. Child was started on breast-feeds. There was no history of TB in the family. On presentation, he was lethargic and weighed 2.3 kg. There was mild respiratory distress with no cyanosis or jaundice. Chest had bilateral crepts with normal heart sounds. He had hepatomegaly. Other systems were normal. Routine hematological investigations were normal with reactive CRP (81 mg/dl). Liver functions showed raised liver enzymes (AST=528 IU/L; ALT=347 IU/L) with normal bilirubin levels. Ultrasound of abdomen showed mild hepatomegaly. Initial chest radiograph showed bilateral miliary shadows in the lung fields (figure 1). Echocardiography showed trivial tricuspid regurgitation with normal biventricular function. Gastric lavage (GL) for acid fast bacilli (AFB) was negative on three occasions. Blood, urine and CSF cultures were sterile. He was treated with pipercillin-tazobactum and ciprofloxacin. Respiratory distress further increased and required oxygen supplementation. On eighth day of admission, he developed severe respiratory distress requiring invasive ventilatory support. Hematological test showed thrombocytopenia. Antibiotics were upgraded to meropenem, vancomycin and second line anti-tuberculous treatment (ATT) in form of amikacin and ofloxacin was added in view of hepatitis but he continued to deteriorate. Endotracheal secretion was sent for GeneXpert test which showed presence of MTB which showed RR. Additional anti-tubercular drugs were added consisting of linezolid and clarithromycin. However the child succumbed to respiratory failure. Mother’s chest radiograph was suggestive of pulmonary TB with GeneXpert on sputum showed presence of MTB that was RR.
Figure 1. Child’s Chest radiogram.
| | | | Discussion | Transmission of TB to the fetus can be either hematological (transplacental) or by aspiration and/or ingestion of infected amniotic fluid. The clinical manifestations of the disease in the neonate are often non-specific (in utero or intrapartum).5 Signs and symptoms appear after first 3 weeks of life at a median age of 28 days (range: 1 to 84 days). Our patient presented to us on Day 21 of life though his symptoms had started by Day 12 of life. The common presenting signs are respiratory distress (77%), hepatic and/or splenic enlargement.6 Our patient also had respiratory distress with hepatomegaly and elevated liver transaminases. The diagnostic criteria of congenital TB were revised by Cantwell which included a proven tuberculous lesion plus one of the following: (1) lesions occurring in the first week of life, (2) primary hepatic complex or caseating hepatic granulomas, (3) maternal genital tract or placental TB, or (4) exclusion of postnatal transmission by thorough investigation of contacts.5 Our patient had a bacteriological confirmed TB and mother was subsequently detected to have pulmonary TB suggestive of congenital mode of inheritance in the child.
The mortality rate is very high, nearly 50%, and is usually due to delayed diagnosis.2 Mantoux test is rarely positive. Diagnosis is confirmed based on positive smear and/or culture results obtained from gastric washing, liver biopsy, lymph node biopsy, spinal fluid, ear discharge or polymerase chain reaction studies. Endometrial biopsy is useful in diagnosis of genital tuberculosis in the mother.
World Health Organization (WHO) recommends feeding under all circumstances for all patients with congenital TB; however, close contact with the baby should be reduced. No specific treatment regimens for congenital TB are advised. For drug-sensitive TB, treatment includes isoniazid, rifampicin, ethambutol and kanamycin or amikacin for the first two months followed by isoniazid and rifampicin for 6-12 months or similar to miliary tuberculosis or isoniazid, rifampicin and pyrazinamide along with streptomycin and kanamycin for 9 to 12 months.4,7 For treatment of early onset DR-TB, the treatment remains challenging.
| | | | Compliance with Ethical Standards | | Funding None | | | | Conflict of Interest None | | |
- Saramba MI, Zhao D. A Perspective of the Diagnosis and Management of Congenital Tuberculosis. J Pathog. 2016;2016:8623825. [CrossRef]
- Abalain ML, Petsaris O, Héry-Arnaud G, Marcorelles P, Couturaud F, Dobrzynski M, et al. Fatal congenital tuberculosis due to a Beijing strain in a premature neonate. J Med Microbiol. 2010;59:733 [CrossRef]
- Espiritu N, Aguirre L, Jave O, Sanchez L, Kirwan DE, Gilman RH. Congenital transmission of multidrug-resistant tuberculosis. Am J Trop Med Hyg. 2014;91:92-95. [CrossRef]
- Kulhari S, Agrawal R, Choudhary D, Babeeta. Congenital transmission of multidrug-resistant tuberculosis: case report. Indian J Med Case Reports. 2016;5:31-34.
- Cantwell MF, Shehab ZM, Costello AM, Sands L, Green WF, Ewing EP Jr, et al. Brief report: congenital tuberculosis. N Engl J Med. 1994;330:1051-1054. [CrossRef]
- Diar H, Velaphi S. Congenital tuberculosis as a proxy to maternal tuberculosis: a case report. J Perinatol. 2009;29:709-711. [CrossRef]
- Tomar R, Gupta A, Prasad TR, Bhalla P, Congenital Tuberculosis. Med J Armed Forces India. 2008;64:78-80. [CrossRef]
DOI: https://doi.org/10.7199/ped.oncall.2022.20
|
| Cite this article as: | | Meshram H. Congenital Drug Resistant Tuberculosis in a Neonate. Pediatr Oncall J. 2022;19. doi: 10.7199/ped.oncall.2022.20 |
|